Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

\(n_{KClO_3}=\dfrac{12,25}{122,5}=0,1\left(mol\right)\)
PTHH: 2KClO3 -to, MnO2-> 2KCl + 3O2
0,1--------------------->0,1--->0,15
=> \(\left\{{}\begin{matrix}V_{O_2}=0,15.24,79=3,7185\left(l\right)\\m_{KCl}=0,1.74,5=7,45\left(g\right)\end{matrix}\right.\)
\(n_P=\dfrac{3,1}{31}=0,1\left(mol\right)\)
PTHH: 4P + 5O2 --to--> 2P2O5
LTL: \(\dfrac{0,1}{4}< \dfrac{0,15}{5}\) => P có cháy hết

\(n_C=\dfrac{1.2}{12}=0.1\left(mol\right)\\ n_S=\dfrac{4}{32}=0.125\left(mol\right)\)
\(2KMnO_4\underrightarrow{t^0}K_2MnO_4+MnO_2+O_2\)
\(C+O_2\underrightarrow{t^0}CO_2\)
\(S+O_2\underrightarrow{t^0}SO_2\)
\(\sum n_{O_2}=n_C+n_S=0.1+0.125=0.225\left(mol\right)\)
\(\Rightarrow n_{KMnO_4}=2n_{O_2}=0.225\cdot2=0.45\left(mol\right)\)
\(m_{KMnO_4}=0.45\cdot158=71.1\left(g\right)\)

\(n_C=\dfrac{m}{M}=\dfrac{3,6}{12}=0,3\left(mol\right)\\ PTHH\left(1\right):C+O_2\underrightarrow{t^o}CO_2\\ Theo.PTHH\left(1\right):n_O=n_C=0,3\left(mol\right)\)
\(PTHH\left(2\right):2KClO_3\underrightarrow{t^o}2KCl+3O_2\\ Theo.PTHH\left(2\right):n_{KClO_3}=\dfrac{2}{3}n_O=\dfrac{2}{3}.0,3=0,2\left(mol\right)\\ \Rightarrow m_{KClO_3}=n.M=0,2.122,5=24,5\left(g\right)\)

\(2KMnO4-->K2MnO4+MnO2+O2\)
\(C+O2-->CO2\)
\(n_C=\frac{4,8}{12}=0,4\left(mol\right)\)
\(n_{O2}=n_C=0,4\left(mol\right)\)
\(n_{KMNO4}=2n_{O2}=0,8\left(mol\right)\)
\(m_{KMnO4}=0,8.158=126,4\left(g\right)\)

a) nO2 = 48/32 = 1,5 (mol)
PTHH: 2KClO3 -> (t°, MnO2) 2KCl + 3O2
nKClO3 = 1,5 : 3 . 2 = 1 (mol)
mKClO3 = 1 . 122,5 = 122,5 (g)
b) nO2 = 49,58/24,79 = 2 (mol)
nKClO3 = 2 : 3 . 2 = 4/3 (mol)
mKClO3 = 4/3 . 122,5 = 490/3 (g)

a) \(n_{SO_2}=\dfrac{6,4}{64}=0,1\left(mol\right)\)
PTHH : S + O2 - to---> SO2
0,1 0,1 0,1 ( mol )
b) \(m_S=0,1.32=3,2\left(g\right)\)
\(V_{O_2}=0,1.22,4=2,24\left(l\right)\)
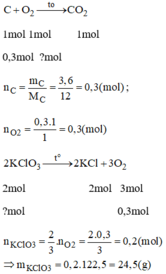

2KClO3-to>2KCl+3O2
0,3---------------------0,45
S+O2-to>SO2
0,45--0,45
n S=\(\dfrac{14,4}{32}\)=0,45 mol
=>m KClO3=0,3.122,5=36,75g