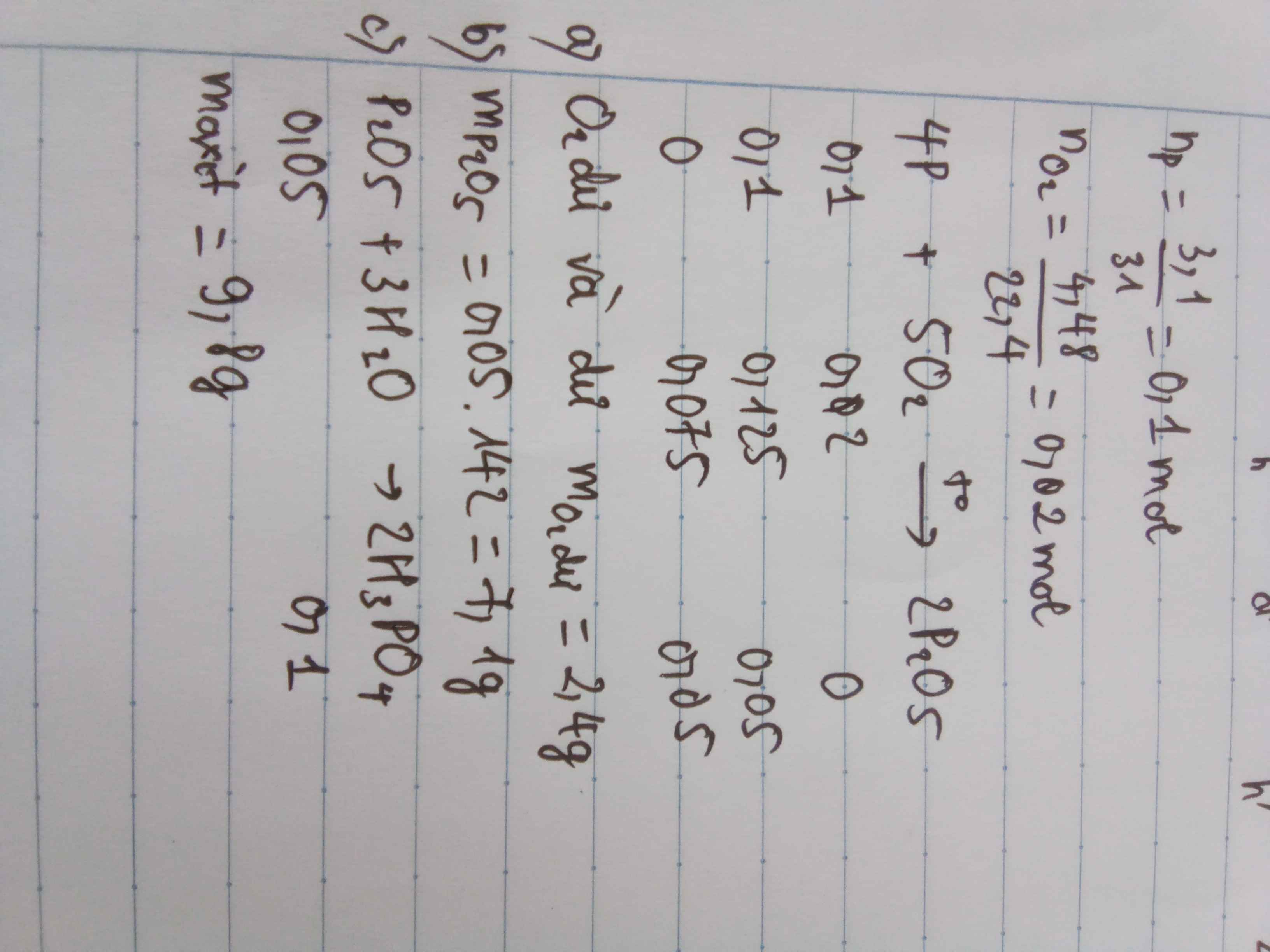Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

nH2 = 0,336/22,4 = 0,015 (mol)
PTHH: 2A + 6HCl -> 2ACl3 + 3H2
nACl3 = nA = 0,015 : 3 . 2 = 0,01 (mol)
M(A) = 0,27/0,01 = 27 (g/mol)
=> A là Al
mAlCl3 = 0,01 . 133,5 = 1,335 (g)
Câu 6.
\(n_{H_2}=\dfrac{0,336}{22,4}=0,015mol\)
\(2A+6HCl\rightarrow2ACl_3+3H_2\)
0,01 0,015
\(\overline{M_A}=\dfrac{0,27}{0,01}=27đvC\)
\(\Rightarrow A\) là Al nhôm.
\(m_{AlCl_3}=0,01\cdot133,5=1,335g\)

\(n_{Mg}=\frac{m}{M}=\frac{9,6}{24}=0,4mol\)
PTHH: \(Mg+H_2SO_4\rightarrow MgSO_4+H_2\uparrow\)
1 : 1 : 1 : 1 mol
0,4 0,4 0,4 0,4 mol
a. \(m_{MgSO_4}=n.M=0,4.\left(24+32+16.4\right)=48g\)
b. \(V_{H_2}=n.22,4=0,4.22,4=8,96l\)
c. \(n_{Fe_2O_3}=\frac{m}{M}=\frac{64}{56.2}+16.3=0,4mol\)
PTHH: \(3H_2+Fe_{2O_3}\rightarrow2Fe+3H_2O\left(ĐK:t^o\right)\)
3 : 1 : 2 : 3 mol
1, 7 0,4 0,8 1,2 mol
\(m_{Fe}=n.M=0,8.56=44,8g\)

\(n_P=\dfrac{3,1}{31}=0,1\left(mol\right)\)
\(n_{O_2}=\dfrac{4,48}{22,4}=0,2mol\)
4P + 5O2 \(\underrightarrow{t^o}\) 2P2O5
\(\dfrac{0,1}{4}< \dfrac{0,2}{5}\) => O2 dư, Photpho đủ
\(n_{O_2}=0,2-0,04=0,16\left(mol\right)\)
\(m_{P_2O_5}=\) 0,05 . 142 = 7,1 ( g )

a, \(n_{Mg}=\dfrac{2,4}{24}=0,1\left(mol\right)\)
\(m_{HCl}=36,5.15\%=5,475\left(g\right)\Rightarrow n_{HCl}=\dfrac{5,475}{36,5}=0,15\left(mol\right)\)
PT: \(Mg+2HCl\rightarrow MgCl_2+H_2\)
Xét tỉ lệ: \(\dfrac{0,1}{1}>\dfrac{0,15}{2}\), ta được Mg dư.
Theo PT: \(n_{H_2}=\dfrac{1}{2}n_{HCl}=0,075\left(mol\right)\Rightarrow V_{H_2}=0,075.22,4=1,68\left(l\right)\)
b, \(n_{Mg\left(pư\right)}=\dfrac{1}{2}n_{HCl}=0,075\left(mol\right)\Rightarrow n_{Mg\left(dư\right)}=0,1-0,075=0,025\left(mol\right)\)
\(\Rightarrow m_{Mg\left(dư\right)}=0,025.24=0,6\left(g\right)\)
c, - Cách 1:
\(n_{MgCl_2}=\dfrac{1}{2}n_{HCl}=0,075\left(mol\right)\Rightarrow m_{MgCl_2}=0,075.95=7,125\left(g\right)\)
- Cách 2:
Theo ĐLBT KL, có: mMg (pư) + mHCl = mMgCl2 + mH2
⇒ mMgCl2 = 2,4 - 0,6 + 5,475 - 0,075.2 = 7,125 (g)

Bài 1)
a \(Fe_2O_3+3H_2SO_4\rightarrow Fe_2\left(SO_4\right)_3+3H_2O\)
\(n_{Fe_2O_3}=\frac{4,8}{216}\approx\text{0,02 (mol)}\)
\(Fe_2O_3+3H_2SO_4\rightarrow Fe_2\left(SO_4\right)_3+3H_2O\)
0,02 0,06
\(m_{H_2SO_4}=98\cdot0,06=5,88\left(g\right)\)
b) \(m_{Fe_2\left(SO_4\right)_3}=0,02\cdot400=\text{290.24}\left(g\right)\)
Câu 2 mai làm
Câu 2
a)\(2Al+3H_2SO_4\rightarrow Al_2\left(SO_4\right)_3+H_2\)
\(n_{Al}=\frac{5,4}{2,7}=0,2\left(mol\right)\)
\(2Al+3H_2SO_4\rightarrow Al_2\left(SO_4\right)_3+H_2\)
0,4 mol 0,6 mol 0,2 mol
\(V_{H_2}=0,2\cdot22,4=4,48\left(l\right)\)
b) \(m_{H_2SO_4}=0,6\cdot98=58,8\left(g\right)\)

PTHH: 2Al + 6HCl\(\rightarrow\) 2AlCl3 + 3H2
a) Ta có: nH2=\(\frac{3,36}{22,5}\)=0,15 (mol)
nAl=0,15 (mol)
Vì: \(\frac{nAl}{2}\)=\(\frac{0,15}{2}\)=0,075
\(\frac{nH2}{3}\)=\(\frac{0,15}{3}\)=0,05
\(\rightarrow\)0,075>0,05 \(\rightarrow\)Tính số mol theo H2
\(\rightarrow\)nAlCl3=\(\frac{2}{3}\).nH2=\(\frac{2}{3}\).0,15=0,1 (mol)
\(\text{mAlCl3=0,1.133,5=13,35 (g)}\)
b) Vì Al dư =>Chất rắn sau phản ứng là Al dư
=>mAl dư=4,05-\(\frac{2}{3}\)
.nH2.27=4,05-2,7=1,35 (g)
a)
\(n_{H_2}=\frac{V_{H_2}}{22,4}=\frac{3,36}{22,4}=0,15\left(mol\right)\)
\(n_{Al}=\frac{m_{Al}}{M_{Al}}=\frac{4,05}{27}=0,15\left(mol\right)\)
\(PTHH:Al+2HCl\rightarrow AlCl_2+H_2\)
\(Theo\) \(PTHH,\) \(ta có:\)
\(n_{AlCl_2}=n_{Al}=n_{H_2}=0,15\left(mol\right)\)
\(m_{AlCl_3}=n_{AlCl_3}.M_{AlCl_3}=0,15.133,5=20,025\left(g\right)\)
b) Là AlCl3 đó bn

a) \(n_{H_2}=\dfrac{1,344}{22,4}=0,06\left(mol\right)\)
\(n_{Cl}=n_{HCl}=2n_{H_2}=2.0,06=0,12\left(mol\right)\)
m=mkim loại+mCl=1,965+0,12.35,5=6,225(gam)
b) \(H_2+CuO\overset{t^0}{\rightarrow}Cu+H_2O\left(1\right)\)
\(yH_2+Fe_xO_y\overset{t^0}{\rightarrow}xFe+yH_2O\left(2\right)\)
Fe+H2SO4\(\rightarrow FeSO_4+H_2\left(3\right)\)
\(m_{Cu}=1,28\left(g\right)\rightarrow n_{Cu}=\dfrac{1,28}{64}=0,02\left(mol\right)\)
-Theo(1): \(n_{H_2\left(1\right)}=n_{CuO}=n_{Cu}=0,02\left(mol\right)\)
\(\rightarrow m_{CuO}=0,02.80=1,6\left(g\right)\rightarrow m_{Fe_xO_y}=3,92-1,6=2,32\left(g\right)\)
\(\rightarrow n_{H_2\left(2\right)}=n_{H_2}-n_{H_2\left(1\right)}=0,06-0,02=0,04\left(mol\right)\)
-Theo(2): \(n_{Fe_xO_y}=\dfrac{1}{y}n_{H_2}=\dfrac{0,04}{y}mol\rightarrow M_{Fe_xO_y}=\dfrac{2,32}{\dfrac{0,04}{y}}=58y\)
\(\rightarrow56x+16y=58y\rightarrow56x=42y\rightarrow\dfrac{x}{y}=\dfrac{42}{56}=\dfrac{3}{4}\)
\(\rightarrow Fe_3O_4\)
%CuO=\(\dfrac{1,6}{3,92}.100\approx40,8\%\)
%Fe3O4=100%-40,8%=59,2%