Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

V C O 2 = n C O 2 . 22,4 = 0,25.22,4 = 5,6 (l)
V O 2 = n O 2 .22,4 = 0,25.22,4 = 5,6 (l)
n
N
2
=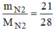 = 0,75(mol)
= 0,75(mol)
→ V N 2 = n N 2 .22,4 = 0,75.22,4= 16,8 (l)
n
C
O
2
=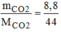 = 0,2 (mol)
= 0,2 (mol)
→ V C O 2 = n C O 2 . 22,4 = 0,2.22,4 = 4,48 (l)
n
H
2
=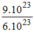 =1,5(mol)
=1,5(mol)
→ V H 2 = n H 2 . 22,4 = 1,5. 22,4 = 33,6 (l)
n
C
O
=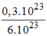 = 0,05(mol)
= 0,05(mol)
→ V C O = n C O . 22,4 = 0,05. 22,4 = 1,12 (l)

\(a,n_{CO_2}=\dfrac{m_{CO_2}}{M_{CO_2}}=\dfrac{11}{44}=0,25\left(mol\right)\\ b,n_{H_2}=\dfrac{9.10^{23}}{6.10^{23}}=1,5\left(mol\right)\\ V_{H_2\left(đktc\right)}=1,5.22,4=33,6\left(l\right)\)

ta có: nCl2=\(\frac{7,1}{71}=0,1mol\)
\(V_{Cl2}=0,1.22,4=2,24\left(l\right)\)
\(n_{CO2}=\frac{8,8}{44}=0,2\left(mol\right)\)
\(V_{CO2}=0,2.22,4=4,48\left(l\right)\)
\(n_{NO2}=\frac{4,6}{46}=0,1\left(mol\right)\)
\(V_{NO2}=0,1.22,4=2,24\left(l\right)\)
\(n_{h^2}=0,1+0,2+0,1=0,4\left(mol\right)\)
\(V_{h^2}=2,24+2,24+4,48=8,96\left(l\right)\)
b) ta có \(n_{O2}=\frac{16}{32}=0,5\left(mol\right)\)
\(n_{N2}=\frac{14}{28}=0,5\left(mol\right)\)
\(\Leftrightarrow n_{h^2}=0,5+0,5=1\left(mol\right)\)
c) vì \(S=n.6.10^{23}\Rightarrow n=\frac{S}{6.10^{23}}\)
\(n_{N2}=\frac{1,5.10^{23}}{6.10^{23}}=0,25\left(mol\right)\)
\(V_{N2}=0,25.22,4=5,6\left(l\right)\)
\(n_{CO2}=\frac{9.10^{23}}{6.10^{23}}=1,5\left(mol\right)\)
\(V_{CO2}=1,5.22,4=33,6\left(l\right)\)
chúc bạn học tốt like mình nha![]()

nSO2=m:M=6,4:64=0,1(mol)
VSO2=n.22,4=0,1.22,4=2,24(l)
nCO2=m:M=4,4:44=0,1(mol)
VCO2=n.22,4=0,1.22,4=2,24(l)
nH2=S:6.1023=1,2.1023:6.1023=0,2(mol)
VH2=n.22,4=0,2.22,4=4,48(l)
\(a.n_{SO_2}=\dfrac{m}{M}=\dfrac{6,4}{64}=0,1\left(mol\right)\\ n_{CO_2}=\dfrac{m}{M}=\dfrac{4,4}{44}=0,1\left(mol\right)\\ \Rightarrow n_{hh}=n_{SO_2}+n_{CO_2}=0,1+0,1=0,2\left(mol\right)\\ \Rightarrow V_{hh}=n.22,4=0,2.22,4=4,48\left(l\right)\)
\(n_{H_2}=\dfrac{1,2.10^{23}}{6.10^{23}}=0,2\left(mol\right)\Rightarrow V_{H_2}=n.22,4=0,2.22,4=4,48\left(l\right)\)

a) VCO2 = 1.22,4 = 22,4l.
VH2 = 2.22,4 = 44,8l.
VO2 = 1,5 .22,4 = 33,6l.
b) Vhh = 22,4.(0,25 + 1,25) = 33,6l.

a.
\(m_{Al}=0.5\cdot27=13.5\left(g\right)\)
\(m_{CO_2}=\dfrac{6.72}{22.4}\cdot44=13.2\left(g\right)\)
\(m_{N_2}=\dfrac{5.6}{22.4}\cdot28=7\left(g\right)\)
\(m_{CaCO_3}=0.25\cdot100=25\left(g\right)\)
b.
\(m_{hh}=\dfrac{3.36}{22.4}\cdot2+\dfrac{5.6}{22.4}\cdot28+0.2\cdot44=16.1\left(g\right)\)
A
\(V_{CO_2}=n\cdot22,4=0,2\cdot22,4=4,48\left(l\right)\\ n_{O_2}=n\cdot22,4=0,25\cdot22,4=5,6\left(l\right)\)
B
\(n_{N_2}=\dfrac{m}{M}=\dfrac{21}{14\cdot2}=0,75\left(mol\right)\\ V_{N_2}=n\cdot22,4=0,75\cdot22,4=16,8\left(l\right)\)
\(n_{CO_2}=\dfrac{m}{M}=\dfrac{8,8}{12+16\cdot2}=0,2\left(mol\right)\\ V_{CO_2}=n\cdot22,4=0,2\cdot22,4=4,48\left(l\right)\)
C
\(n_{H_2}=\dfrac{9\cdot10^{23}}{6\cdot10^{23}}=1,5\left(mol\right)\\ V_{H_2}=n\cdot22,4=1,5\cdot22,4=33,6\left(l\right)\)
\(n_{CO_2}=\dfrac{0,3\cdot10^{23}}{6\cdot10^{23}}=0,05\left(mol\right)\\ V_{CO_2}=n\cdot22,4=0,05\cdot22,4=1,12\left(l\right)\)