8, Khử oxit sắt từ ( Fe3O4 ) bằng khí hidro ở nhiệt độ cao thu được 30,24g sắt. Tính khối lượng oxit sắt từ cần dùng
Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.


a, \(Fe_3O_4+4H_2\underrightarrow{t^o}3Fe+4H_2O\)
b, \(n_{Fe}=\dfrac{11,2}{56}=0,2\left(mol\right)\)
Theo PT: \(n_{Fe_3O_4}=\dfrac{1}{3}n_{Fe}=\dfrac{1}{15}\left(mol\right)\Rightarrow m_{Fe_3O_4}=\dfrac{1}{15}.232=\dfrac{232}{15}\left(g\right)\)
c, \(n_{H_2}=\dfrac{4}{3}n_{Fe}=\dfrac{4}{15}\left(mol\right)\Rightarrow V_{H_2}=\dfrac{4}{15}.22,4=\dfrac{448}{75}\left(l\right)\)
d, \(Zn+2HCl\rightarrow ZnCl_2+H_2\)
\(n_{Zn}=n_{H_2}=\dfrac{4}{15}\left(mol\right)\Rightarrow m_{Zn}=\dfrac{4}{15}.65=\dfrac{52}{3}\left(g\right)\)
\(n_{HCl}=2n_{H_2}=\dfrac{8}{15}\left(mol\right)\Rightarrow m_{HCl}=\dfrac{8}{15}.36,5=\dfrac{292}{15}\left(g\right)\)

a) \(n_{Fe}=\dfrac{3,36}{56}=0,06\left(mol\right)\)
PTHH: 3Fe + 2O2 --to--> Fe3O4
0,06->0,04------->0,02
=> mFe3O4 = 0,02.232 = 4,64 (g)
b) VO2 = 0,04.22,4 = 0,896 (l)

n Fe3O4=\(\dfrac{4,64}{232}=0,02mol\)
3Fe+2O2-to>Fe3O4
0,06----0,04---0,02
=>m O2=0,04.32=1,28g
\(n_{Fe_3O_4}=\dfrac{m}{M}=\dfrac{4,64}{232}=0,02mol\)
\(3Fe+2O_2\rightarrow\left(t^o\right)Fe_3O_4\)
0,06 0,02 ( mol )
\(m_{Fe}=n_{Fe}.M_{Fe}=0,06.56=3,36g\)

a. \(n_{Fe_3O_4}=\dfrac{6,96}{232}=0,03\left(mol\right)\)
PTHH : 3Fe + 2O2 -to-> Fe3O4
0,09 0,06 0,03
\(m_{Fe}=0,09.56=5,04\left(g\right)\)
\(V_{O_2}=0,06.22,4=1,344\left(l\right)\)
b. PTHH : 2KCl + 3O2 -> 2KClO3
0,06 0,04
\(m_{KClO_3}=0,04.122,5=4,9\left(g\right)\)

Bài 3:
\(n_{H_2}=\dfrac{8,96}{22,4}=0,4\left(mol\right)\)
a, PTHH: Fe3O4 + 4H2 --to--> 3Fe + 4H2O
0,1<------0,4
Zn + 2HCl ---> ZnCl2 + H2
0,4<-------------------------0,4
b, mFe3O4 = 0,1.232 = 23,2 (g)
c, mZn = 0,4.65 = 26 (g)
Bài 4:
\(n_{Zn}=\dfrac{6,5}{65}=0,1\left(mol\right)\)
a, PTHH: Zn + 2HCl ---> ZnCl2 + H2
0,1---->0,2---------------->0,1
b, VH2 = 0,1.22,4 = 2,24 (l)
c, \(C_{M\left(HCl\right)}=\dfrac{0,2}{0,2}=1M\)

\(n_{Fe_2O_3}=\dfrac{16}{160}=0,1\left(mol\right)\)
PTHH: Fe2O3 + 3H2 --to--> 2Fe + 3H2O
0,1 0,3 0,2
\(V_{H_2}=0,3.22,4=6,72\left(l\right)\)
\(n_{O_2}=\dfrac{2,24}{22,4}=0,1\left(mol\right)\)
PTHH: 3Fe + 2O2 --to--> Fe3O4
LTL: \(\dfrac{0,2}{3}>\dfrac{0,1}{2}\rightarrow\) Fe dư
Theo pthh: \(n_{Fe\left(pư\right)}=\dfrac{3}{2}n_{O_2}=\dfrac{3}{2}.0,1=0,15\left(mol\right)\)
\(\rightarrow m_{Fe\left(dư\right)}=\left(0,2-0,15\right).56=2,8\left(g\right)\)
a.\(n_{Fe_2O_3}=\dfrac{16}{160}=0,1mol\)
\(Fe_2O_3+3H_2\rightarrow\left(t^o\right)2Fe+3H_2O\)
0,1 0,3 0,2 ( mol )
\(V_{H_2}=0,3.22,4=6,72l\)
b.\(n_{O_2}=\dfrac{2,24}{22,4}=0,1mol\)
\(3Fe+2O_2\rightarrow\left(t^o\right)Fe_3O_4\)
\(\dfrac{0,2}{3}\) > \(\dfrac{0,1}{2}\) ( mol )
0,15 0,1 ( mol )
Chất dư là Fe
\(m_{Fe\left(dư\right)}=\left(0,2-0,15\right).56=2,8g\)

a, \(3Fe+2O_2\underrightarrow{t^o}Fe_3O_4\)
b, \(n_{Fe}=\dfrac{16,8}{56}=0,3\left(mol\right)\)
Theo PT: \(n_{O_2}=\dfrac{2}{3}n_{Fe}=0,2\left(mol\right)\Rightarrow V_{O_2}=0,2.22,4=4,48\left(l\right)\)
\(n_{Fe_3O_4}=\dfrac{1}{3}n_{Fe}=0,1\left(mol\right)\Rightarrow m_{Fe_3O_4}=0,1.232=23,2\left(g\right)\)
c, \(n_{CuO}=\dfrac{16}{80}=0,2\left(mol\right)\)
PT: \(CuO+H_2\underrightarrow{t^o}Cu+H_2O\)
Xét tỉ lệ: \(\dfrac{0,2}{1}< \dfrac{0,3}{1}\), ta được H2 dư.
Theo PT: \(n_{Cu}=n_{CuO}=0,2\left(mol\right)\Rightarrow m_{Cu}=0,2.64=12,8\left(g\right)\)
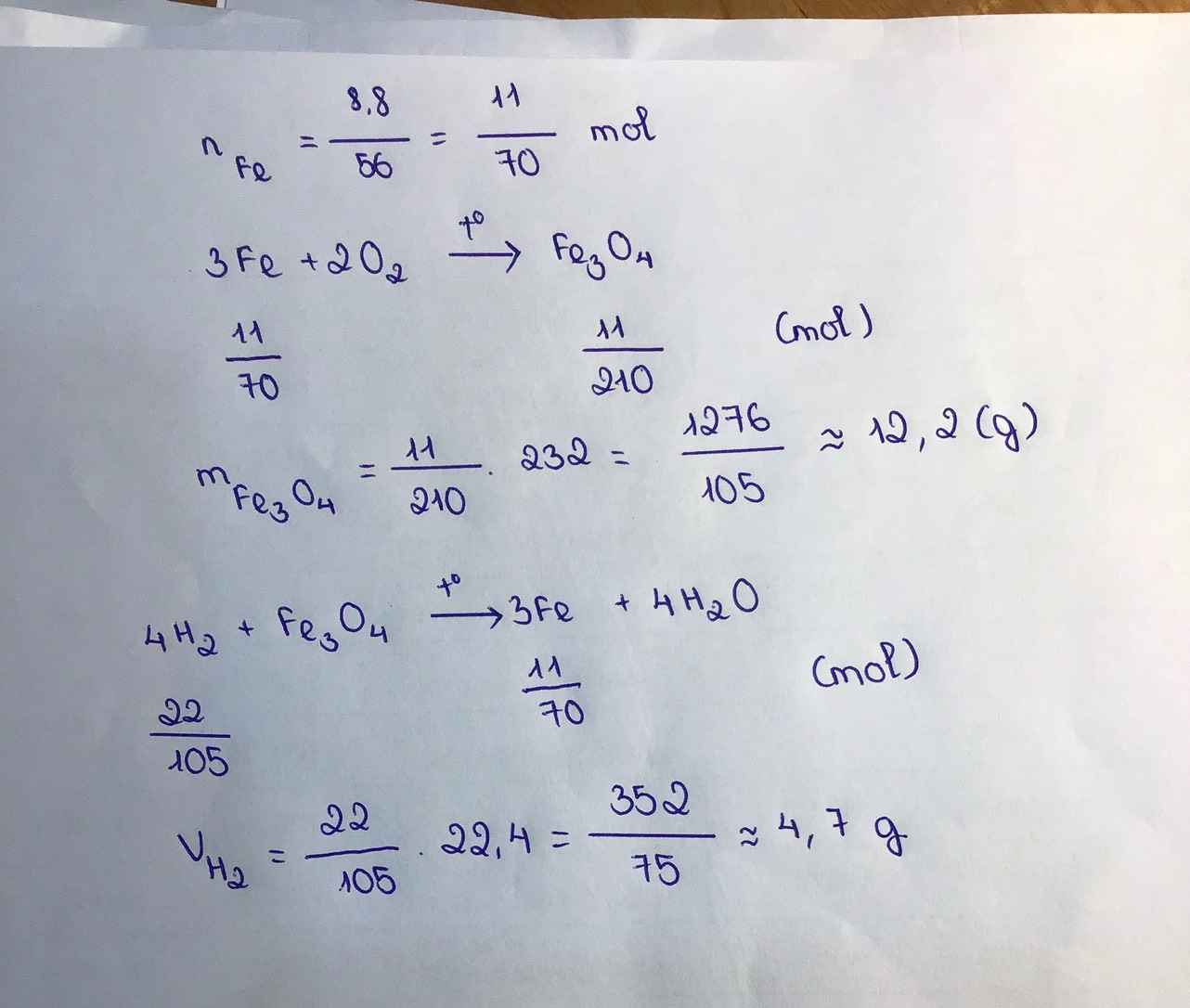
\(PTHH:Fe_3O_4+4H_2\rightarrow^{t^o}3Fe+4H_2O\\ n_{Fe}=\dfrac{30,24}{56}=0,54\left(mol\right)\\ \Rightarrow n_{Fe_3O_4}=\dfrac{1}{3}n_{Fe}=0,18\left(mol\right)\\ \Rightarrow m_{Fe_3O_4}=0,18\cdot232=41,76\left(g\right)\)