Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

a, \(n_{P_2O_5}=\dfrac{21,3}{142}=0,15\left(mol\right)\)
PT: \(P_2O_5+3H_2O\rightarrow2H_3PO_4\)
Theo PT: \(n_{H_3PO_4}=2n_{P_2O_5}=0,3\left(mol\right)\)
\(\Rightarrow m_{H_3PO_4}=0,3.98=29,4\left(g\right)\)
b, m dd sau pư = 21,3 + 300 = 321,3 (g)
\(\Rightarrow C\%_{H_3PO_4}=\dfrac{29,4}{321,3}.100\%\approx9,15\%\)

\(n_P=\dfrac{6,2}{31}=0,2\left(mol\right)\\ n_{O_2}=\dfrac{8,96}{22,4}=0,4\left(mol\right)\\ PTHH:4P+5O_2\underrightarrow{t^o}2P_2O_5\\ LTL:\dfrac{0,2}{4}< \dfrac{0,4}{5}\Rightarrow O_2dư\)
\(n_{O_2\left(pư\right)}=\dfrac{5}{4}n_P=\dfrac{5}{4}.0,2=0,25\left(mol\right)\\ n_{O_2\left(dư\right)}=0,4-0,25=0,15\left(mol\right)\)
\(n_{P_2O_5\left(lt\right)}=\dfrac{1}{2}n_P=\dfrac{1}{2}.0,2=0,1\left(mol\right)\\ m_{P_2O_5\left(lt\right)}=0,1.142=14,2\left(g\right)\\ m_{P_2O_5\left(tt\right)}=0,1.142.80\%=11,36\left(g\right)\)

a, \(4P+5O_2\underrightarrow{t^o}2P_2O_5\)
b, \(n_P=\dfrac{6,2}{31}=0,2\left(mol\right)\)
\(n_{O_2}=\dfrac{8,96}{22,4}=0,4\left(mol\right)\)
Xét tỉ lệ: \(\dfrac{0,2}{4}< \dfrac{0,4}{5}\), ta được O2 dư.
Theo PT: \(n_{O_2\left(pư\right)}=\dfrac{5}{4}n_P=0,25\left(mol\right)\Rightarrow n_{O_2\left(dư\right)}=0,4-0,25=0,15\left(mol\right)\)
\(\Rightarrow m_{O_2\left(dư\right)}=0,15.32=4,8\left(g\right)\)
c, Theo PT: \(n_{P_2O_5}=\dfrac{1}{2}n_P=0,1\left(mol\right)\Rightarrow m_{P_2O_5}=0,1.142=14,2\left(g\right)\)
d, \(m_{P_2O_5}=14,2.80\%=11,36\left(g\right)\)

\(n_P=\dfrac{3,1}{31}=0,1\left(mol\right)\)
\(n_{O_2}=\dfrac{4,48}{22,4}=0,2mol\)
4P + 5O2 \(\underrightarrow{t^o}\) 2P2O5
\(\dfrac{0,1}{4}< \dfrac{0,2}{5}\) => O2 dư, Photpho đủ
\(n_{O_2}=0,2-0,04=0,16\left(mol\right)\)
\(m_{P_2O_5}=\) 0,05 . 142 = 7,1 ( g )

a) 4P + 5O2 --to--> 2P2O5
b) \(n_{P_2O_5}=\dfrac{34,08}{142}=0,24\left(mol\right)\)
4P + 5O2 --to--> 2P2O5
0,48<-0,6<------0,24
=> mO2 = 0,6.32 = 19,2 (g)
c)
C1: mP = 0,48.31 = 14,88(g)
C2:
Theo ĐLBTKL: mP + mO2 = mP2O5
=> mP = 34,08-19,2 = 14,88(g)
d)
VO2 = 0,6.22,4 = 13,44 (l)
=> Vkk = 13,44 :20% = 67,2 (l)

1) Gọi số mol P2O5 là a (mol)
PTHH: P2O5 + 3H2O --> 2H3PO4
a----------------->2a
\(m_{H_3PO_4\left(tổng\right)}=98.2a+\dfrac{10.200}{100}=196a+20\left(g\right)\)
mdd sau pư = 142a + 200 (g)
=> \(C\%_{dd.sau.pư}=\dfrac{196a+20}{142a+200}.100\%=17,93\%\)
=> a = 0,093 (mol)
=> mP2O5 = 0,093.142 = 13,206 (g)
2)
a) \(n_{O_2}=\dfrac{10,08}{22,4}=0,45\left(mol\right)\)
PTHH: 2KMnO4 --to--> K2MnO4 + MnO2 + O2
0,9<-----------0,45<----0,45<----0,45
=> \(m_{KMnO_4\left(Pư\right)}=0,9.158=142,2\left(g\right)\)
=> \(m_{KMnO_4\left(tt\right)}=\dfrac{142,2.100}{80}=177,75\left(g\right)\)
=> \(m=\dfrac{177,75.100}{90}=197,5\left(g\right)\)
b)
X \(\left\{{}\begin{matrix}m_{K_2MnO_4}=0,45.197=88,65\left(g\right)\\m_{MnO_2}=0,45.87=39,15\left(g\right)\\m_{KMnO_4}=177,75-142,2=35,55\left(g\right)\\m_{tạp.chất}=197,5.10\%=19,75\left(g\right)\end{matrix}\right.\)

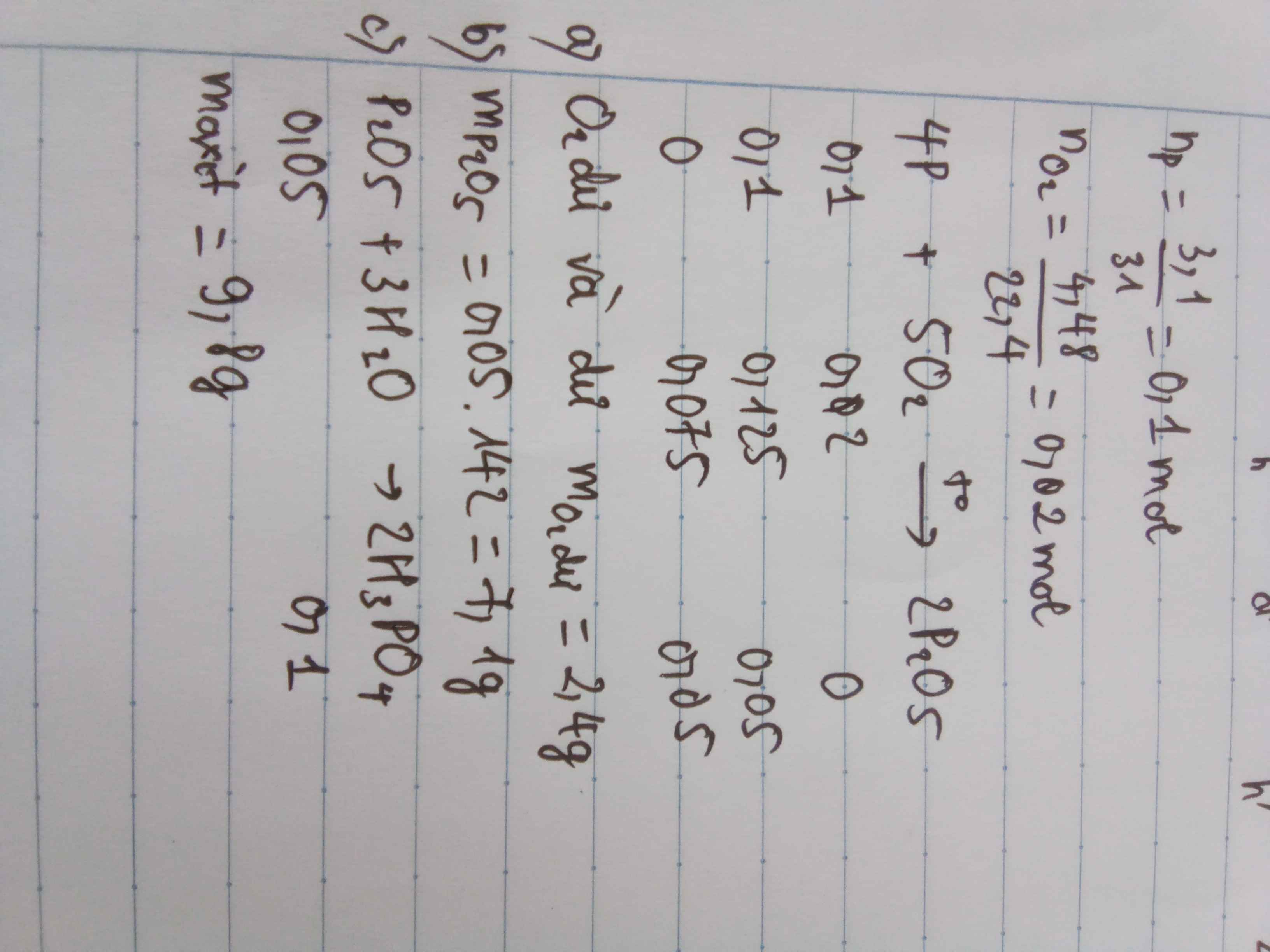
a) 4P + 5O2 --to--> 2P2O5
b) \(n_P=\dfrac{24,8}{31}=0,8\left(mol\right)\)
PTHH: 4P + 5O2 --to--> 2P2O5
0,8--------------->0,4
=> mP2O5 = 0,4.142 = 56,8 (g)
c)
PTHH: P2O5 + 3H2O --> 2H3PO4
0,4--------------->0,8
=> mH3PO4 = 0,8.98 = 78,4 (g)
mong mọi người giúp nhanh ạ