Hoà tan 10 gam hỗn hợp Al và Ag trong dung dịch HCl phản ứng kết thúc thu 6,72 lít H2(đktc). Xác định khối lượng từng kim loại trong hỗn hợp
Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.


a)
Gọi : \(\left\{{}\begin{matrix}n_{Al}=a\left(mol\right)\\n_{Fe}=b\left(mol\right)\end{matrix}\right.\)⇒ 27a + 56b = 1,66(1)
\(2Al + 6HCl \to 2AlCl_3 + 3H_2\\ Fe +2 HCl \to FeCl_2 + H_2\)
Theo PTHH :
\(n_{H_2} = 1,5a + b = \dfrac{1,12}{22,4} = 0,05(2)\)
Từ (1)(2) suy ra a = 0,02 ; b = 0,02
Vậy :
\(\%m_{Al} = \dfrac{0,02.27}{1,66}.100\% = 32,53\%\\ \%m_{Fe} = 100\% - 32,53\% = 67,47\%\)
a)
\(n_{HCl} = 2n_{H_2} = 0,05.2 = 0,1(mol)\\ \Rightarrow C\%_{HCl} = \dfrac{0,1.36,5}{100}.100\% = 3,65\%\)

Đặt \(\begin{matrix}n_{Al}=a\left(mol\right)\\n_{Fe}=b\left(mol\right)\end{matrix}=>27a+56b=13,8\left(1\right)\)
Pt : \(2Al+6HCl\rightarrow2AlCl_3+3H_2\)
a \(\dfrac{3}{2}\)a
\(Fe+2HCl\rightarrow FeCl_2+H_2\)
b b
\(n_{H2}=\dfrac{10,08}{22,4}=0,45\left(mol\right)=>1,5a+b=0,45\left(2\right)\)
Từ (1),(2) ta có hệ phương trình : \(\left\{{}\begin{matrix}27a+56b=13,8\\1,5a+b=0,45\end{matrix}\right.\Rightarrow\left\{{}\begin{matrix}a=0,2\\b=0,15\end{matrix}\right.\)
\(m_{Al}=0,2.27=5,4\left(g\right)\)
\(m_{Fe}=0,15.56=8,4\left(g\right)\)
Chúc bạn học tốt

Ta có: 27nAl + 56nFe = 13,8 (1)
\(n_{H_2}=\dfrac{10,08}{22,4}=0,45\left(mol\right)\)
Theo ĐLBT e, có: 3nAl + 2nFe = 2nH2 = 0,9 (2)
Từ (1) và (2) \(\Rightarrow\left\{{}\begin{matrix}n_{Al}=0,2\left(mol\right)\\n_{Fe}=0,15\left(mol\right)\end{matrix}\right.\)
\(\Rightarrow\left\{{}\begin{matrix}m_{Al}=0,2.27=5,4\left(g\right)\\m_{Fe}=0,15.56=8,4\left(g\right)\end{matrix}\right.\)

\(\left\{{}\begin{matrix}n_{Cu}\\n_{Fe}\end{matrix}\right.\Rightarrow\left\{{}\begin{matrix}64x+56y=24,4\\2x+3y=\dfrac{6,72}{22,4}.3\end{matrix}\right.\Rightarrow\left\{{}\begin{matrix}x=0,285\\y=0,11\end{matrix}\right.\)
\(\left\{{}\begin{matrix}\%m_{Cu}=74,75\%\\\%m_{Fe}=25,25\%\end{matrix}\right.\)

PTHH: \(Zn+2HCl\rightarrow ZnCl_2+H_2\uparrow\)
Ta có: \(n_{H_2}=\dfrac{6,72}{22,4}=0,3\left(mol\right)\)
\(\Rightarrow\left\{{}\begin{matrix}n_{Zn}=0,3\left(mol\right)=n_{ZnCl_2}\\n_{HCl}=0,6\left(mol\right)\end{matrix}\right.\) \(\Rightarrow\left\{{}\begin{matrix}\%m_{Zn}=\dfrac{0.3\cdot65}{35,5}\cdot100\%\approx54,93\%\\\%m_{Cu}=45,07\%\\C\%_{HCl}=\dfrac{0,6\cdot36,5}{500}\cdot100\%=4,38\%\\m_{ZnCl_2}=0,3\cdot136=40,8\left(g\right)\end{matrix}\right.\)
Mặt khác: \(\left\{{}\begin{matrix}m_{Cu}=35,5-0,3\cdot65=16\left(g\right)\\m_{H_2}=0,3\cdot2=0,6\left(g\right)\end{matrix}\right.\)
\(\Rightarrow m_{dd}=m_{KL}+m_{ddHCl}-m_{Cu}-m_{H_2}=518,9\left(g\right)\)
\(\Rightarrow C\%_{ZnCl_2}=\dfrac{40,8}{518,9}\cdot100\%\approx7,86\%\)

Chất rắn A là Ag vì ko phản ứng với HCl
PTHH: \(Zn+2HCl\rightarrow ZnCl_2+H_2\uparrow\)
Ta có: \(n_{H_2}=\dfrac{2,24}{22,4}=0,1\left(mol\right)=n_{Zn}\) \(\Rightarrow\left\{{}\begin{matrix}m_{Zn}=0,1\cdot65=6,5\left(g\right)\\m_{Ag}=10-6,5=3,5\left(g\right)\end{matrix}\right.\)

m(rắn)=mAg=3(g); nH2=6,72/22,4=0,3(mol)
2 Al +3 H2SO4 -> Al2(SO4)3 + 3 H2
nAl=2/3. 0,3=0,2(mol) => mAl=0,2.27=5,4(g)
=> \(\%mAl=\dfrac{5,4}{5,4+3}.100\approx64,3\%\)
=> CHỌN B
\(n_{H_2}=0,3\left(mol\right)\)
\(2Al+3H_2SO_{4\left(l\right)}\rightarrow Al_2\left(SO_4\right)_3+3H_2\)
0,2<---------------------------------------0,3
\(m_{Al}=0,2.27=5,4\left(g\right)\)
\(m_{r_{bđ}}=5,4+3=8,4\left(g\right)\)
\(\%m_{Al}=\dfrac{5,4.100\%}{8,4}\approx64,3\%\)
=>B
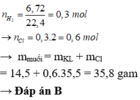
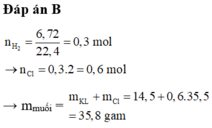
Ag không pư với dd HCl.
PT: \(2Al+6HCl\rightarrow2AlCl_3+3H_2\)
Ta có: \(n_{H_2}=\dfrac{6,72}{22,4}=0,3\left(mol\right)\)
Theo PT: \(n_{Al}=\dfrac{2}{3}n_{H_2}=0,2\left(mol\right)\)
\(\Rightarrow\left\{{}\begin{matrix}m_{Al}=0,2.27=5,4\left(g\right)\\m_{Ag}=10-5,4=4,6\left(g\right)\end{matrix}\right.\)
Bạn tham khảo nhé!
PTHH: \(2Al+6HCl\rightarrow2AlCl_3+3H_2\uparrow\)
Ta có: \(n_{Al}=\dfrac{2}{3}n_{H_2}=\dfrac{2}{3}\cdot\dfrac{6,72}{22,4}=0,2\left(mol\right)\)
\(\Rightarrow m_{Al}=0,2\cdot27=5,4\left(g\right)\) \(\Rightarrow m_{Ag}=4,6\left(g\right)\)