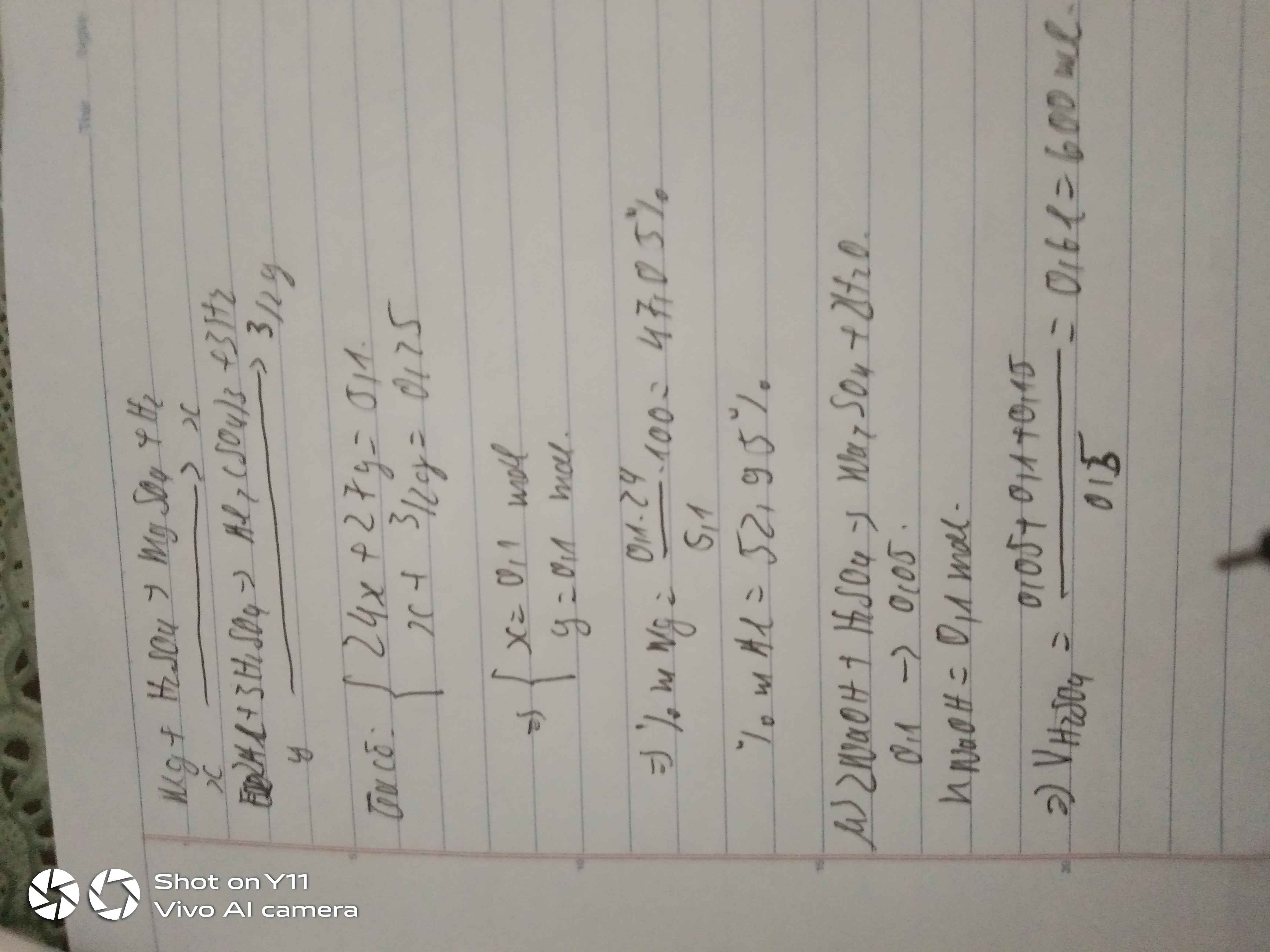Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

PT: \(2Al+3H_2SO_4\rightarrow Al_2\left(SO_4\right)_3+3H_2\)
Ta có: \(n_{H_2}=\dfrac{3,7185}{24,79}=0,15\left(mol\right)\)
a, \(n_{Al}=\dfrac{2}{3}n_{H_2}=0,1\left(mol\right)\Rightarrow m_{Al}=0,1.27=2,7\left(g\right)\)
b, \(n_{Al_2\left(SO_4\right)_3}=\dfrac{1}{3}n_{H_2}=0,05\left(mol\right)\)
\(\Rightarrow m_{Al_2\left(SO_4\right)_3}=0,05.342=17,1\left(g\right)\)
c, \(n_{H_2SO_4}=n_{H_2}=0,15\left(mol\right)\)
\(\Rightarrow V_{H_2SO_4}=\dfrac{0,15}{1,5}=0,1\left(l\right)=100\left(ml\right)\)

a.\(n_{Mg}=\dfrac{4,8}{24}=0,2mol\)
\(Mg+2HCl\rightarrow MgCl_2+H_2\)
0,2 0,2 ( mol )
\(V_{H_2}=0,2.24,79=4,958l\)
b.\(n_{Na}=\dfrac{6,9}{23}=0,3mol\)
\(2Na+2H_2O\rightarrow2NaOH+H_2\)
0,3 0,15 ( mol )
\(V_{H_2}=0,15.24,79=3,7185l\)
c.\(2Al+3H_2SO_4\rightarrow Al_2\left(SO_4\right)_3+3H_2\)
0,05 0,075 ( mol )
\(V_{H_2}=0,075.24,79=1,85925l\)

a) Mg + H2SO4 --> MgSO4 + H2
b) \(n_{Mg}=\dfrac{14,4}{24}=0,6\left(mol\right)\)
PTHH: Mg + H2SO4 --> MgSO4 + H2
0,6--->0,6------->0,6----->0,6
=> \(m_{H_2SO_4}=0,6.98=58,8\left(g\right)\)
c)
PTHH: 2H2 + O2 --to--> 2H2O
0,6-->0,3
=> VO2 = 0,3.24,79 = 7,437 (l)
=> Vkk = 7,437.5 = 37,185 (l)

a,Fe + 2HCl → FeCl + H2 (1)
FeO + 2HCl → FeCl + H2O (2)
nH2 = 3,36/ 22,4 = 0,15 ( mol)
Theo (1) nH2 = nFe = 0,15 ( mol)
mFe = 0,15 x 56 = 8.4 (g)
m FeO = 12 - 8,4 = 3,6 (g)
a, \(n_{H_2}=\frac{3,36}{22,4}=0,15\left(mol\right)\)
\(Fe+2HCl->FeCl_2+H_2\left(1\right)\)
\(FeO+2HCl->FeCl_2+H_2O\left(2\right)\)
theo (1) \(n_{Fe}=n_{H_2}=0,15\left(mol\right)\)
=> \(m_{Fe}=0,15.56=8,4\left(g\right)\)
=> \(m_{FeO}=12-8,4=3,6\left(g\right)\)

ta thấy : nFe =nH2 = 0,15
=> mFe =0,15 x 56 = 8,4g
%Fe=8,4/12 x 100 = 70%
=>%FeO = 100 - 70 = 30%
b) BTKLra mdd tìm mct of HCl
c) tìm mdd sau pứ -mH2 nha bạn