Tính phần trăm khối lượng các nguyên tố trong HNO3,CaCO3
Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.


\(CaCO_3\\ \%m_{Ca}=\dfrac{40}{40+12+3.16}.100=40\%\\ \%m_C=\dfrac{12}{40+12+16.3}.100=12\%\\ \Rightarrow\%m_O=100\%-\left(40\%+12\%\right)=48\%\\ H_2SO_4\\ \%m_H=\dfrac{2.1}{2.1+32+4.16}.100\approx2,041\%\\ \%m_S=\dfrac{32}{2.1+32+4.16}.100\approx32,653\%\\ \%m_O=\dfrac{4.16}{2.1+32+4.16}.100\approx65,306\%\\ Fe_2O_3\\ \%m_{Fe}=\dfrac{56.2}{56.2+16.3}.100=70\%\\ \Rightarrow\%m_O=100\%-70\%=30\%\)
CaCO3
\(\%M_{\dfrac{Ca}{CaCO_3}}=\dfrac{40}{100}.100\%=40\%\)
\(\%M_{\dfrac{C}{CaCO_3}}=\dfrac{12}{100}.100\%=12\%\)
\(\%M_{\dfrac{O}{CaCO_3}}=100\%-\left(40\%+12\%\right)=48\%\)
H2SO4
\(\%M_{\dfrac{H_2}{H_2SO_4}}=\dfrac{2}{98}.100\%=2,04\%\)
\(\%M_{\dfrac{S}{H_2SO_4}}=\dfrac{32}{98}.100\%=32,65\%\)
\(\%M_{\dfrac{O}{H_2SO_4}}=100\%-\left(2,04\%+32,65\%\right)=65,31\%\)
Fe2O3
\(\%M_{\dfrac{Fe}{Fe_2O_3}}=\dfrac{112}{160}.100\%=70\%\)
\(\%M_{\dfrac{O}{Fe_2O_3}}=100\%-70\%=30\%\)

`a,` \(K.L.P.T_{Fe_2O_3}=56.2+16.3=160< amu>.\)
\(\%Fe=\dfrac{56.2.100}{160}=70\%\)
\(\%O=100\%-70\%=30\%\)
`b,`\(K.L.P.T_{CaCO_3}=40+12+16.3=100< amu>.\)
\(\%Ca=\dfrac{40.100}{100}=40\%\)
\(\%C=\dfrac{12.100}{100}=12\%\)
\(\%O=100\%-40\%-12\%=48\%\)
`c,` \(K.L.P.T_{HCl}=1+35,5=36,5< amu>.\)
\(\%H=\dfrac{1.100}{36,5}\approx2,74\%\)
\(\%Cl=100\%-2,74\%=97,26\%\)
a: \(\%Fe=\dfrac{56\cdot2}{56\cdot2+16\cdot3}=70\%\)
=>%O=30%
b: \(\%Ca=\dfrac{40}{40+12+16\cdot3}=40\%\)
\(\%C=\dfrac{12}{100}=12\%\)
%O=100%-12%-40%=48%
c: %H=1/36,5=2,74%
=>%Cl=97,26%

Ta có: MCaCO3 = 40 + 12 + (16 × 3) = 100g
Trong 1mol CaCO3 có 1mol Ca, 1 mol C và 3 mol O
Do đó: MCa = 1 × 40 = 40g
MC = 1 × 12 = 12g
MO = 3 × 16 = 48g
Thành phần % các nguyên tố có trong hợp chất CaCO3 là:
%Ca = (40/100) × 100% = 40%
%C = (12/100) × 100% = 12%
%O = (16 × 3/100) × 100% = 48%
Vậy ....

\(M_{NaOH}=40(g/mol)\\ \%_{Na}=\dfrac{23}{40}.100\%=57,5\%\\ \%_O=\dfrac{16}{40}.100\%=40\%\\ \%_H=\dfrac{1}{40}.100\%=2,5\%\\ M_{H_2CO_3}=2+12+16.3=62(g/mol)\\ \%_H=\dfrac{2}{62}.100\%=3,23\%\\ \%_C=\dfrac{12}{62}.100\%=19,35\%\\ \%_O=100\%-3,23\%-19,35\%=77,42\%\)
\(M_{CaCO_3}=40+12+16.3=100(g/mol)\\ \%_{Ca}=\dfrac{40}{100}.100\%=40\%\\ \%_C=\dfrac{12}{100}.100\%=12\%\\ \%_O=\dfrac{48}{100}.100\%=48\%\\ M_{KNO_3}=39+14+16.3=101(g/mol)\\ \%_K=\dfrac{39}{101}.100\%=38,61\%\\ \%_N=\dfrac{14}{101}.100\%=13,86\%\\ \%_O=100\%-38,61\%-13,86\%=47,53\%\)

\(PTK_{CaCO_3}=NTK_{Ca}+NTK_C+3.NTK_O=40+12+3.16=100\left(đ.v.C\right)\\ \%m_{Ca}=\dfrac{NTK_{Ca}}{PTK_{CaCO_3}}.100\%=\dfrac{40}{100}.100=40\%\\ \%m_C=\dfrac{NTK_C}{PTK_{CaCO_3}}.100\%=\dfrac{12}{100}.100=12\%\\ \%m_O=100\%-\left(\%m_{Ca}+\%m_C\right)=100\%-\left(40\%+12\%\right)=48\%\)

\(PTK_{CuO}=64+16=80\left(đvC\right)\)
\(\%m_{Cu}=\) \(\dfrac{64}{80}.100=80\%\)
\(\%m_O=100-80=20\%\)
\(PTK_{MgCO_3}=24+12+3.16=84\left(đvC\right)\)
\(\%m_{Mg}=\dfrac{24}{84}.100=28,57\%\)
\(\%m_C=\dfrac{12}{84}.100=14,28\%\)
\(\%m_O=\dfrac{3.16}{84}.100=57,14\%\)
các ý còn lại làm tương tự
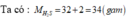
\(M_{HNO_3}=1+14+16.3=63\left(\dfrac{g}{mol}\right)\\ \%m_H=\dfrac{1.100\%}{63}=1,6\%\\ m_N=\dfrac{14.100\%}{63}=22,2\%\\ m_O=100\%-\left(1,6\%+22,2\%\right)=76,2\%\\ M_{CaCO_3}=40+12+16.3=100\left(\dfrac{g}{mol}\right)\\ \%m_{Ca}=\dfrac{40.100\%}{100}=40\%\\ \%m_C=\dfrac{12.100\%}{100}=12\%\\ \%m_O=100-\left(40\%+12\%\right)=48\%\)