Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

\(n_{SO_2}=\frac{V_{SO_2}}{22,4}=\frac{2,24}{22,4}=0,1\left(mol\right)\)
\(=>m_{SO_2}=n_{SO_2}.M_{SO_2}=0,1.64=6,4\left(g\right)\)
\(n_{O_2}=\frac{V_{O_2}}{22,4}=\frac{3,36}{22,4}=0,15\left(mol\right)\)
\(=>m_{O_2}=n_{O_2}.M_{O_2}=0,15.32=4,8\left(g\right)\)

a, khối lượng của 2,5 mol CuO là:
\(m=n.M=2,5.80=200\left(g\right)\)
b, số mol của 4,48 lít khí CO2 (đktc) là:
\(n=\dfrac{V}{22,4}=\dfrac{4,48}{22,4}=0,2\left(mol\right)\)

N phân tử = 1 mol phân tử
\(\Rightarrow n_{O2}=1mol;n_{N_2}=2mol;n_{CO_2}=1,5mol\)
\(\Rightarrow m_{hh}=1.32+2.28+1,5.44=154g\)
b. \(m_{hh}=0,1.56+0,2.64+0,3.65+0,25.27=44,65g\)
c. \(n_{O_2}=\dfrac{2,24}{22,4}=0,1mol\)
\(n_{H_2}=\dfrac{1,12}{22,4}=0,05mol\)
\(n_{HCl}=\dfrac{6,72}{22,4}=0,3mol\)
\(n_{CO_2}=\dfrac{0,56}{22,4}=0,025mol\)
\(\Rightarrow m_{hh}=0,1.32+0,05.2+0,3.36,5+0,025.44=15,35g\)

Trong A :
\(n_{CO_2}=n_X=a\left(mol\right)\)
Trong B:
\(n_{N_2}=2b\left(mol\right),n_{CO_2}=3b\left(mol\right)\)
\(n_A=2a=0.1\left(mol\right)\Rightarrow a=0.05\)
\(n_B=5b=0.05\left(mol\right)\Rightarrow b=0.01\)
\(m=0.05\cdot44+0.05\cdot X+0.02\cdot28+0.03\cdot44=4.18\left(g\right)\)
\(\Rightarrow X=2\)
\(X:H_2\)

\(n_{O2}=\dfrac{16,8}{22,4}=0,75\left(mol\right)\)
\(m_{O2}=0,75.32=24\left(g\right)\)
⇒ \(M_{O2}=\dfrac{24}{0,75}=32\)(g/mol)
\(n_{SO2}=\dfrac{10,08}{22,4}=0,45\left(mol\right)\)
\(m_{SO2}=0,45.64=28,8\left(g\right)\)
⇒ \(M_{SO2}=\dfrac{28,8}{0,45}=64\)(g/mol)
\(n_{Cl2}=\dfrac{7,84}{22,4}=0,35\left(mol\right)\)
\(m_{Cl2}=0,35.71=24,85\left(g\right)\)
⇒ \(M_{Cl2}=\dfrac{24,85}{0,35}=71\)(g/mol)
Chúc bạn học tốt

\(1,\\ a,m_{hh}=3.44+2.28=188(g)\\ b,m_{hh}=\dfrac{2,24}{22,4}.64+\dfrac{1,12}{22,4}.32=8(g)\\ 2,\\ a,V_{hh}=(\dfrac{4,4}{44}+\dfrac{0,4}{2}).22,4=6,72(l)\\ b,V_{hh}=(\dfrac{6.10^{23}}{6.10^{23}}+\dfrac{3.10^{23}}{6.10^{23}}).22,4=33,6(l)\)
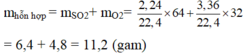
\(n_{SO_2\left(dktc\right)}=\dfrac{V}{22,4}=\dfrac{2,24}{22,4}=0,1\left(mol\right)\\ m_{SO_2}=n\cdot M=0,1\cdot\left(32+16\cdot2\right)=6,4\left(g\right)\)
\(n_{O_2\left(dktc\right)}=\dfrac{V}{22,4}=\dfrac{1,12}{22,4}=0,05\left(mol\right)\\ m_{O_2}=n\cdot M=0,05\cdot32=1,6\left(g\right)\)
\(=>m_{hh}=1,6+6,4=8\left(g\right)\)
\(n_{SO_2}=\dfrac{V_{\left(\text{đ}ktc\right)}}{22,4}=\dfrac{2,24}{22,4}=0,1\left(mol\right)\\ \Rightarrow m_{SO_2}=n.M=0,1.64=6,4\left(g\right)\\ n_{O_2}=\dfrac{V_{\left(\text{đ}ktc\right)}}{22,4}=\dfrac{1,12}{22,4}=0,05\left(mol\right)\\ \Rightarrow m_{O_2}=n.M=0,05.32=1,6\left(g\right)\\ \Rightarrow m_{hh}=m_{SO_2}+m_{O_2}=6,4+1,6=8\left(g\right)\)