Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

\(a.n_{CO_2}=\dfrac{2,24}{22,4}=0,1\left(mol\right)\\ CO_2+Ba\left(OH\right)_2\rightarrow BaCO_3+H_2O\\ 0,1...........0,1.............0,1..........0,1\left(mol\right)\\ b.m_{kt}=m_{BaCO_3}=0,1.197=19,7\left(g\right)\\ c.C_{MddBa\left(OH\right)_2}=\dfrac{0,1}{0,2}=0,5\left(M\right)\)

Ta có: \(\left\{{}\begin{matrix}n_{CO_2}=\dfrac{2,24}{22,4}=0,1\left(mol\right)\\n_{NaOH}=0,2\cdot1=0,2\left(mol\right)\end{matrix}\right.\) \(\Rightarrow\) Tạo muối trung hòa
a) PTHH: \(CO_2+2NaOH\rightarrow Na_2CO_3+H_2O\)
b) Theo PTHH: \(n_{Na_2CO_3}=n_{CO_2}=0,1mol\) \(\Rightarrow m_{Na_2CO_3}=0,1\cdot106=10,6\left(g\right)\)
a) PTHH: CO2 + 2NaOH -----> Na2CO3 + H2O (1)
b) nCO2 = \(\dfrac{2,24}{22,4}\)= 0,1 (mol)
Đổi 200ml = 0,2l
nNaOH = Cm x V = 1 x 0,2 = 0,2 (mol)
Lập tỷ lệ: \(\dfrac{nCO2}{1}\)=\(\dfrac{0,1}{1}\)=\(\dfrac{0,2}{2}\)=\(\dfrac{nNaOH}{2}\)
Sau phản ứng, CO2 và NaOH hết. Các chất được tính theo CO2 ( hoặc NaOH )
Theo (1): nNa2CO3 = nCO2 = 0,1 (mol)
-> mNa2CO3 = 0,1 x 106 = 10,6 (g)

2.
a, \(n_{HCl}=0,2.3,5=0,7\left(mol\right)\)
PTHH: CuO + 2HCl → CuCl2 + H2O
Mol: x 2x
PTHH: Fe2O3 + 6HCl → 2FeCl3 + 3H2O
Mol: y 6y
Ta có: \(\left\{{}\begin{matrix}80x+160y=20\\2x+6y=0,7\end{matrix}\right.\Leftrightarrow\left\{{}\begin{matrix}x=0,05\\y=0,1\end{matrix}\right.\)
b, \(m_{CuO}=0,05.80=4\left(g\right);m_{Fe_2O_3}=20-4=16\left(g\right)\)
c,
PTHH: CuO + 2HCl → CuCl2 + H2O
Mol: 0,05 0,05
PTHH: Fe2O3 + 6HCl → 2FeCl3 + 3H2O
Mol: 0,1 0,2
\(m_{CuCl_2}=0,05.135=6,75\left(g\right)\)
\(m_{FeCl_3}=0,1.162,5=16,25\left(g\right)\)
1.
a, \(n_{CO_2}=\dfrac{2,24}{22,4}=0,1\left(mol\right)\)
PTHH: CO2 + Ba(OH)2 → BaCO3 + H2O
Mol: 0,1 0,1 0,1
b, \(C_{M_{ddBa\left(OH\right)_2}}=\dfrac{0,1}{0,2}=0,5M\)
c, \(m_{BaCO_3}=0,1.197=19,7\left(g\right)\)

\(\text{Đặt }\left\{{}\begin{matrix}n_{Al}=x\left(mol\right)\\n_{Fe}=y\left(mol\right)\end{matrix}\right.\\ n_{H_2}=\dfrac{5,6}{22,4}=0,25\left(mol\right)\\ a,PTHH:\left\{{}\begin{matrix}2Al+6HCl\rightarrow2AlCl_3+3H_2\\Fe+2HCl\rightarrow FeCl_2+H_2\end{matrix}\right.\\ b,\text{Theo đề ta có HPT: }\left\{{}\begin{matrix}27x+56y=8,3\\\dfrac{3}{2}x+y=0,25\end{matrix}\right.\Rightarrow\left\{{}\begin{matrix}x=0,1\left(mol\right)\\y=0,1\left(mol\right)\end{matrix}\right.\\ \Rightarrow\left\{{}\begin{matrix}\%_{Al}=\dfrac{0,1\cdot27}{8,3}\approx32,53\%\\\%_{Fe}\approx67,47\%\end{matrix}\right.\)
\(c,\left\{{}\begin{matrix}n_{AlCl_3}=0,1\left(mol\right)\\n_{FeCl_2}=0,1\left(mol\right)\end{matrix}\right.\Rightarrow\left\{{}\begin{matrix}m_{AlCl_3}=0,1\cdot133,5=13,35\left(g\right)\\m_{FeCl_2}=0,1\cdot127=12,7\left(g\right)\end{matrix}\right.\\ \Rightarrow\sum m_{muối}=13,35+12,7=26,05\left(g\right)\)

\(a.Mg+2HCl\rightarrow MgCl_2+H_2\\ b.n_{H_2}=0,2\left(mol\right)\\ TheoPT:n_{Mg}=n_{H_2}=0,2\left(mol\right)\\ \Rightarrow m_{Mg}=0,2.24=4,8\left(g\right)\\ c.n_{HCl}=2n_{H_2}=0,4\left(mol\right)\\ \Rightarrow m_{HCl}=0,4.36,5=14,6\left(g\right)\)

\(n_{CuCl_2}=\dfrac{60,75}{135}=0,45mol\\ a)CuCl_2+2NaOH\rightarrow Cu\left(OH\right)_2+2NaCl\)
0,45 0,9 0,45 0,9
\(b)m_X=m_{Cu\left(OH\right)_2}=0,45.81=36,45g\\
c)m_{ddNaOH}=\dfrac{0,9.40}{15\%}\cdot100\%=240g\\
d)m_{ddNaCl}=60,75+240-36,45=264,3g\\
C_{\%NaCl}=\dfrac{0,9.58,5}{264,3}\cdot100\%=19,92\%\\
e)n_{H_2SO_4}=\dfrac{245.20\%}{100\%.98}=0,5mol\\
H_2SO_4+Cu\left(OH\right)_2\rightarrow CuSO_4+2H_2O\\
\Rightarrow\dfrac{0,5}{1}>\dfrac{0,45}{1}\Rightarrow H_2SO_4.dư\)
\(\Rightarrow\)Dung dịch acid \(H_2SO_4\) làm tan hết chất X\(\left(Cu\left(OH\right)_2\right)\)
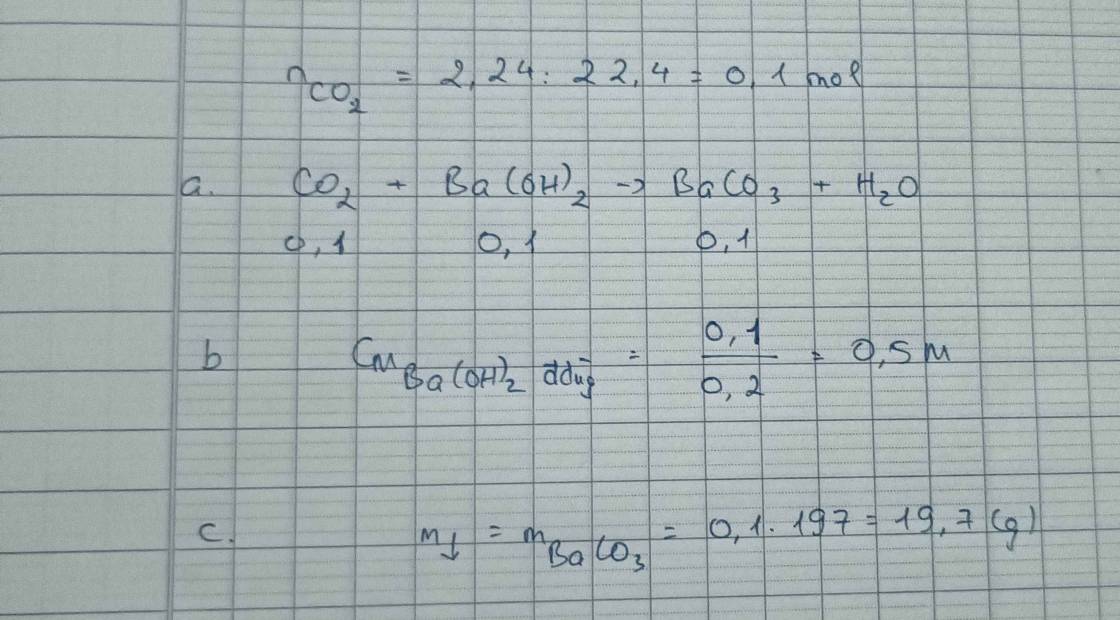
\(B1\\ n_{CaO}=\dfrac{5,6}{56}=0,1\left(mol\right)\\ a,CaO+2HCl\rightarrow CaCl_2+H_2O\\ n_{CaCl_2}=n_{H_2O}=n_{CaO}=0,1\left(mol\right)\\ n_{HCl}=0,1.2=0,2\left(mol\right)\\ b,m_{HCl}=0,2.36,5=7,3\left(g\right)\\ m_{CaCl_2}=0,1.111=11,1\left(g\right)\\ m_{H_2O}=0,1.18=1,8\left(g\right)\)
\(B2\\ n_{CO_2}=\dfrac{2,24}{22,4}=0,1\left(mol\right)\\a, CaO+CO_2\rightarrow\left(t^o\right)CaCO_3\\ b,n_{CaO}=n_{CO_2}=n_{CaCO_3}=0,1\left(mol\right)\\ m_{CaO}=0,1.56=5,6\left(g\right)\\ m_{CaCO_3}=100.0,1=10\left(g\right)\)