Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

a)
\(n_{FeSO_4.7H_2O}=\dfrac{41,7}{278}=0,15\left(mol\right)\)
=> \(n_{FeSO_4}=0,15\left(mol\right)\)
=> \(m_{FeSO_4}=0,15.152=22,8\left(g\right)\)
b) mdd sau pha trộn = 41,7 + 207 = 248,7 (g)
c) \(C\%=\dfrac{22,8}{248,7}.100\%=9,168\%\)
\(V_{dd}=\dfrac{248,7}{1,023}=243,1085\left(ml\right)=0,2431085\left(l\right)\)
\(C_M=\dfrac{0,15}{0,2431085}=0,617M\)

Câu 6:
\(m_{dd.bđ}=1,1.200=220\left(g\right)\)
\(n_{FeSO_4.7H_2O}=\dfrac{83,4}{278}=0,3\left(mol\right)\Rightarrow n_{FeSO_4}=0,3\left(mol\right)\)
=> \(C\%_{dd.bđ}=\dfrac{0,3.152}{220}.100\%=20,73\%\)
Câu 7:
\(m_{MgCl_2\left(dd.ở.60^oC\right)}=\dfrac{500.37,5}{100}=187,5\left(g\right)\)
=> \(m_{H_2O}=500-187,5=312,5\left(g\right)\)
Giả sử có a mol MgCl2.6H2O tách ra
\(n_{MgCl_2\left(dd.ở.10^oC\right)}=\dfrac{187,5}{95}-a=\dfrac{75}{38}-a\left(mol\right)\)
=> \(m_{MgCl_2\left(dd.ở.10^oC\right)}=95\left(\dfrac{75}{38}-a\right)=187,5-95a\left(g\right)\)
\(n_{H_2O\left(tách.ra\right)}=6a\left(mol\right)\)
\(m_{H_2O\left(dd.ở.10^oC\right)}=312,5-18.6a\)=312,5 - 108a (g)
=> \(S_{10^oC}=\dfrac{187,5-95a}{312,5-108a}.100=53\left(g\right)\)
=> \(a=\dfrac{4375}{7552}\left(mol\right)\)
=> \(m_{MgCl_2.6H_2O}=\dfrac{4375}{7552}.203=117,6\left(g\right)\)

a, \(n_K=\dfrac{3,9}{39}=0,1\left(mol\right)\)
PTHH: 2K + 2H2O ---> 2KOH + H2
0,1---------------->0,1----->0,05
\(m_{ct}=m_{KOH}=0,1.56=5,6\left(g\right)\\ m_{dd}=m_K+m_{H_2O}-m_{H_2}=96,2+3,9-0,05.2=100\left(g\right)\)
\(C\%_{KOH}=\dfrac{5,6}{100}.100\%=5,6\%\\ b,m_{dd}=100+50=150\left(g\right)\\ C\%_{KOH}=\dfrac{5,6}{150}.100\%=3,37\%\)
c, Gọi \(m_{H_2O}=a\left(g\right)\)
\(\Rightarrow C\%_{KOH}=\dfrac{5,6}{100+a}.100\%=2,8\%\\ \Leftrightarrow a=100\left(g\right)\)
d, Gọi \(m_{KOH}=a\left(g\right)\)
\(\Rightarrow C\%_{KOH}=\dfrac{5,6+a}{100+a}.100\%=22,4\%\\ \Leftrightarrow a=21,65\left(g\right)\)

\(a,m_{ct}=30\left(g\right)\\ m_{dm}=120\left(g\right)\\ m_{dd}=120+30=150\left(g\right)\\ b,C\%_{đường}=\dfrac{30}{150}.100\%=20\%\\ c,C\%_{đường}=\dfrac{30}{150+50}.100\%=15\%\)
\(d,m_{dd}=\dfrac{30}{10\%}=300\left(g\right)\\ m_{H_2O\left(thêm\right)}=300-150=150\left(g\right)\)
e, Gọi \(m_{đường\left(thêm\right)}=a\left(g\right)\)
\(\Rightarrow C\%=\dfrac{30+a}{150+a}.100\%=30\%\\ \Leftrightarrow a=21,4285\left(g\right)\)

a)
\(n_{K_2O}=\dfrac{18,8}{94}=0,2\left(mol\right)\)
PTHH: K2O + H2O --> 2KOH
0,2-->0,2---->0,4
mct = 0,4.56 = 22,4 (g)
mdm = 81,2 - 0,2.18 = 77,6 (g)
mdd = 22,4 + 77,6 = 100 (g)
b)
\(C\%=\dfrac{22,4}{100}.100\%=22,4\%\)
c)
\(C\%=\dfrac{22,4}{50+100}.100\%=14,933\%\)
d)
\(m_{dd\left(sau.khi.thêm\right)}=\dfrac{22,4.100}{11,2}=200\left(g\right)\)
=> mH2O(thêm) = 200 - 100 = 100 (g)
e) Gọi khối lượng KOH thêm là x (g)
Có: \(C\%_{\left(dd.sau.khi.thêm\right)}=\dfrac{22,4+x}{100+x}.100\%=30\%\)
=> x = 10,857 (g)

\(n_{NaCl\left(tv\right)}=\dfrac{29.25}{58.5}=0.5\left(mol\right)\)
\(n_{NaCl\left(bđ\right)}=0.15\cdot0.5=0.075\left(mol\right)\)
\(\Rightarrow n_{NaCl}=0.5+0.075=0.575\left(mol\right)\)
\(C_{M_{NaCl}}=\dfrac{0.575}{0.252}=2.3\left(M\right)\)
a ơi ở phần tính mol NaCl ban đầu 2 số đấy từ đâu ra vậy ạ?

a/
\(n_{Na_2O}=\dfrac{9,3}{62}=0,15\left(mol\right)\)
\(Na_2O+H_2O\rightarrow2NaOH\)
0,15 0,3 (mol)
\(m_{NaOH}=0,3.40=12\left(g\right)\)
\(m_A=90,7+9,3=100\left(g\right)\)
\(C\%_{NaOH}=\dfrac{12}{100}.100\%=12\%\)
b/
m\(_{FeSO_4}=\dfrac{16.200}{100}=32\left(g\right)\)
\(\rightarrow m_{FeSO_4}=\dfrac{32}{152}=\dfrac{4}{19}\left(mol\right)\)
\(2NaOH+FeSO_4\rightarrow Na_2SO_4+Fe\left(OH\right)_2\downarrow\)
bđ: 0,3 \(\dfrac{4}{19}\) 0 0 (mol)
pư: 0,3 0,15 0,15 0,15 (mol)
dư: 0 \(\dfrac{23}{380}\) (mol)
\(m_{Fe\left(OH\right)_2}=0,15.90=13,5\left(g\right)\)
\(m_C=100+200-13,5=286,5\left(g\right)\)
\(m_{Na_2SO_4}=0,15.142=21,3\left(g\right)\)
\(\rightarrow C\%_{Na_2SO_4}=\dfrac{21,3}{286,5}.100\%\approx7,4\%\)
\(m_{FeSO_4\left(dư\right)}=\dfrac{23}{380}.152=9,2\left(g\right)\)
\(\rightarrow C\%_{FeSO_4\left(dư\right)}=\dfrac{9,2}{286,5}.100\%\approx3,2\%\)

a, \(C\%_{KCl}=\dfrac{20}{20+60}.100\%=25\%\)
b, \(C\%=\dfrac{40}{40+150}.100\%\approx21,05\%\)
c, \(C\%_{NaOH}=\dfrac{60}{60+240}.100\%=20\%\)
d, \(C\%_{NaNO_3}=\dfrac{30}{30+90}.100\%=25\%\)
e, \(m_{NaCl}=150.60\%=90\left(g\right)\)
f, \(m_{ddA}=\dfrac{25}{10\%}=250\left(g\right)\)
g, \(n_{NaOH}=120.20\%=24\left(g\right)\)
Gọi: nNaOH (thêm vào) = a (g)
\(\Rightarrow\dfrac{a+24}{a+120}.100\%=25\%\Rightarrow a=8\left(g\right)\)
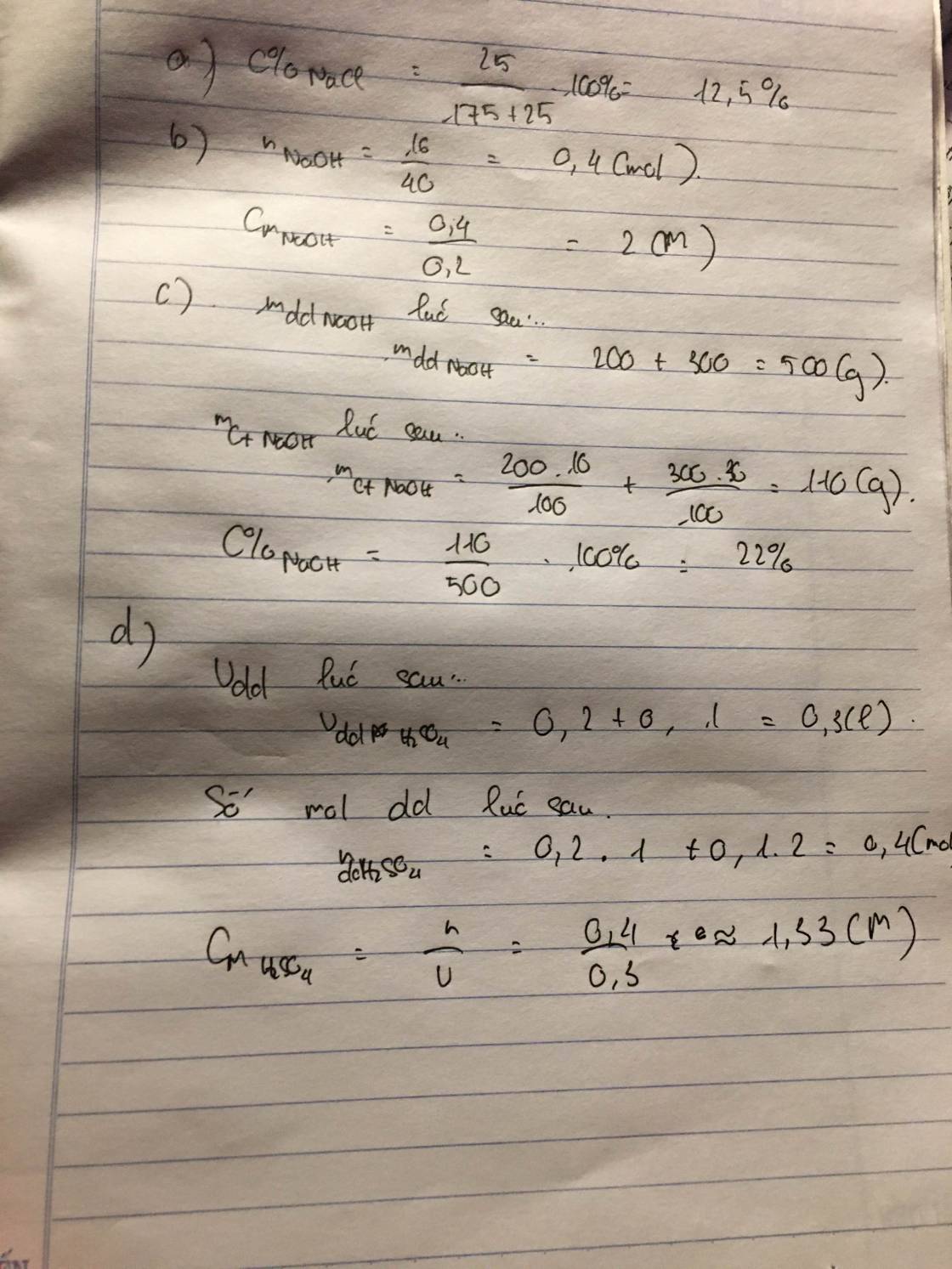
\(m_{ddFeSO_4\left(sau\right)}=50+150=200\left(g\right)\\ m_{FeSO_4\left(tổng\right)}=\dfrac{152}{278}.50+150.9\%\approx40,84\left(g\right)\\ C\%_{ddFeSO_4}\approx\dfrac{40,84}{200}.100\%\approx20,42\%\)