Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

a/ Ta có: \(n_{Mg}=\dfrac{4.8}{24}=0.2\left(mol\right)\)
PTHH:
\(2Mg+O_2\underrightarrow{t^o}2MgO\)
2 1
0.2 x
\(=>x=\dfrac{0.2\cdot1}{2}=0.1=n_{O_2}\)
\(=>V_{O_2\left(đktc\right)}=0.1\cdot22.4=2.24\left(l\right)\)
b/ \(2Mg+O_2\underrightarrow{t^o}2MgO\)
2 2
0.2 y
\(=>y=\left(0.2\cdot2\right):2=0.2=n_{MgO}\)
\(=>m_{MgO}=0.2\cdot\left(24+16\right)=8\left(g\right)\)

a) \(n_{Al}=\dfrac{10,8}{27}=0,4\left(mol\right)\)
PTHH: 2Al + 6HCl --> 2AlCl3 + 3H2
0,4------------>0,4---->0,6
=> \(V_{H_2}=0,6.22,4=13,44\left(l\right)\)
b)
\(m_{AlCl_3}=0,4.133,5=53,4\left(g\right)\)
c)
PTHH: CuO + H2 --to--> Cu + H2O
0,6------>0,6
=> mCu = 0,6.64 = 38,4 (g)

2Zn+O2-to>2ZnO
0,1---0,05----0,1
n Zn=0,1 mol
nO2=0,025 mol
=>VO2=0,05.22,4=1,12l
=>mZnO=0,1.81=8,1g
c)Zn dư
=>m ZnO=0,05.81=4,05g
2Zn+O2-to>2ZnO
0,1---0,05----0,1
n Zn=6,5/65=0,1 mol
n O2=0,8/32=0,025 mol
=>VO2=0,05.22,4=1,12l
=>mZnO=0,1.81=8,1g
c)Zn dư
=>m ZnO=0,05.81=4,05g

nP2O5= 28,4/ 142=0,2(mol)
PTHH: 4P + 5 O2 -to-> 2 P2O5
a) nP=4/2 . nP2O5= 2. 0,2=0,4(mol)
=>mP=31.0,4=12,4(g)
b) nO2=5/2. 0,2=0,5(mol)
=>V(O2,đktc)=0,5.22,4=11,2(l)
Vì: Vkk=5.V(O2)
=>Vkk=5.11,2=56(l)
\(4P+5O_2\buildrel{{t^o}}\over\longrightarrow 2P_2O_5\\ n_{P_2O_5}=\frac{28,4}{142}=0,2(mol)\\ n_P=2n_{P_2O_5}=0,2.2=0,4(mol)\\ a/ m_P=0,4.31=12,4(g)\\ b/\\ n_{O_2}=2,5.n_{P_2O_5}=2,5.0,2=0,5(mol)\\ V_{O_2}=0,5.22,4=11,2(l)\\ V_{kk}=5.V_{O_2}=11,2.5=56(l) \)

\(n_{KMnO4}=\dfrac{15,8}{158}=0,1\left(mol\right)\)
a) Pt : \(2KMnO_4\underrightarrow{t^o}K_2MnO_4+MnO_2+O_2|\)
2 1 1 1
0,1 0,05
b) \(n_{O2}=\dfrac{0,1.1}{2}=0,05\left(mol\right)\)
\(V_{O2\left(dktc\right)}=0,05.22,4=1,12\left(l\right)\)
c) \(n_{Fe}=\dfrac{5,6}{56}=0,1\left(mol\right)\)
Pt : \(3Fe+2O_2\underrightarrow{t^o}Fe_3O_4|\)
3 2 1
0,1 0,05 0,025
Lập tỉ số so sánh : \(\dfrac{0,1}{3}>\dfrac{0,05}{2}\)
⇒ Fe dư , O2 phản ứng hết
⇒ Tính toán dựa vào số mol của O2
\(n_{Fe3O4}=\dfrac{0,05.1}{2}=0,025\left(mol\right)\)
⇒ \(m_{Fe3O4}=0,025.232=5,8\left(g\right)\)
Chúc bạn học tốt

\(n_{Zn}=\dfrac{m_{Zn}}{M_{Zn}}=\dfrac{19,5}{65}=0,3mol\)
\(Zn+\dfrac{1}{2}O_2\rightarrow\left(t^o\right)ZnO\)
1 1/2 1 (mol)
0,3 0,15 0,3 ( mol )
PƯ trên thuộc loại phản ứng hóa hợp
\(m_{ZnO}=n_{ZnO}.M_{ZnO}=0,3.81=24,3g\)
\(V_{O_2}=n_{O_2}.22,4=0,15.22,4=3,36l\)

a) nFe= 0,25(mol)
PTHH: Fe + H2SO4 -> FeSO4 + H2
0,25______0,25______0,25__0,25(mol)
b) V(H2,đktc)=0,25.22,4=5,6(l)
c) mH2SO4= 0,25.98= 24,5(g)

\(n_{Al}=\dfrac{5,4}{27}=0,2\left(mol\right)\\a, PTHH:4Al+3O_2\rightarrow\left(t^o\right)2Al_2O_3\\ b,n_{O_2}=\dfrac{3}{4}.n_{Al}=\dfrac{3.0,2}{4}=0,15\left(mol\right)\\ \Rightarrow V_{O_2\left(đktc\right)}=0,15.22,4=3,36\left(l\right)\\ c,2KMnO_4\rightarrow\left(t^o\right)K_2MnO_4+MnO_2+O_2\\ n_{KMnO_4}=2.n_{O_2}=2.0,15=0,3\left(mol\right)\\ \Rightarrow m_{KMnO_4}=158.0,3=47,4\left(g\right)\)

Bài 1:
a) nFe = \(\frac{25,2}{56}= 0,45\) mol
Pt: 3Fe + ..2O2 --to--> Fe3O4
0,45 mol-> 0,3 mol
VO2 = 0,3 . 22,4 = 6,72 (lít)
b) Pt: CH4 + ....2O2 --to--> CO2 + 2H2O
......0,15 mol<-0,3 mol
mCH4 = 0,15 . 16 = 2,4 (g)
Bài 2:
a) nAl = \(\frac{5,4}{27}= 0,2\) mol
Pt: 4Al + 3O2 --to--> 2Al2O3
0,2 mol->0,15 mol-->0,1 mol
mAl2O3 = 0,1 . 102 = 10,2 (g)
b) VO2 = 0,15 . 22,4= 3,36 (lít)
Mà: VO2 = 20%Vkk = 0,2Vkk
=> Vkk = \(\frac{VO2}{0,2}=\frac{3,36}{0,2}=16,8 \) (lít)
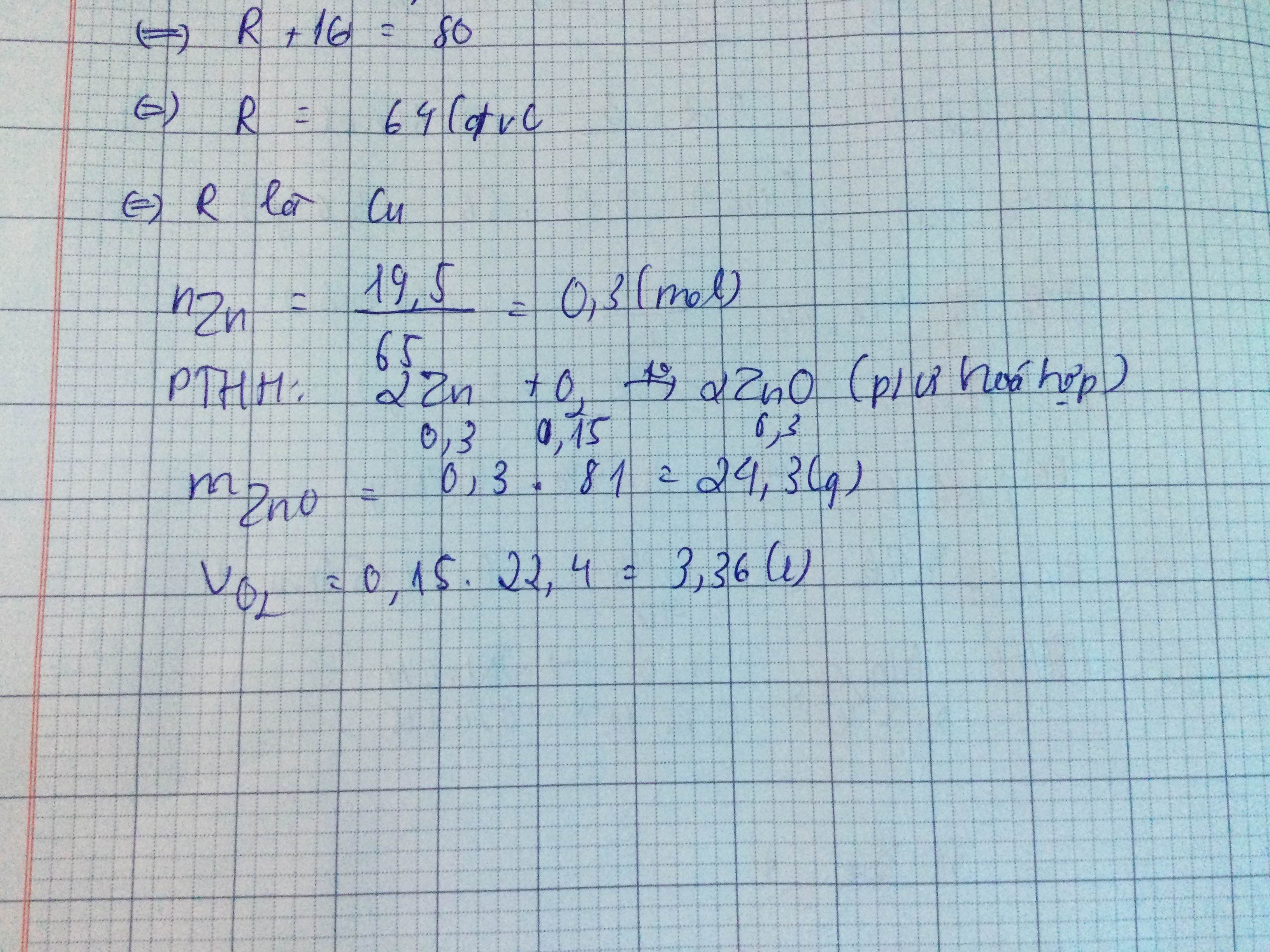
Ta có: \(n_P=\dfrac{6,2}{31}=0,2\left(mol\right)\)
a. \(PTHH:4P+5O_2\overset{t^o}{--->}2P_2O_5\)
Theo PT: \(n_{O_2}=\dfrac{5}{4}.n_P=\dfrac{5}{4}.0,2=0,25\left(mol\right)\)
\(\Rightarrow V_{O_2}=0,25.22,4=5,5\left(lít\right)\)
b. Theo PT: \(n_{P_2O_5}=\dfrac{1}{2}.n_P=\dfrac{1}{2}.0,2=0,1\left(mol\right)\)
\(\Rightarrow m_{P_2O_5}=0,1.142=14,2\left(g\right)\)
\(n_P=\dfrac{6,2}{31}=0,2\left(mol\right)\\ 4P+5O_2-^{t^o}\rightarrow2P_2O_5\\ a.n_{O_2}=\dfrac{5}{4}n_P=0,25\left(mol\right)\\ \Rightarrow m_{O_2}=0,25.32=8\left(g\right)\\ b.BTKLm_P+m_{O_2}=m_{P_2O_5}\\ \Rightarrow m_{P_2O_5}=6,2+8=14,2\left(g\right)\)