Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

Coi $n_A = 1(mol) \Rightarrow m_A = 1.2.13,5 = 27(gam)$
$m_{NH_3} + m_{O_2} + m_{N_2} = 27$
$\Rightarrow \dfrac{7}{8}m_{O_2} + m_{O_2} + \dfrac{3}{6} (m_{O_2} + m_{NH_3} ) = 27$
$\Rightarrow \dfrac{7}{8}m_{O_2} + m_{O_2} + \dfrac{3}{6} (m_{O_2} + \dfrac{7}{8}m_{O_2} ) = 27$
$\Rightarrow \dfrac{45}{16}m_{O_2} = 27 \Rightarrow m_{O_2} = 9,6(gam)$
Suy ra:
$m_{NH_3} = 8,4 ; m_{N_2} = 9$
Suy ra : $n_{O_2} = 0,3(mol) ; n_{NH_3} = \dfrac{42}{85}(mol)$
$\%V_{O_2} = \dfrac{0,3}{1}.100\% = 30\%$
$\%V_{NH_3} = 49,41\%$
$\%V_{N_2} = 20,59\%$

a.\(M_A=23.2=46\) ( g/mol )
b.\(M_B=2,7.16=43,2\) ( g/mol )
c.\(M_C=2.29=58\) ( g/mol )
d.\(M_D=2.17=34\) ( g/mol )
e.\(M_E=1,32.44=58,08\) ( g/mol )
f.\(M_F=2,71.34=92,14\) ( g/mol )
g.\(M_G=1,5.32=48\) ( g/mol )
h.\(M_H=0,41.71=29,11\) ( g/mol )

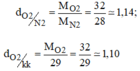
a.\(\dfrac{M_{O_2}}{M_{H_2}}=\dfrac{32}{2}=16\)
b.\(\dfrac{M_{N_2}}{29}=\dfrac{28}{29}=0,96\)