Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

PTHH :
\(Al+3HCl\rightarrow AlCl_3+1,5H_2\uparrow\)
x 3x
\(Fe+2HCl\rightarrow FeCl_2+H_2\uparrow\)
x 2x
Gọi số mol của Al là x => \(m_{Al}=27x\)
Gọi số mol của Fe là y => \(m_{Fe}=56y\)
Ta thấy cứ 1 mol HCl lại tạo ra 0,5 H2
Mà : \(n_{H_2}=\dfrac{4,48}{22,4}=0,2\left(mol\right)\)
\(\Rightarrow n_{HCl}=0,4\left(mol\right)\)
Ta có HPT : \(\left\{{}\begin{matrix}27x+56y=5,5\\3x+2y=0,4\end{matrix}\right.\)
Giải HPT ta được : \(x=0,1\left(mol\right),y=0,2\left(mol\right)\)
Ta có : \(m_{Al}=2,7\left(g\right)\)
\(m_{Fe}=11,2\left(g\right)\)
a ) \(\%Al=\dfrac{2,7}{11,2+2,7}.100\%\approx19,4\%\)
\(\Rightarrow\%Fe=100\%-19,4\%=80,6\%.\)
b ) Ta có : \(m_{HCl}=14,6\left(g\right)\)
Ta có : \(m_{dd\left(HCl\right)}=\dfrac{m_{HCl}}{C\%}=\dfrac{14,6}{\dfrac{14,6}{100}}=100\left(ml\right)\)
m\(m_{H_2}=0,4.2=0,8\)
\(m_{dd\left(sau\right)}=100+11,2+2,7-0,8=113,1\left(ml\right)\)
\(m_{AlCl_3}=0,1.133,5=13,35\left(g\right)\)
\(m_{FeCl_2}=0,2.127=25,4\left(g\right)\)
\(C\%=\dfrac{25,4+13,35}{113,1}.100\%\approx34,26\%.\)

Đặt \(n_{Al}=x(mol);n_{Fe}=y(mol)\)
\(\Rightarrow 27x+56y=5,5(1)\\ n_{H_2}=\dfrac{4,48}{22,4}=0,2(mol)\\ 2Al+6HCl\to 2AlCl_3+3H_2\\ Fe+2HCl\to FeCl_2+H_2\\ \Rightarrow 1,5x+y=0,2(2)\\ (1)(2)\Rightarrow x=0,1(mol);y=0,05(mol)\\ a,\begin{cases} \%_{Al}=\dfrac{0,1.27}{5,5}.100\%=49,09\%\\ \%_{Fe}=100\%-49,09\%=50,91\% \end{cases}\\ b,\Sigma n_{HCl}=3x+2y=0,4(mol)\\ \Rightarrow m_{dd_{HCl}}=\dfrac{0,4.36,5}{14,6\%}=100(g)\)
\(n_{AlCl_3}=0,1(mol);n_{FeCl_2}=0,05(mol)\\ \Rightarrow C\%_{AlCl_3}=\dfrac{0,1.133,5}{0,1.27+100-0,15.2}.100\%=13,04\%\\ C\%_{FeCl_2}=\dfrac{0,05.127}{0,05.56+100-0,05.2}.100\%=6,18\%\)

thôi thì mình làm cho bn vậy, câu a ko làm dc đâu, làm câu b thôi, làm sao biết dc chất nào dư khi chỉ có số mol 1 chất?
nK2SO3=0.1367(mol)
mddH2SO4=Vdd.D=200.1,04=208(g)
K2SO3+H2SO4-->K2SO4+H2O+SO2
0.1367----0.1367----0.1367---------0.1367 (mol)
mddspu=100+208-0,1367.64=299.2512(g) ; mK2SO4=0,1367.174=23.7858(g)
==>C%=23.7858.100/299.512=7.94%
2)pt bn tự ghi nhé
ta có hệ pt: 56a+27b=11 và a+3b/2=8.96/22.4==>a=0.1, b=0.2
==>%Fe=0.1x56x100/11=50.9%
%Al=100%-50.9%=49.1%
b)nH2SO4= 0.7(mol)==>VddH2SO4=0.7/2=0.35(L)

\(\text{Đặt }n_{Al}=x(mol);n_{Fe}=y(mol)\\ \Rightarrow 27x+56y=13,9(1)\\ n_{H_2}=\dfrac{7,84}{22,4}=0,35(mol)\\ a,PTHH:2Al+6HCl\to 2AlCl_3+3H_2(1)\\ Fe+2HCl\to FeCl_2+H_2(2)\\ b,\text{Từ 2 PT: }1,5x+y=0,35(2)\\ (1)(2)\Rightarrow x=0,1(mol);y=0,2(mol)\\ \Rightarrow m_{Al}=0,1.27=2,7(g)\\ m_{Fe}=0,2.56=11,2(g)\)
\(c,n_{HCl(1)}=3n_{Al}=0,3(mol);n_{AlCl_3}=0,1(mol);n_{H_2(1)}=0,15(mol)\\ \Rightarrow m_{dd_{HCl(1)}}=\dfrac{0,3.36,5}{14,6\%}=75(g)\\ \Rightarrow C\%_{AlCl_3}=\dfrac{0,1.133,5}{2,7+75-0,15.2}.100\%=17,25\%\)
\(n_{HCl(2)}=2n_{Fe}=0,4(mol);n_{FeCl_2}=n_{H_2(2)}=n_{Fe}=0,2(mol)\\ \Rightarrow m{dd_{HCl(2)}}=\dfrac{0,4.36,5}{14,6\%}=100(g)\\ \Rightarrow C\%_{FeCl_2}=\dfrac{0,2.127}{11,2+100-0,2.2}.100\%=22,92\%\)
a) 2Al + 6HCl --> 2AlCl3 + 3H2
Fe + 2HCl --> FeCl2 + H2
b) Gọi số mol Al, Fe lần lượt là a,b
=> 27a + 56b = 13,9
\(n_{H_2}=\dfrac{7,84}{22,4}=0,35\left(mol\right)\)
2Al + 6HCl --> 2AlCl3 + 3H2
a----->3a--------->a------->1,5a______(mol)
Fe + 2HCl --> FeCl2 + H2
b------>2b-------->b----->b__________(mol)
=> 1,5a + b = 0,35
=> \(\left\{{}\begin{matrix}a=0,1=>m_{Al}=0,1.27=2,7\left(g\right)\\b=0,2=>m_{Fe}=0,2.56=11,2\left(g\right)\end{matrix}\right.\)
c) nHCl = 3a + 2b = 0,7 (mol)
=> mHCl = 0,7.36,5 = 25,55(g)
=> \(m_{ddHCl}=\dfrac{25,55.100}{14,6}=175\left(g\right)\)
\(m_{dd\left(saupu\right)}=13,9+175-2.0,35=188,2\left(g\right)\)
\(\left\{{}\begin{matrix}m_{AlCl_3}=0,1.133,5=13,35\left(g\right)\\m_{FeCl_2}=0,2.127=25,4\left(g\right)\end{matrix}\right.\)
=> \(\left\{{}\begin{matrix}C\%\left(AlCl_3\right)=\dfrac{13,35}{188,2}.100\%=7,1\%\\C\%\left(FeCl_2\right)=\dfrac{25,4}{188,2}.100\%=13,5\%\end{matrix}\right.\)

\(a)2Al+6HCl\rightarrow2AlCl_3+3H_2\\ Fe+2HCl\rightarrow FeCl_2+H_2\\ b)n_{H_2}=\dfrac{5,6}{22,4}=0,25mol\\ n_{Al}=a;n_{Fe}=b\\ \left\{{}\begin{matrix}3a+b=0,25\\27a+56b=8,3\end{matrix}\right.\\ a=\dfrac{19}{470};b=\dfrac{121}{940}\\ \%m_{Al}=\dfrac{\dfrac{19}{470}\cdot27}{8,3}\cdot100=13,15\%\\ \%m_{Fe}=100-13,15=86,85\%\\ c)n_{HCl}=3\cdot\dfrac{19}{470}+2\cdot\dfrac{121}{940}=\dfrac{89}{235}mol\\ m_{ddHCl=}=\dfrac{\dfrac{89}{235}\cdot36,5}{7,3}\cdot100=189g\\ d)n_{AlCl_3}=n_{Al}=\dfrac{19}{470}mol\\ n_{Fe}=n_{FeCl_2}=\dfrac{121}{940}mol\)
\(m_{dd}=8,3+189-0,25.2=196,8g\\ C_{\%AlCl_3}=\dfrac{\dfrac{19}{470}\cdot133,8}{196,8}\cdot100=2,8\%\\ C_{\%FeCl_2}=\dfrac{\dfrac{121}{940}127}{196,8}\cdot100=8,3\%\)

a)
$Mg + 2HCl \to MgCl_2 + H_2$
$MgO + 2HCl \to MgCl_2 + H_2o$
b)
Theo PTHH : $n_{Mg} = n_{H_2} = \dfrac{4,48}{22,4} = 0,2(mol)$
$m_{Mg} = 0,2.24 = 4,8(gam)$
$m_{MgO} = m_{hh} - m_{Mg} = 12,8 - 4,8 = 8(gam)$
c)
$n_{MgO} = \dfrac{8}{40} = 0,2(mol)$
$n_{HCl} = 2n_{Mg} + 2n_{MgO} = 0,8(mol)$
$m_{dd\ HCl} = \dfrac{0,8.36,5}{14,6\%} = 200(gam)$

a) \(2Al+6HCl\rightarrow2AlCl_3+3H_2\)
\(Fe+2HCl\rightarrow FeCl_2+H_2\)
\(n_{H_2}=\dfrac{8,96}{22,4}=0,4\left(mol\right)\)
Gọi \(\left\{{}\begin{matrix}n_{Al}:x\left(mol\right)\\n_{Fe}:y\left(mol\right)\end{matrix}\right.\)
Ta có : \(\left\{{}\begin{matrix}27x+56y=11\\1,5x+y=0,4\end{matrix}\right.\)
=> x=0,2 ; y=0,1
\(\%m_{Al}=\dfrac{0,2.27}{11}.100==49,09\%\)
\(\%m_{Fe}=50,91\%\)
b) \(\Sigma n_{HCl}=3x+2y=0,8\left(mol\right)\)
=> \(V_{HCl}=\dfrac{0,8}{2}=0,4\left(lít\right)\)
c) \(CM_{AlCl_3}=\dfrac{0,2}{0,4}=0,5M\)
\(CM_{FeCl_2}=\dfrac{0,1}{0,4}=0,25M\)

a) $Zn + 2HCl \to ZnCl_2 + H_2$
$ZnO + 2HCl \to ZnCl_2 + H_2O$
b)
Theo PTHH : $n_{Zn} = n_{H_2} = \dfrac{4,48}{22,4} = 0,2(mol)$
$m_{Zn} = 0,2.65 = 13(gam)$
$m_{ZnO} = 21,1 - 13 = 8,1(gam)$
c) $n_{ZnO} = 0,1(mol)$
Theo PTHH : $n_{HCl} = 2n_{Zn} + 2n_{ZnO} = 0,6(mol)$
$m_{dd\ HCl} = \dfrac{0,6.36,5}{16,6\%} = 132(gam)$
d) $m_{dd\ sau\ pư} = 21,1 + 132 - 0,2.2 = 152,7(gam)$
$n_{ZnCl_2} = n_{Zn} + n_{ZnO} = 0,3(mol)$
$C\%_{ZnCl_2} = \dfrac{0,3.136}{152,7}.100\% = 26,72\%$
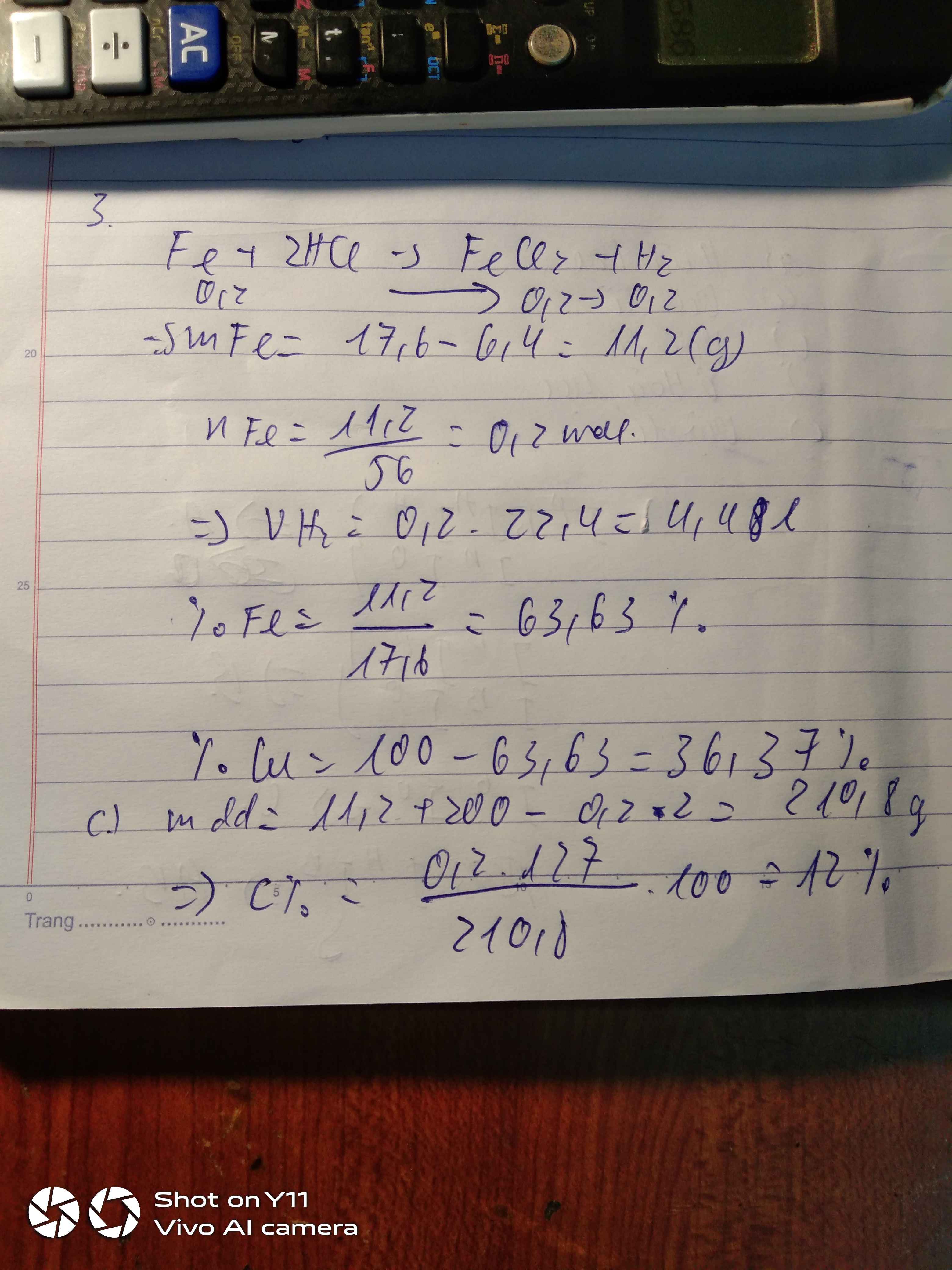
gọi x,y lần lượt là số mol của Al và Fe
PTHH 2Al + 6HCl ---> 2AlCl3 + 3H2
x 3x x 1,5x ( mol)
Fe + 2HCl ----> FeCl2 + H2
y 2y y y (mol)
ta có hệ PT : \(\left\{{}\begin{matrix}27x+56y=5,5\\1,5x+y=\dfrac{4,48}{22,4}=0,2\end{matrix}\right.\Rightarrow\left\{{}\begin{matrix}x=0,1\left(mol\right)\\y=0,05\left(mol\right)\end{matrix}\right.\)
=> mAl = n . M = 0,1 . 27 = 2,7 (g)
=> mFe = n . M = 0,05 . 56 =2,8
=> \(\%m_{Al}=\dfrac{2,7}{5,5}\cdot100=49,09\%\)
\(\rightarrow\%m_{Fe}=100-49,09=50,91\%\)
b) ta có : \(\sum n_{HCl}=3x+2y=0,4\left(mol\right)\Rightarrow m_{HCl}=0,4\cdot36,5=14,6\left(g\right)\)
\(\Rightarrow mdd_{HCl}=\dfrac{14,6\cdot100}{14,6}=100\left(g\right)\)
mddsau phản ứng = mAl+Fe + mddHCl - mH2
= 2,8 +2,7 + 100 - 0,2 . 2
= 105,1 (g)
\(m_{AlCl_3}=n\cdot M=0,1\cdot133,5=13,35\Rightarrow C\%=\dfrac{13,15}{105,1}\cdot100\%=12,7\%\)
\(m_{FeCl_2}=n.M=0,05\cdot127=6,35\Rightarrow C\%=\dfrac{6,35}{105,1}\cdot100=6,04\%\)
n H2 = 0,2 mol.
2Al + 6HCl -> 2AlCl3 + 3H2
a -> ....3a...........a........1,5a (mol)
Fe + 2HCl -> FeCl2 + H2
b -> ...2b..........b.........b (mol)
Theo đề bài, ta có hpt:
27a + 56b = 5,5
1,5a + b = 0,2
Giải hệ, được: a = 0,1; b = 0,05.
a/ m Al = 27a = 2,7g.
=> %Al = 49,09 %.
=> % Fe = 50,91 %. .
b/ m AlCl3 = 13,35g.
m FeCl2 = 6,35g.
Áp dụng đ. luật bảo toàn khối lượng:
mdd sau = mdd trước - m khí
.............= mdd HCl - m H2
.............= 100 - 2.0,2
.............= 99,6g
=> C% AlCl3 = 13,4 %.
.....C% FeCl2 = 6,38 %.