Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

\(n_{H_2}=\dfrac{V_{H_2}}{22,4}=\dfrac{2,4}{22,4}\approx0,11\left(mol\right)\)
\(n_{O_2}=\dfrac{V_{O_2}}{22,4}=\dfrac{1,6}{22,4}\approx0,07\)
\(2H_2+O_2\rightarrow2H_2O\)
2 mol-1mol---2 mol
Ta có: \(\dfrac{n_{H_2}}{2}=\dfrac{0,11}{2}\)
\(\dfrac{n_{O_2}}{1}=\dfrac{0,07}{1}\)
\(\Rightarrow\dfrac{n_{H_2}}{2}< \dfrac{n_{O_2}}{1}\)
Vậy \(O_2\) dư
Số mol O2 dư:
\(n_{O_2}=\dfrac{0,07.1}{2}=0,035\left(mol\right)\)
Khối lượng O2 dư
\(m_{O_2}=0,035.32=1,12\left(g\right)\)
Khối lượng nước thu được:
\(n_{H_2O}=\dfrac{0,07.2}{2}=0,07\left(mol\right)\)
\(\Rightarrow m_{H_2O}=n_{H_2O}.M_{H_2O}=0,07.18=1,26\left(g\right)\)

a) PTHH: 2Al + 6HCl -> 2AlCl3 + 3 H2
b) nHCl=0,6(mol); nAl=0,3(mol)
Ta có: 0,3/2 > 0,6/6
=> HCl hết, Al dư, tính theo nHCl
c) nH2= 3/6 . nHCl=3/6 . 0,6= 0,3(mol)
=> V=V(H2,đktc)=0,3.22,4= 6,72(l)

\(a) Zn + H_2SO_4 \to ZnSO_4 + H_2\\ n_{Zn} = \dfrac{13}{65} = 0,2 < n_{H_2SO_4} = \dfrac{200.24,5\%}{98} = 0,5 \to H_2SO_4\ dư\\ n_{H_2SO_4\ pư} =n_{Zn} = 0,2(mol)\\ \Rightarrow m_{H_2SO_4\ dư} = (0,5 - 0,2).98 = 29,4(gam)\\ c) n_{FeSO_4} = n_{H_2} = n_{Zn} = 0,2(mol)\\ m_{FeSO_4} = 0,2.152 = 30,4(gam)\\ V_{H_2} = 0,2.22,4 = 4,48(lít)\)

\(n_{H_2}=\dfrac{V_{H_2}}{22,4}=\dfrac{4,48}{22,4}=0,2mol\)
\(n_{Fe_2O_3}=\dfrac{m_{Fe_2O_3}}{M_{Fe_2O_3}}=\dfrac{23,2}{160}=0,145mol\)
\(Fe_2O_3+3H_2\rightarrow\left(t^o\right)2Fe+3H_2O\)
0,145 > 0,2 ( mol )
1/15 0,2 2/15 ( mol 0
Chất còn dư là \(Fe_2O_3\)
\(m_{Fe_2O_3\left(du\right)}=n_{Fe_2O_3\left(du\right)}.M_{Fe_2O_3}=\left(0,145-\dfrac{1}{15}\right).160=12,53g\)
\(m_{Fe}=n_{Fe}.M_{Fe}=\dfrac{2}{15}.56=7,4666g\)

a, \(n_{Mg}=\dfrac{2,4}{24}=0,1\left(mol\right)\)
\(m_{HCl}=36,5.15\%=5,475\left(g\right)\Rightarrow n_{HCl}=\dfrac{5,475}{36,5}=0,15\left(mol\right)\)
PT: \(Mg+2HCl\rightarrow MgCl_2+H_2\)
Xét tỉ lệ: \(\dfrac{0,1}{1}>\dfrac{0,15}{2}\), ta được Mg dư.
Theo PT: \(n_{H_2}=\dfrac{1}{2}n_{HCl}=0,075\left(mol\right)\Rightarrow V_{H_2}=0,075.22,4=1,68\left(l\right)\)
b, \(n_{Mg\left(pư\right)}=\dfrac{1}{2}n_{HCl}=0,075\left(mol\right)\Rightarrow n_{Mg\left(dư\right)}=0,1-0,075=0,025\left(mol\right)\)
\(\Rightarrow m_{Mg\left(dư\right)}=0,025.24=0,6\left(g\right)\)
c, - Cách 1:
\(n_{MgCl_2}=\dfrac{1}{2}n_{HCl}=0,075\left(mol\right)\Rightarrow m_{MgCl_2}=0,075.95=7,125\left(g\right)\)
- Cách 2:
Theo ĐLBT KL, có: mMg (pư) + mHCl = mMgCl2 + mH2
⇒ mMgCl2 = 2,4 - 0,6 + 5,475 - 0,075.2 = 7,125 (g)

\(n_K=\dfrac{3,8}{39}=\dfrac{19}{195}mol\)
\(n_{H_2O}=\dfrac{101,8}{18}=\dfrac{509}{90}mol\)
\(2K+2H_2O\rightarrow2KOH+H_2\)
19/195 < 509/90 ( mol )
19/195 19/195 19/195 ( mol )
Chất dư là H2O
\(m_{H_2O\left(dư\right)}=\left(\dfrac{509}{90}-\dfrac{19}{195}\right).18\approx100,04g\)
\(m_{KOH}=\dfrac{19}{195}.56\approx5,45g\)

pt 2Na+2H20-->2NaOH+H2
ta có nNa=3,45:23=0,15 mol
nH2O=200/18=100/9 mol
theo pt thì H2O dư
theo pt nNAOH=nNa=0,15 mol \(\Rightarrow\)mNAOH=0,15*40=6mol
theo pt nH2=1/2nNa=0,075 mol\(\Rightarrow\)mh2=0,15 mol
mdd sau pứ =3,45+200-0,15=203,3 mol
C%NaOH=6:203,3*100%=3%
CÂU C MÌNH K BIẾT ĐÚNG KHÔNG NỮA ....
c,
mdd sau phản ứng=mNa+mH20-mH2
=3,45+200-0,075.2=203,3g
=>C%=(0,15.40).100/203,3=2,95%
Vậy C%=2,95%
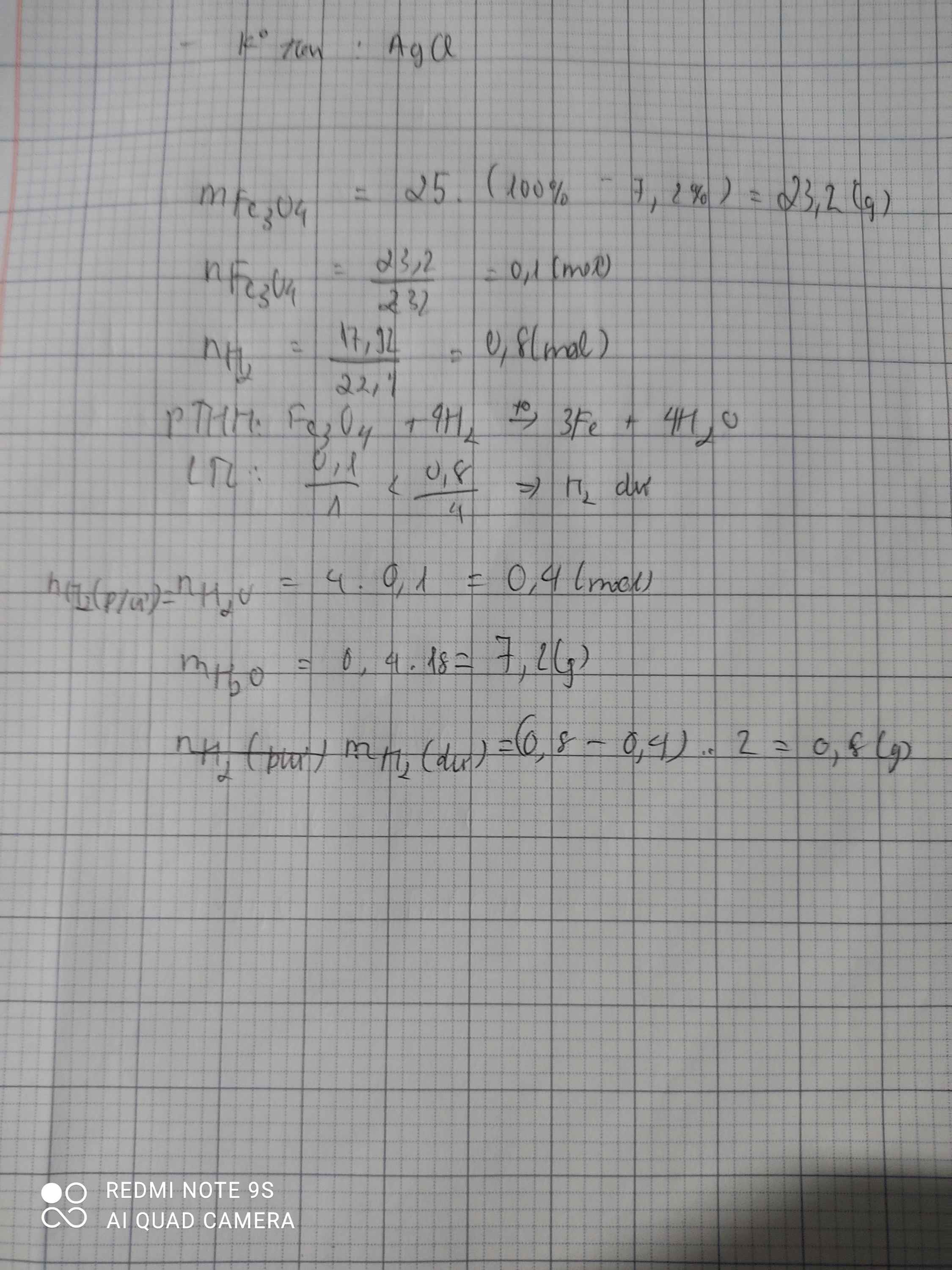

Ta có:
\(n_{Na_2O}=\dfrac{6,2}{62}=0,1\left(mol\right)\)
\(n_{H_2O}=\dfrac{3,6}{18}=0,2\left(mol\right)\left(phần.này.mik.sửa.lại.đề\right)\)
a. \(PTHH:Na_2O+H_2O--->2NaOH\)
Ta thấy: \(\dfrac{0,1}{1}< \dfrac{0,2}{1}\)
Vậy H2O dư.
Theo PT: \(n_{NaOH}=2.n_{Na_2O}=2.0,1=0,2\left(mol\right)\)
\(\Rightarrow m_{NaOH}=40.0,2=8\left(g\right)\)
b. Ta có: \(n_{H_2O_{PỨ}}=n_{Na_2O}=0,1\left(mol\right)\)
\(\Rightarrow n_{H_2O_{dư}}=0,2-0,1=0,1\left(mol\right)\)
\(\Rightarrow m_{H_2O_{dư}}=0,1.18=1,8\left(g\right)\)
onwcamr ơn nha