Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

1 ) CAO +H2O => CA(OH)2 (1)
2K + 2H2O => 2KOH + H2(2)
n (H2) =1,12/22,4 =0,05
theo ptpư 2 : n(K) = 2n (h2) =2.0.05=0,1(mol)
=> m (K) =39.0,1=3,9 (g)
% K= 3,9/9,5 .100% =41,05%
%ca =100%-41,05%=58,95%
xo + 2hcl =>xcl2 +h2o
10,4/X+16 15,9/x+71
=> giải ra tìm đc X bằng bao nhiêu thì ra

Gọi \(\left\{{}\begin{matrix}n_{Zn}=a\left(mol\right)\\n_{Fe}=b\left(mol\right)\end{matrix}\right.\)
\(n_{H_2}=\dfrac{15,68}{22,4}=0,7\left(mol\right)\\ m_{HCl}=200.27,375\%=54,75\left(g\right)\\ n_{HCl}=\dfrac{54,75}{36,5}=1,5\left(mol\right)\)
PTHH:
Zn + 2HCl ---> ZnCl2 + H2
a ----> 2a --------> a -----> a
Fe + 2HCl ---> FeCl2 + H2
b ---> 2b -------> b ------> b
Hệ pt \(\left\{{}\begin{matrix}65a+56b=43,7\\a+b=0,7\end{matrix}\right.\Leftrightarrow\left\{{}\begin{matrix}a=0,5\left(mol\right)\\b=0,2\left(mol\right)\end{matrix}\right.\)
\(\rightarrow\left\{{}\begin{matrix}m_{Zn}=0,5.65=32,5\left(g\right)\\m_{Fe}=0,2.56=11,2\left(g\right)\end{matrix}\right.\)
\(m_{dd}=43,7+200-0,7.2=242,3\left(g\right)\\ \rightarrow\left\{{}\begin{matrix}C\%_{ZnCl_2}=\dfrac{0,5.136}{242,3}=28,06\%\\C\%_{FeCl_2}=\dfrac{0,2.127}{242,3}=10,48\%\\C\%_{HCl\left(dư\right)}=\dfrac{\left(1,5-0,5.2-0,2.2\right).36,5}{242,3}=1,51\%\end{matrix}\right.\)
\(n_{H_2}=\dfrac{15,68}{22,4}=0,7\left(mol\right)\\
pthh:\left\{{}\begin{matrix}Zn+H_2SO_4->ZnSO_4+H_2\\Fe+H_2SO_4->FeSO_{\text{ 4 }}+H_2\end{matrix}\right.\)
gọi số mol Zn là x , số mol Fe là y
=> 65x+56y=43,7
=> a+b=0,7
=>a=0,5 , b =0,2
=> \(m_{Zn}=0,5.65=32,5\\ m_{Fe}=43,7-32,5=11,2\left(G\right)\)

a,Fe + 2HCl → FeCl + H2 (1)
FeO + 2HCl → FeCl + H2O (2)
nH2 = 3,36/ 22,4 = 0,15 ( mol)
Theo (1) nH2 = nFe = 0,15 ( mol)
mFe = 0,15 x 56 = 8.4 (g)
m FeO = 12 - 8,4 = 3,6 (g)
a, \(n_{H_2}=\frac{3,36}{22,4}=0,15\left(mol\right)\)
\(Fe+2HCl->FeCl_2+H_2\left(1\right)\)
\(FeO+2HCl->FeCl_2+H_2O\left(2\right)\)
theo (1) \(n_{Fe}=n_{H_2}=0,15\left(mol\right)\)
=> \(m_{Fe}=0,15.56=8,4\left(g\right)\)
=> \(m_{FeO}=12-8,4=3,6\left(g\right)\)

ta thấy : nFe =nH2 = 0,15
=> mFe =0,15 x 56 = 8,4g
%Fe=8,4/12 x 100 = 70%
=>%FeO = 100 - 70 = 30%
b) BTKLra mdd tìm mct of HCl
c) tìm mdd sau pứ -mH2 nha bạn

a, Chất rắn là MgO
\(n_{H_2}=\dfrac{8,96}{22,4}=0,4\left(mol\right)\)
PTHH: \(Na+H_2O\rightarrow NaOH+\dfrac{1}{2}H_2\)
0,8<-------------0,8<-------0,4
\(m=0,8.23+8=26,4\left(g\right)\)
\(b,m_{dd}=0,8.23+200-0,4.2=217,6\left(g\right)\\ \rightarrow C\%_{NaOH}=\dfrac{0,8.40}{217,6}.100\%=14,7\%\)
Tk:
a)2Al+ 6HCl→ 2AlCl3 +3H2↑
0,1________________0,15
Mg+ 2HCl→ MgCl2+ H2↑
0,2_______________0,2
MgO+ 2HCl→MgCl2+H2O
2Al+ 2NaOH+2H2O→ 2NaAlO2+ 3H2↑
0,1____________________________0,15
nHCl pư= 0,5.2.100110 =0,91 mol
nMgO=0,91−0,1.3−0,2.22= 0,105 mol
⇒ a= 0,1.27+0,2.24+ 0,105.40=11,7 g
b)
Dd B gồm:_______HCl dư ______AlCl3______MgCl2
_________________0,09________ 0,1_________0,305
NaOH+ HCl→ NaCl+ H2O
0,09 ___0,09
2NaOH+ MgCl2→ Mg(OH)2↓+ 2NaCl
0,61 ___0,305
3NaOH+ AlCl3→ 3NaCl+ Al(OH)3↓
0,3______ 0,1
VNaOH=0,09+0,61+0,32=0,5l

\(n_{Mg}=\dfrac{4.8}{24}=0.2\left(mol\right)\)
\(Mg+2HCl\rightarrow MgCl_2+H_2\)
\(0.2.......0.4........................0.2\)
\(C_{M_{HCl}}=\dfrac{0.4}{0.2}=2\left(M\right)\)
\(n_{CuO}=\dfrac{32}{80}=0.4\left(mol\right)\)
\(CuO+H_2\underrightarrow{^{^{t^0}}}Cu+H_2O\)
Lập tỉ lệ : \(\dfrac{0.4}{1}>\dfrac{0.2}{1}\)
=> CuO dư
\(m_{cr}=m_{CuO\left(dư\right)}+m_{Cu}=32-0.2\cdot80+0.2\cdot64=28.8\left(g\right)\)
\(\%Cu=\dfrac{0.2\cdot64}{28.8}\cdot100\%=44.44\%\)
\(\%CuO\left(dư\right)=55.56\%\)

Fe+2HCl->FeCl2+H2
0,125--0,25---0,125-0,125
m HCl=9,125 g=>n HCl=\(\dfrac{9,125}{26,5}\)=0,25 mol
=>m Fe=0,125.56=7g
=>VH2=0,125.22,4=2,8l
=>C%FeCl2=\(\dfrac{0,125.127}{7+182,5-0,25}\).100=8,388%

\(a) Fe + 2HCl \to FeCl_2\\ b) n_{HCl} = \dfrac{182,5.5\%}{36,5} = 0,25(mol)\\ n_{FeCl_2} = n_{H_2} = n_{Fe} = \dfrac{1}{2}n_{HCl} = 0,125(mol)\\ \Rightarrow m_{Fe} = 0,125.56 = 7(gam) ; V = 0,125.22,4 = 2,8(lít)\\ c) m_{dd\ sau\ phản\ ứng} = m_{Fe} + m_{dd\ HCl} - m_{H_2} = 7 + 182,5 - 0,125.2 = 189,25(gam)\\ C\%_{FeCl_2} = \dfrac{0,125.127}{189,25}.100\% = 8,39\%\)
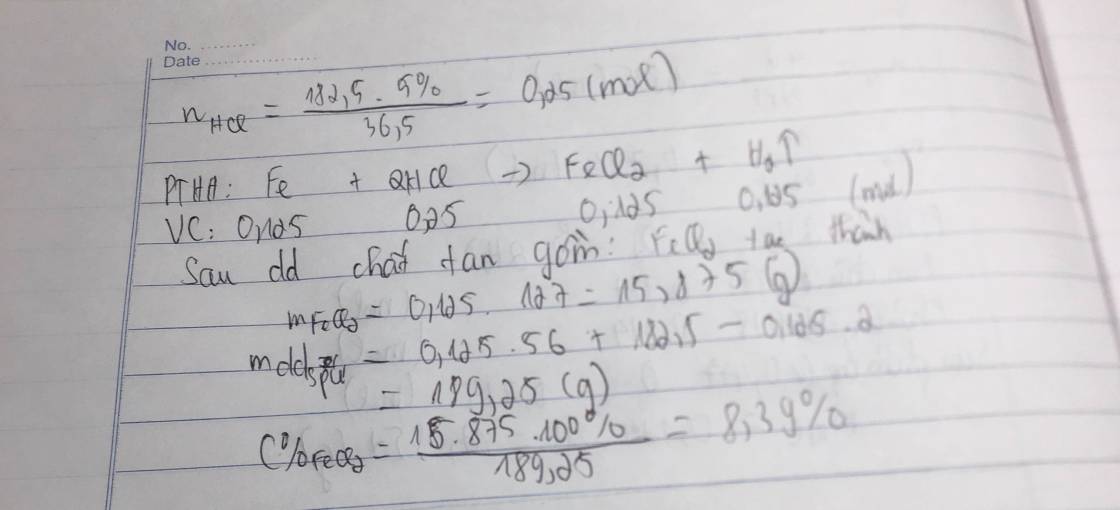
a) \(n_{H_2}=\dfrac{6,72}{22,4}=0,3\left(mol\right)\)
Chất rắn không tan là FeO
PTHH: 2K + 2H2O --> 2KOH + H2
0,6<--------------0,6<-0,3
=> m = 0,6.39 + 14,4 = 37,8 (g)
b) \(C_{M\left(dd.KOH\right)}=\dfrac{0,6}{2}=0,3M\)
c)
\(n_{FeO}=\dfrac{14,4}{72}=0,2\left(mol\right)\)
\(n_{HCl}=\dfrac{182,5.10\%}{36,5}=0,5\left(mol\right)\)
PTHH: FeO + 2HCl --> FeCl2 + H2O
Xét tỉ lệ: \(\dfrac{0,2}{1}< \dfrac{0,5}{2}\) => FeO hết, HCl dư
PTHH: FeO + 2HCl --> FeCl2 + H2O
0,2-->0,4------>0,2
mdd sau pư = 14,4 + 182,5 = 196,9 (g)
\(\left\{{}\begin{matrix}C\%_{FeCl_2}=\dfrac{0,2.127}{196,9}.100\%=12,9\%\\C\%_{HCl\left(dư\right)}=\dfrac{\left(0,5-0,4\right).36,5}{196,9}.100\%=1,85\%\end{matrix}\right.\)
a. \(n_{H_2}=\dfrac{6.72}{22,4}=0,3\left(mol\right)\)
PTHH : 2K + 2H2O -> 2KOH + H2
0,6 0,6 0,3
\(m_K=0,6.39=23,4\left(g\right)\)
\(m_{hh}=23,4+14,4=37,8\left(g\right)\)
b. \(C_M=\dfrac{0.6}{2}=0,3\left(M\right)\)
c. \(n_{FeO}=\dfrac{14.4}{72}=0,2\left(mol\right)\)
\(n_{HCl}=\dfrac{182,5.10\%}{36,5}=0,5\left(mol\right)\)
PTHH : FeO + 2HCl -> FeCl2 + H2
0,2 0,2
Ta thấy : \(\dfrac{0.2}{1}< \dfrac{0.5}{2}\) => FeO đủ , HCl dư
\(m_{FeCl_2}=0,2.127=25,4\left(g\right)\)
\(C\%=\dfrac{25,4}{14,4+182,5}.100\%=12,8\%\)