Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

a.
\(4P+5O_2\underrightarrow{t^o}2P_2O_5\)
b.
\(CH_4+2O_2\underrightarrow{t^o}CO_2+2H_2O\)
c.
\(2Al+6HCl\rightarrow2AlCl_3+3H_2\)
d.
\(Zn+2HCl\rightarrow ZnCl_2+H_2\)

a) \(n_{Zn}=\dfrac{9,75}{65}=0,15\left(mol\right)\)
PTHH: Zn + 2HCl --> ZnCl2 + H2
0,15-->0,3------>0,15-->0,15
=> mHCl = 0,3.36,5 = 10,95 (g)
b)
mZnCl2 = 0,15.136 = 20,4 (g)
c)
PTHH: Fe2O3 + 3H2 --to--> 2Fe + 3H2O
0,05<---0,15------->0,1
=> mFe2O3 = 0,05.160 = 8 (g)
mFe = 0,1.56 = 5,6 (g)
a.b.\(n_{Zn}=\dfrac{m_{Zn}}{M_{Zn}}=\dfrac{9,75}{65}=0,15mol\)
\(Zn+2HCl\rightarrow ZnCl_2+H_2\)
0,15 0,3 0,15 0,15 ( mol )
\(m_{HCl}=n_{HCl}.M_{HCl}=0,3.36,5=10,95g\)
\(m_{ZnCl_2}=n_{ZnCl_2}.M_{ZnCl_2}=0,15.136-20,4g\)
c.\(Fe_2O_3+3H_2\rightarrow2Fe+3H_2O\)
0,05 0,15 0,1 ( mol )
\(m_{Fe_2O_3}=n_{Fe_2O_3}.M_{Fe_2O_3}=0,05.160=8g\)
\(m_{Fe}=n_{Fe}.M_{Fe}=0,1.56=5,6g\)

\(n_{Fe_2O_3}=\dfrac{12}{160}=0,075\left(mol\right)\\a, Fe_2O_3+3H_2\rightarrow\left(t^o\right)2Fe+3H_2O\\ b,n_{H_2}=3.0,075=0,225\left(mol\right)\\ V_{H_2\left(đkc\right)}=24,79.0,225=5,57775\left(l\right)\\ c,n_{Fe}=2.0,075=0,15\left(mol\right)\\ m_{Fe}=0,15.56=8,4\left(g\right)\)

a) $2Mg + O_2 \xrightarrow{t^o} 2MgO$
b)
$2Fe + O_2 \xrightarrow{t^o} 2FeO$
$4Fe + 3O_2 \xrightarrow{t^o} 2Fe_2O_3$
$3Fe + 2O_2 \xrightarrow{t^o} Fe_3O_4$
c)
$Zn + 2HCl \to ZnCl_2 + H_2$

\(n_{O_2}=\dfrac{3,7185}{24,79}=0,15\left(mol\right)\\ 3Fe+2O_2\rightarrow\left(t^o\right)Fe_3O_4\\ n_{Fe}=\dfrac{3}{2}.n_{O_2}=1,5.0,15=0,225\left(mol\right)\\ \Rightarrow m_{Fe}=0,225.56=12,6\left(g\right)\\ n_{Fe_3O_4}=\dfrac{n_{O_2}}{2}=\dfrac{0,15}{2}=0,075\left(mol\right)\\ \Rightarrow m_{Fe_3O_4}=232.0,075=17,4\left(g\right)\)

1) \(2KClO_3\rightarrow2KCl+3O_2\)
2)
a) \(CuO+H_2\xrightarrow[]{t^o}Cu+H_2O\)
b) \(2Na+2H_2O\rightarrow2NaOH+H_2\)
c) \(Zn+H_2SO_4\rightarrow ZnSO_4+H_2\)
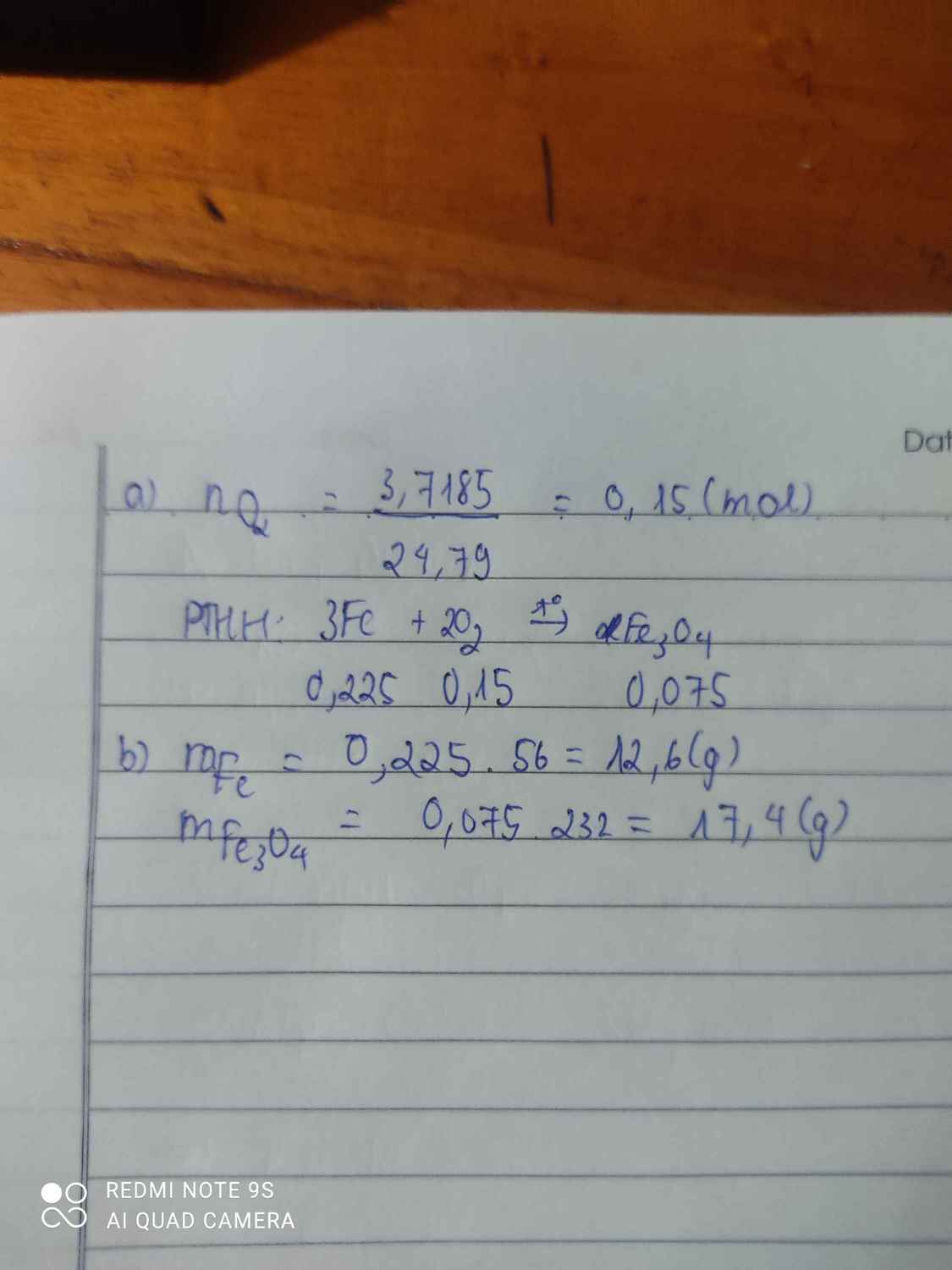
a.
\(Fe_2O_3+3H_2\underrightarrow{t^o}2Fe+3H_2O\)
b.
\(Mg+2HCl\rightarrow MgCl_2+H_2\)
c.
\(2KMnO_4\underrightarrow{p.h}K_2MnO_4+MnO_2+O_2\)