
Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.






1.
\(n_{KNO_3}=0.15\cdot0.1=0.015\left(mol\right)\)
\(m_{KNO_3}=0.015\cdot101=1.515\left(g\right)\)
2.
\(m_{KOH}=200\cdot20\%=40\left(g\right)\)
Sau khi pha :
\(m_{dd_{KOH}}=\dfrac{40}{16\%}=250\left(g\right)\)
\(m_{H_2O\left(tv\right)}=250-200=50\left(g\right)\)
3.
\(n_{NaOH}=2\cdot1=2\left(mol\right)\)
Sau khi pha :
\(V_{dd_{NaOH}}=\dfrac{2}{0.1}=20\left(l\right)\)
\(V_{H_2o\left(tv\right)}=20-2=18\left(l\right)\)
3)
$n_{KNO_3} = 0,15.0,1 = 0,015(mol)$
$m_{KNO_3} = 0,015.101 = 1,515(gam)$
4)
$m_{KOH} = 200.20\% = 40(gam)$
$m_{dd\ KOH\ 16\%} = \dfrac{40}{16\%} = 250(gam)$
$\Rightarrow m_{H_2O} = 250 -200= 50(gam)$
5)
$n_{NaOH} = 2.1 = 2(mol)$
$V_{dd\ NaOH} = \dfrac{2}{0,1} = 20(lít)$
$\Rightarrow V_{H_2O} = 20 - 2 = 18(lít)$
4)




 mình cần gấp
mình cần gấp
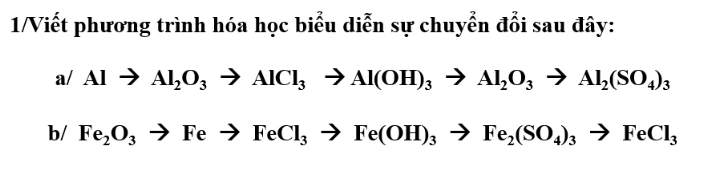

 giúp mình với mình cần gấp ạ
giúp mình với mình cần gấp ạ






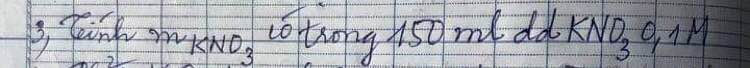
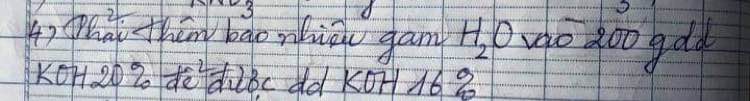




\(n_{H_2}=\dfrac{13,44}{22,4}=0,6mol\\ a)2Al+6HCl\rightarrow2AlCl_3+3H_2\)
0,4 1,2 0,4 0,6
\(Al_2O_3+6HCl\rightarrow2AlCl_3+3H_2O\\ b)m_{Al}=0,4.27=10,8g\\ \%m_{Al}=\dfrac{10,8}{21}\cdot100\%=51,43\%\\ \%m_{Al_2O_3}=100\%-51,43\%=48,57\%\\ c)m_{Al_2O_3}=21-10,8=10,2g\\ n_{Al_2O_3}=\dfrac{10,2}{102}=0,1mol\\ Al_2O_3+6HCl\rightarrow2AlCl_3+3H_2O\)
0,1 0,6 0,2 0,3
\(n_{HCl}=1,2+0,6=1,8mol\\ m_{HCl}=1,8.36,5=65,7g\\ C_{\%HCl}=\dfrac{65,7}{200}\cdot100\%=32,85\%\\ d)m_{dd}=21+200-0,6.2=219,8g\\ n_{AlCL_3}=0,4+0,2=0,6mol\\ m_{AlCl_3}=0,6.133,5=80,1g\\ C_{\%AlCl_3}=\dfrac{80,1}{219,8}\cdot100\%=36,44\%\)
ngầu dữ:)