Giải bài 16 từ câu số 15 đến 45 giùm mik nha mọi ng
Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.


Câu 15: (mãi mới nghĩ ra :v)
\(\dfrac{\left(a+b\right)^2}{ab}+\dfrac{\left(b+c\right)^2}{bc}+\dfrac{\left(c+a\right)^2}{ca}\ge9+2\left(\dfrac{a}{b+c}+\dfrac{b}{c+a}+\dfrac{c}{a+b}\right)\)
\(\Leftrightarrow\dfrac{a^2+2ab+b^2}{ab}+\dfrac{b^2+2bc+b^2}{bc}+\dfrac{c^2+2ca+a^2}{ca}\ge9+2\left(\dfrac{a}{b+c}+\dfrac{b}{c+a}+\dfrac{c}{a+b}\right)\)
\(\Leftrightarrow\dfrac{a}{b}+2+\dfrac{b}{a}+\dfrac{b}{c}+2+\dfrac{c}{b}+\dfrac{c}{a}+2+\dfrac{a}{c}\ge9+2\left(\dfrac{a}{b+c}+\dfrac{b}{c+a}+\dfrac{c}{a+b}\right)\)
\(\Leftrightarrow\dfrac{a}{b}+\dfrac{b}{a}+\dfrac{b}{c}+\dfrac{c}{b}+\dfrac{c}{a}+\dfrac{a}{c}\ge3+2\left(\dfrac{a}{b+c}+\dfrac{b}{c+a}+\dfrac{c}{a+b}\right)\)
\(\Leftrightarrow a\left(\dfrac{1}{b}+\dfrac{1}{c}\right)+b\left(\dfrac{1}{c}+\dfrac{1}{a}\right)+c\left(\dfrac{1}{a}+\dfrac{1}{b}\right)\ge3+2\left(\dfrac{a}{b+c}+\dfrac{b}{c+a}+\dfrac{c}{a+b}\right)\)
-Áp dụng BĐT Caushy Schwarz ta có:
\(\left\{{}\begin{matrix}\dfrac{1}{b}+\dfrac{1}{c}\ge\dfrac{\left(1+1\right)^2}{b+c}=\dfrac{4}{b+c}\\\dfrac{1}{c}+\dfrac{1}{a}\ge\dfrac{\left(1+1\right)^2}{c+a}=\dfrac{4}{c+a}\\\dfrac{1}{a}+\dfrac{1}{b}\ge\dfrac{\left(1+1\right)^2}{a+b}=\dfrac{4}{a+b}\end{matrix}\right.\)
-Từ đó suy ra: \(a\left(\dfrac{1}{b}+\dfrac{1}{c}\right)+b\left(\dfrac{1}{c}+\dfrac{1}{a}\right)+c\left(\dfrac{1}{a}+\dfrac{1}{b}\right)\ge\dfrac{4a}{b+c}+\dfrac{4b}{c+a}+\dfrac{4c}{a+b}\)
-Ta c/m rằng: \(\dfrac{4a}{b+c}+\dfrac{4b}{c+a}+\dfrac{4c}{a+b}\ge3+2\left(\dfrac{a}{b+c}+\dfrac{b}{c+a}+\dfrac{c}{a+b}\right)\)
\(\Leftrightarrow2\left(\dfrac{a}{b+c}+\dfrac{b}{c+a}+\dfrac{c}{a+b}\right)+2\left(\dfrac{a}{b+c}+\dfrac{b}{c+a}+\dfrac{c}{a+b}\right)\ge3+2\left(\dfrac{a}{b+c}+\dfrac{b}{c+a}+\dfrac{c}{a+b}\right)\)
\(\Leftrightarrow2\left(\dfrac{a}{b+c}+\dfrac{b}{c+a}+\dfrac{c}{a+b}\right)\ge3\)
\(\Leftrightarrow2\left(\dfrac{a}{b+c}+1+\dfrac{b}{c+a}+1+\dfrac{c}{a+b}+1-3\right)\ge3\)
\(\Leftrightarrow2\left(\dfrac{a+b+c}{b+c}+\dfrac{b+c+a}{c+a}+\dfrac{c+a+b}{a+b}\right)-6\ge3\)
\(\Leftrightarrow2\left(a+b+c\right)\left(\dfrac{1}{b+c}+\dfrac{1}{c+a}+\dfrac{1}{a+b}\right)\ge9\left(1\right)\)
-Áp dụng BĐT Caushy Schwarz cho VT của BĐT ta được:
\(2\left(a+b+c\right)\left(\dfrac{1}{b+c}+\dfrac{1}{c+a}+\dfrac{1}{a+b}\right)\ge2\left(a+b+c\right)\left(\dfrac{\left(1+1+1\right)^2}{a+b+b+c+c+a}\right)=2\left(a+b+c\right)\dfrac{9}{2\left(a+b+c\right)}=9\)
\(\Rightarrow\)BĐT (1) đúng.
\(\Rightarrowđpcm\)
-Dấu "=" xảy ra khi \(a=b=c\)

52+16<x9<45+25
=>68<x9<70
=>7.9+5<x9<7.9+7 mà 7.9+5<7.9+6<7.9+7
Nên 7.9+6=x9
69=x9
x=69/9=23/3
Vậy x=23/3


Bài 9
Công
\(A=F.s=100.30000=3000000J\)
Công suất
\(P=\dfrac{A}{t}=\dfrac{3000000}{40\cdot60}=1250W\)
Bài 10
Hiệu suất
\(H=\dfrac{A_i}{A_{tp}}=\dfrac{55.10.1,5}{275.5}.100\%=60\%\)
Lực cản
\(F_c=\dfrac{A_{tp}-A_i}{l}=110N\)
Bài 11
Lực kéo
\(F=\dfrac{P}{s}=\dfrac{500000}{10}=50000N\)
Công thực hiện trong 120s
\(A=P.t=500000.120=60000kJ\)
Bài 12)
Nl đun sôi nước là
\(Q=Q_1+Q_2=m_1c_1\Delta t+m_2c_2\Delta t\\ =0,3.880+2,5.4200\left(100-30\right)=753480J\)
Bài 13)
Ta có ptcbn
\(Q_{thu}=Q_{tỏa}\\ =2,5.4200\left(30-t\right)=0,6.380\left(100-30\right)\\ \Rightarrow t=28,48^o\)
--> nước nóng lên 1,52 oC
Bài 14)
Công
\(A=F.s=2000.3000=6000000J\)
Công suất
\(P=\dfrac{A}{t}=\dfrac{6000000}{10.60}=10kW\)

Hồi hôm qua bạn đã làm cho mình bài 1 và bài 10 rồi bây giờ bạn chịu khó làm cho mình những câu còn lại đi nha. Cám ơn bạn.![]()
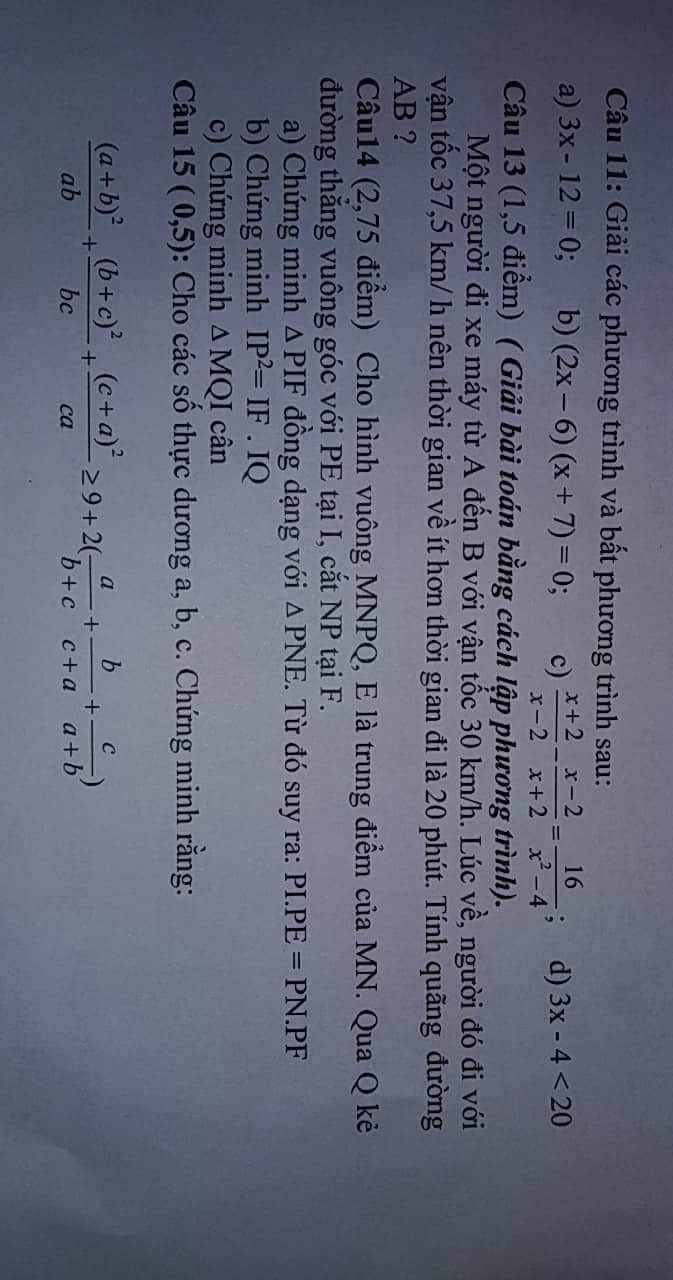




\(15)2K + 2H_2O \to 2KOH + H_2\\ 16) 2Na + 2H_2O \to 2NaOH + H_2\\ 17) Ca + 2H_2O \to Ca(OH)_2 + H_2\\ 18) Ba + 2H_2O \to Ba(OH)_2 + H_2\\ 19) Mg + 2AgNO_3 \to Mg(NO_3)_2 + 2Ag\\ 20) 2Al + 3CuCl_2 \to 3Cu + 2AlCl_3\\ 21) 2Al + 3H_2SO_4 \to Al_2(SO_4)_3 + 3H_2\\ 22) Zn + 2HCl \to ZnCl_2 + H_2\\ 23) Fe + 2HCl \to FeCl_2 + H_2\\ 24) 2Al + 2H_3PO_4 \to 2AlPO_4 + 3H_2\\ 25) Fe_2O_3 + 6HCl \to 2FeCl_3 + 3H_2O\\ 26) CuO + 2HCl \to CuCl_2 + H_2O\\ 27) 2Fe + 3Cl_2 \xrightarrow{t^o} 2FeCl_3\\\)
\(28) 4Cr + 3O_2 \xrightarrow{t^o} 2Cr_2O_3\\ 29) 2Fe + 3Br_2 \xrightarrow{t^o} 2FeBr_3\\ 30) 2H_2 + O_2 \xrightarrow{t^o} 2H_2O\\ 31) 2C_2H_2 + 5O_2 \xrightarrow{t^o} 4CO_2 + 2H_2O\\ 32) C_2H_4 + 3O_2 \xrightarrow{t^o} 2CO_2 + 2H_2O\\ 33) CH_4 + 2O_2 \xrightarrow{t^o} CO_2 + 2H_2O\\ 34) BaCl_2 + 2AgNO_3 \to 2AgCl + Ba(NO_3)_2\\ 35) 6NaOH + Fe_2(SO_4)_3 \to 2Fe(OH)_3 + 3Na_2SO_4\\ 36) Na_2CO_3 + Ca(OH)_2 \to CaCO_3 + 2NaOH\\ 37) 2HCl +CaCO_3 \to CaCl_2 + CO_2 + H_2O\\ 38) CuCl_2 + 2AgNO_3 \to 2AgCl + Cu(NO_3)_2\\ 39) Na_2SO_4 + BaCl_2 \to BaSO_4 + 2NaCl\\ 40) H_2 + CuO \xrightarrow{t^o} Cu + H_2O\\ 41) 3H_2 + Fe_2O_3 \xrightarrow{t^o} 2Fe + 3H_2O\\ 42) H_2 + FeO \xrightarrow{t^o} Fe + H_2O\\ 43) 4H_2 + Fe_3O_4 \xrightarrow{t^o}3Fe + 4H_2O\\ 44) H_2 + PbO \xrightarrow{t^o} Pb + H_2O\\ 45) 2Al + 3CuO \xrightarrow{t^o} Al_2O_3 + 3Cu\)