Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

a.\(n_{hh}=\dfrac{6,72}{22,4}=0,3mol\)
Gọi \(\left\{{}\begin{matrix}n_{CH_4}=x\\n_{C_2H_4}=y\end{matrix}\right.\)
\(CH_4+2O_2\rightarrow\left(t^o\right)CO_2+2H_2O\)
x x ( mol )
\(C_2H_4+3O_2\rightarrow\left(t^o\right)2CO_2+2H_2O\)
y 2y ( mol )
\(n_{CaCO_3}=\dfrac{40}{100}=0,4mol\)
\(Ca\left(OH\right)_2+CO_2\rightarrow CaCO_3+H_2O\)
0,4 0,4 ( mol )
Ta có:
\(\left\{{}\begin{matrix}x+y=0,3\\x+2y=0,4\end{matrix}\right.\) \(\Leftrightarrow\left\{{}\begin{matrix}x=0,2\\y=0,1\end{matrix}\right.\)
\(\%V_{CH_4}=\dfrac{0,2}{0,3}.100=66,67\%\)
\(\%V_{C_2H_4}=100\%-66,67\%=33,33\%\)
b.\(C_2H_4+Br_2\rightarrow C_2H_4Br_2\)
0,1 0,1 ( mol )
\(m_{Br_2}=0,1.160:10\%=160g\)

a)
nBr2 = 0,2.0,2 = 0,04 (mol)
nCaCO3 = \(\dfrac{10}{100}=0,1\left(mol\right)\)
PTHH: C2H4 + Br2 --> C2H4Br2
0,04<--0,04---->0,04
C2H4 + 3O2 --to--> 2CO2 + 2H2O
0,04--------------->0,08
CH4 + 2O2 --to--> CO2 + 2H2O
0,02<-------------0,02
Ca(OH)2 + CO2 --> CaCO3 + H2O
0,1<------0,1
=> \(\left\{{}\begin{matrix}\%V_{CH_4}=\dfrac{0,02}{0,02+0,04}.100\%=33,33\%\\\%V_{C_2H_4}=\dfrac{0,04}{0,02+0,04}.100\%=66,67\%\end{matrix}\right.\)
b) mC2H4Br2 = 0,04.188 = 7,52 (g)

Theo gt ta có: $n_{O_2}=0,6(mol);n_{hh}=0,25(mol)$
a, $CH_4+2O_2\rightarrow CO_2+2H_2O$
$C_2H_4+3O_2\rightarrow 2CO_2+2H_2O$
Gọi số mol CH4 và C2H4 lần lượt là a;b(mol)
Ta có: $a+b=0,25;2a+3b=0,6\Rightarrow a=0,15;b=0,1$
b, Suy ra $\%V_{CH_4}=60\%;\%V_{C_2H_4}=40\%$
c, Ta có: $n_{CaCO_3}=n_{CO_2}=0,15+0,1.2=0,35(mol)\Rightarrow m_{CaCO_3}=35(g)$
\(a)\\ CH_4 + 2O_2 \xrightarrow{t^o} CO_2 + 2H_2O\\ C_2H_4 + 3O_2 \xrightarrow{t^o} 2CO_2 + 2H_2O\\ b)\ V_{CH_4} = a(lít) ; V_{C_2H_4} = b(lít)\\ \Rightarrow a + b = 5,6(1)\\ V_{O_2} = 2a + 3b = 13,44(2)\\ (1)(2)\Rightarrow a = 3,36 ; b = 2,24\\ \%V_{CH_4} = \dfrac{3,36}{5,6}.100\% = 60\%\\ \%V_{C_2H_4} = 40\%\\ c) V_{CO_2} = a + 2b = 7,84(lít)\\\)
\(CO_2 + Ca(OH)_2 \to CaCO_3 + H_2O\\ n_{CaCO_3} = n_{CO_2} = \dfrac{7,84}{22,4} = 0,35(mol)\\ \Rightarrow m_{CaCO_3} = 0,35.100 = 35(gam)\)

\(n_{hhkhí}=\dfrac{2,8}{22,4}=0,125\left(mol\right)\\ n_{O_2}=\dfrac{6,72}{22,4}=0,3\left(mol\right)\\ Gọi\left\{{}\begin{matrix}n_{CH_4}=a\left(mol\right)\\n_{C_2H_2}=b\left(mol\right)\end{matrix}\right.\)
PTHH:
CH4 + 2O2 \(\underrightarrow{t^o}\) CO2 + 2H2O
a 2a a
2C2H2 + 5O2 \(\underrightarrow{t^o}\) 4CO2 + 2H2O
b 2,5b 2b
Hệ phương trình: \(\left\{{}\begin{matrix}a+b=0,125\\2a+2,5b=0,3\end{matrix}\right.\Leftrightarrow\left\{{}\begin{matrix}a=0,025\left(mol\right)\\b=0,1\left(mol\right)\end{matrix}\right.\)
\(\%V_{C_2H_2}=\dfrac{0,1}{0,125}=80\%\\ \%_{CH_4}=100\%-80\%=20\%\)
nCO2 = 2.0,025 + 2.0,1 = 0,25 (mol)
PTHH: Ca(OH)2 + CO2 -> CaCO3 + H2O
0,25 0,25
=> mCaCO3 = 0,25.100 = 25 (g)

\(n_{hh}=\dfrac{V_{hh}}{22,4}=\dfrac{1,68}{22,4}=0,075mol\)
Gọi \(\left\{{}\begin{matrix}n_{CH_4}=x\\n_{C_2H_4}=y\end{matrix}\right.\) \(\Rightarrow\left\{{}\begin{matrix}n_{CO_2\left(CH_4\right)}=x\\n_{CO_2\left(C_2H_4\right)}=2y\end{matrix}\right.\)
\(n_{CaCO_3}=\dfrac{m_{CaCO_3}}{M_{CaCO_3}}=\dfrac{10}{100}=0,1mol\)
\(Ca\left(OH\right)_2+CO_2\rightarrow CaCO_3+H_2O\)
x+2y x+2y ( mol )
Ta có:
\(\left\{{}\begin{matrix}22,4x+22,4y=1,68\\x+2y=0,1\end{matrix}\right.\) \(\Leftrightarrow\left\{{}\begin{matrix}x=0,05\\y=0,025\end{matrix}\right.\)
\(\%CH_4=\dfrac{0,05}{0,075}.100=66,66\%\)
\(\%C_2H_4=100\%-66,66\%=33,34\%\)
\(m_{CH_4}=0,05.16=0,8g\)
\(m_{C_2H_4}=0,025.28=0,7g\)

Gọi số mol của CH 4 trong hỗn hợp là X, của C 2 H 4 trong hỗn hợp là y.
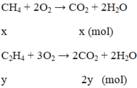
Ta có hệ: x + y = 0,15 mol và y = x + 2y = n CO 2 = n CaCO 3 = 0,2 mol
⇒ x = 0,1; y = 0,05
⇒ % V C H 4 = 66,67% ; % V C 2 H 4 = 33,33%.


a) Gọi \(\left\{{}\begin{matrix}n_{CH_4}=a\left(mol\right)\\n_{C_2H_4}=b\left(mol\right)\end{matrix}\right.\)
=> \(a+b=\dfrac{6,72}{22,4}=0,3\) (1)
\(n_{O_2}=\dfrac{17,92}{22,4}=0,8\left(mol\right)\)
PTHH: CH4 + 2O2 --to--> CO2 + 2H2O
a--->2a----------->a
C2H4 + 3O2 --to--> 2CO2 + 2H2O
b---->3b---------->2b
=> \(2a+3b=0,8\) (2)
(1)(2) => a = 0,1; b = 0,2
=> \(\left\{{}\begin{matrix}\%V_{CH_4}=\dfrac{0,1}{0,3}.100\%=33,33\%\\\%V_{C_2H_4}=\dfrac{0,2}{0,3}.100\%=66,67\%\end{matrix}\right.\)
b) \(n_{CO_2}=a+2b=0,5\left(mol\right)\)
PTHH: Ca(OH)2 + CO2 --> CaCO3 + H2O
0,5------>0,5
=> \(m_{CaCO_3}=0,5.100=50\left(g\right)\)