Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

a) $Mg + H_2SO_4 \to MgSO_4 + H_2$
b)
Theo PTHH : $n_{Mg} = n_{MgSO_4} = n_{H_2SO_4} = 0,14.1,2 = 0,168(mol)$
$m_{Mg} = 0,168.24 = 4,032(gam)$
$m_{MgSO_4} = 0,168.120 = 20,16(gam)$
c)
$n_{H_2} = n_{H_2SO_4} = 0,168(mol)$
$V_{H_2} = 0,168.22,4=3,7632(lít)$

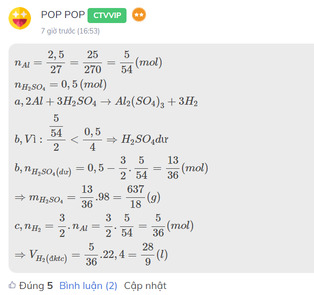
\(n_{Al}=\dfrac{2,5}{27}=\dfrac{25}{270}=\dfrac{5}{54}\left(mol\right)\\ n_{H_2SO_4}=0,5\left(mol\right)\\ a,2Al+3H_2SO_4\rightarrow Al_2\left(SO_4\right)_3+3H_2\\ b,Vì:\dfrac{\dfrac{5}{54}}{2}< \dfrac{0,5}{4}\Rightarrow H_2SO_4dư\\ b,n_{H_2SO_4\left(dư\right)}=0,5-\dfrac{3}{2}.\dfrac{5}{54}=\dfrac{13}{36}\left(mol\right)\\ \Rightarrow m_{H_2SO_4}=\dfrac{13}{36}.98=\dfrac{637}{18}\left(g\right)\\ c,n_{H_2}=\dfrac{3}{2}.n_{Al}=\dfrac{3}{2}.\dfrac{5}{54}=\dfrac{5}{36}\left(mol\right)\\ \Rightarrow V_{H_2\left(đktc\right)}=\dfrac{5}{36}.22,4=\dfrac{28}{9}\left(l\right)\)

a: \(2Al+3H_2SO_4\rightarrow Al_2\left(SO_4\right)_3+3H_2\)
b: \(n_{Al}=\dfrac{2.5}{27}< \dfrac{1}{4}\)
=>H2SO4 dư, Al đủ
\(m_{H_2SO_4}=0.25\cdot98=24.5\left(g\right)\)
c: \(n_{Al_2\left(SO_4\right)_3}=\dfrac{2.5}{54}=\dfrac{5}{108}\left(mol\right)\)
\(\Leftrightarrow n_{H_2}=\dfrac{5}{36}\left(mol\right)\)
\(V_{H_2}=\dfrac{5}{36}\cdot22.4=\dfrac{28}{9}\left(lít\right)\)
Mình thấy bạn Thịnh tính lượng dư sai
Đây là bài mình từng làm, bạn tham khảo nhé!

PTHH: \(2Al+3H_2SO_4\rightarrow Al_2\left(SO_4\right)_3+3H_2\uparrow\)
Làm gộp các phần còn lại
Ta có: \(n_{Al}=\dfrac{5,4}{27}=0,2\left(mol\right)\) \(\Rightarrow\left\{{}\begin{matrix}n_{Al_2\left(SO_4\right)_3}=0,1mol\\n_{H_2SO_4}=n_{H_2}=0,3mol\end{matrix}\right.\)
\(\Rightarrow\left\{{}\begin{matrix}V_{H_2}=0,3\cdot22,4=6,72\left(l\right)\\m_{Al_2\left(SO_4\right)_3}=0,1\cdot342=34,2\left(g\right)\\m_{H_2SO_4}=0,3\cdot98=29,4\left(g\right)\end{matrix}\right.\)

a) \(n_{Zn}=\dfrac{19,5}{65}=0,3\left(mol\right)\)
\(n_{HCl}=\dfrac{18,25}{36,5}=0,5\left(mol\right)\)
PTHH : Zn + 2HCl -> ZnCl2 + H2
0,25 0,5 0,5 0,5
Xét tỉ lệ : \(\dfrac{0,3}{1}>\dfrac{0,5}{2}\) => Zn dư , HCl đủ
b) \(m_{Zn\left(dư\right)}=\left(0,3-0,25\right).65=3,25\left(g\right)\)
c) \(m_{ZnCl_2}=0,25.136=34\left(g\right)\)
\(V_{H_2}=0,25.22,4=5,6\left(l\right)\)
\(n_{Zn}=\dfrac{19,5}{65}=0,3\left(mol\right)\\ n_{HCl}=\dfrac{18,25}{36,5}=0,5\left(mol\right)\\ a,Zn+2HCl\rightarrow ZnCl_2+H_2\\b, Vì:\dfrac{0,5}{2}< \dfrac{0,3}{1}\Rightarrow Zndư\\ n_{Zn\left(dư\right)}=0,3-\dfrac{0,5}{2}=0,05\left(mol\right)\\ \Rightarrow m_{Zn\left(dư\right)}=0,05.65=3,25\left(g\right)\\ c,n_{ZnCl_2}=n_{H_2}=\dfrac{0,5}{2}=0,25\left(mol\right)\\ \Rightarrow m_{ZnCl_2}=0,25.136=34\left(g\right)\\ V_{H_2\left(đktc\right)}=0,25.22,4=5,6\left(l\right)\)

\(nAl=\dfrac{8,1}{27}=0,3\left(mol\right)\)
\(nHCl=\dfrac{21,9}{36,5}=0,6\left(mol\right)\)
PTHH:
\(2Al+6HCl\rightarrow2AlCl_3+3H_2\)
2 6 2 3 (mol)
0,2 0,6 0,2 0,3 (mol)
LTL : 0,3 / 2 > 0,6/6
=> Al dư sau pứ , HCl đủ vs pứ
\(mAl_{\left(dư\right)}=\left(0,3-0,2\right).27=2,7\left(g\right)\)
\(mAlCl_3=0,2.98=19,6\left(g\right)\)
\(H_2+CuO\rightarrow Cu+H_2O\)
1 1 1 1 (mol)
0,3 0,3 0,3 0,3 (mol)
=> \(mCu=0,3.64=19,2\left(g\right)\)
\(n_{Al}=\dfrac{8,1}{27}=0,3\left(mol\right)\\
n_{HCl}=\dfrac{21,9}{36,5}=0,6\left(mol\right)\\
pthh:2Al+6HCl\rightarrow2AlCl_3+3H_2\uparrow\)
\(LTL:\dfrac{0,3}{2}>\dfrac{0,6}{6}\)
=> Al dư HCl hết
theo pthh : \(n_{Al\left(p\text{ư}\right)}=\dfrac{1}{3}n_{HCl}=0,2\left(mol\right)\\ m_{Al\left(d\right)}=\left(0,3-0,2\right).27=2,7\left(g\right)\)
theo pthh : \(n_{AlCl_3}=\dfrac{1}{3}n_{HCl}=0,1\left(mol\right)\\
m_{AlCl_3}=0,1.133,5=13,35\left(g\right)\)
theo pthh : \(n_{H_2}=\dfrac{1}{2}n_{HCl}=0,3\left(mol\right)\)
pthh: \(CuO+H_2\underrightarrow{t^o}H_2O+Cu\)
0,3 0,3
\(m_{Cu}=0,3.64=19,2\)

nZn=19,5/65=0,3(mol)
mHCl=18,25/36,5=0,5(mol)
pt: Zn+2HCl--->ZnCl2+H2
1______2
0,3_____0,5
Ta có: 0,3/1>0,5/2
=>Zn dư
mZn dư=0,05.65=3,25(mol)
Theo pt: nH2=1/2nHCl=1/2.0,5=0,25(mol)
=>VH2=0,25.22,4=5,6(l)
nZn = 0,3 mol
nHCl = 0,5 mol
Zn + 2HCl → ZnCl2 + H2
Đặt tỉ lệ ta có
0,3 < \(\dfrac{0,52}{2}\)
⇒ Zn dư và dư 3,25 gam
⇒ VH2 = 0,25.22,4 = 5,6 (l)

a)
n Al = 10,8/27 = 0,4(mol)
2Al + 6HCl → 2AlCl3 + 3H2
n H2 = \(\dfrac{3}{2}\)n Al = 0,6(mol)
=> V H2 = 0,6.22,4 = 13,44(lít)
b) n AlCl3 = n Al = 0,4(mol)
=> m AlCl3 = 0,4.133,5 = 53,4(gam)
c) n CuO = 16/80 = 0,2(mol)
CuO + H2 \(\xrightarrow{t^o}\) Cu + H2O
n CuO = 0,2 < n H2 = 0,6 => H2 dư
n H2 pư = n Cu = n CuO = 0,2 mol
Suy ra:
m H2 dư = (0,6 -0,2).2 = 0,8(gam)
m Cu = 0,2.64 = 12,8(gam)
a) nAl=0,4(mol)
PTHH: 2Al + 6HCl -> 2AlCl3 + 3H2
nH2= 3/2 . nAl=3/2 . 0,4=0,6(mol)
=>V(H2,đktc)=0,6 x 22,4= 13,44(l)
b) nAlCl3= nAl=0,4(mol)
=>mAlCl3=133,5 x 0,4= 53,4(g)
c) nCuO=0,2(mol)
PTHH: CuO + H2 -to-> Cu + H2O
Ta có: 0,2/1 < 0,6/1
=> H2 dư, CuO hết, tính theo nCuO
=> nH2(p.ứ)=nCu=nCuO=0,2(mol)
=>nH2(dư)=0,6 - 0,2=0,4(mol)
=> mH2(dư)=0,4. 2=0,8(g)
mCu=0,2.64=12,4(g)
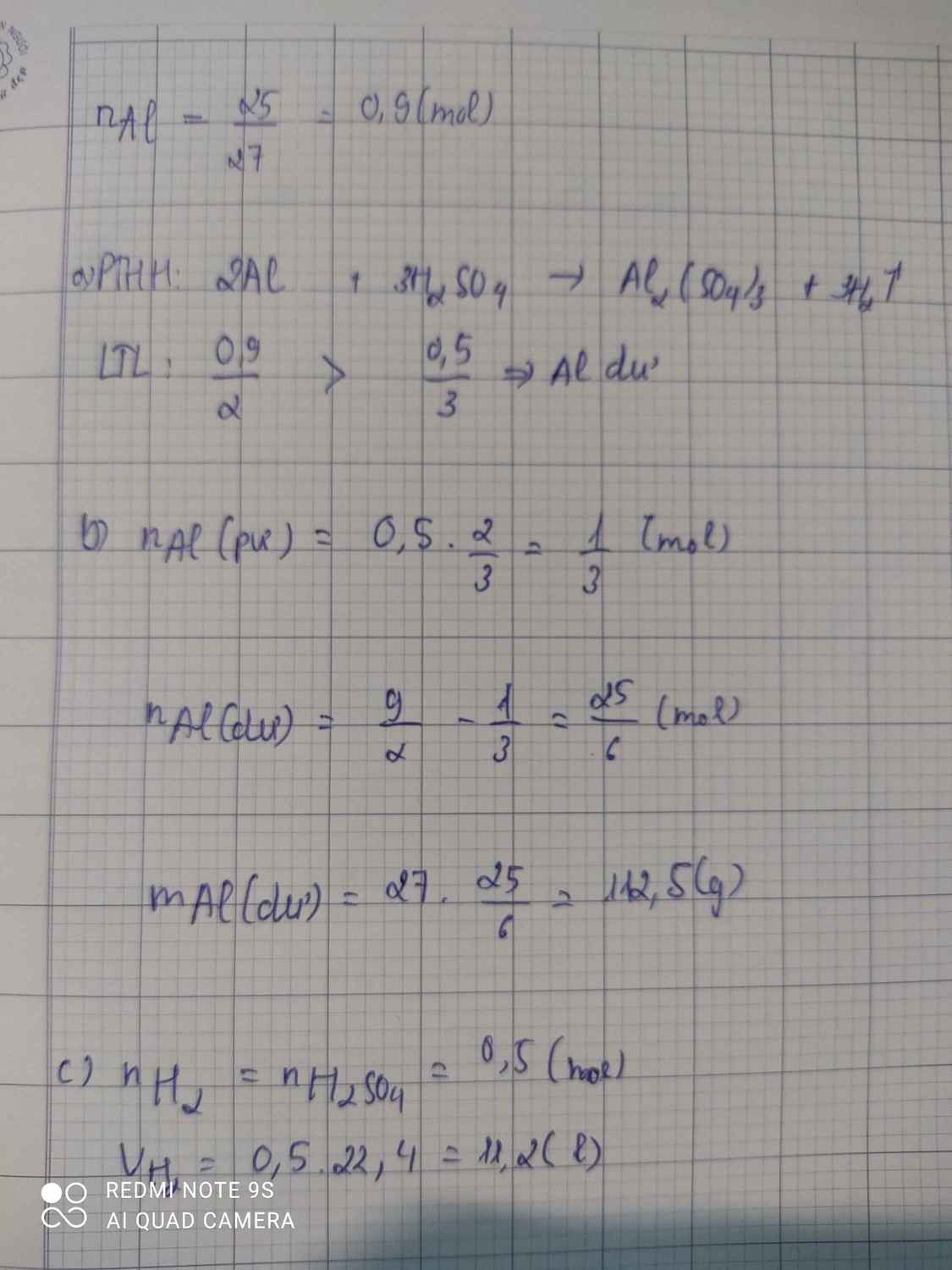

$a) 2Al + 3H_2SO_4 \to Al_2(SO_4)_3 + 3H_2$
$b) n_{Al} = \dfrac{12,15}{27} =0,45(mol) ; n_{H_2SO_4} = \dfrac{109,5.20\%}{98} = 0,2234(mol)$
Ta thấy :
$n_{Al} : 2 > n_{H_2SO_4} : 3$ nên Al dư
$n_{Al\ pư} = \dfrac{2}{3}n_{H_2SO_4} = 0,15(mol)$
$\Rightarrow m_{Al\ dư} = 12,15 - 0,15.27 = 8,1(gam)$
c) $n_{Al_2(SO_4)_3} = \dfrac{1}{3}n_{H_2SO_4} = 0,0747(mol)$
$m_{Al_2(SO_4)_3} = 0,0747.342 = 25,5474(gam)$
d) $n_{H_2} = n_{H_2SO_4} = 0,2234(mol)$
$V_{H_2} = 0,2234.22,4 = 5,00416(lít)$