Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

Ta có: \(n_{Fe}=\dfrac{28}{56}=0,5\left(mol\right)\)
a, PT: \(Fe+H_2SO_4\rightarrow FeSO_4+H_2\)
______0,5____0,5_____0,5_____0,5 (mol)
b, mH2SO4 = 0,5.98 = 49 (g)
c, mFeSO4 = 0,5.152 = 76 (g)
d, \(CuO+H_2\underrightarrow{t^o}Cu+H_2O\)
____0,5__0,5 (mol)
⇒ mCu = 0,5.64 = 32 (g)
Bạn tham khảo nhé!

500ml = 0,5l
\(n_{HCl}=0,2.0,5=0,1\left(mol\right)\)
a) Pt : \(Fe+2HCl\rightarrow FeCl_2+H_2|\)
1 2 1 1
0,05 0,1 0,05 0,05
b) \(n_{Fe}=\dfrac{0,1.1}{2}=0,05\left(mol\right)\)
⇒ \(m_{Fe}=0,05.56=2,8\left(g\right)\)
c) \(n_{H2}=\dfrac{0,1.1}{2}=0,05\left(mol\right)\)
\(V_{H2\left(dktc\right)}=0,05.22,4=1,12\left(l\right)\)
d) \(n_{FeCl2}=\dfrac{0,05.1}{1}=0,05\left(mol\right)\)
⇒ \(m_{FeCl2}=0,05.127=6,35\left(g\right)\)
Chúc bạn học tốt

\(n_{HCl}=0,2.0,5=0,1(mol)\\ a,Fe+2HCl\to FeCl_2+H_2\\ \Rightarrow n_{Fe}=n_{FeCl_2}=\dfrac{1}{2}n_{HCl}=0,05(mol)\\ b,m_{Fe}=0,05.56=2,8(g)\\ c,m_{FeCl_2}=0,05.127=6,35(g)\)

\(n_{H2}=\dfrac{3,36}{22,4}=0,15\left(mol\right)\)
a) \(Fe+2HCl\rightarrow FeCl_2+H_2|\)
1 2 1 1
0,15 0,15 0,15
b) \(n_{Fe}=\dfrac{0,15.1}{1}=0,15\left(mol\right)\)
⇒ \(m_{Fe}=01,5.56=8,4\left(g\right)\)
c) \(n_{FeCl2}=\dfrac{0,2.1}{1}=0,2\left(mol\right)\)
⇒ \(m_{FeCl2}=0,2.127=25,4\left(g\right)\)
Chúc bạn học tốt

\(n_{Mg}=\dfrac{6}{24}=0,25\left(mol\right)\\ Mg+2HCl\rightarrow MgCl_2+H_2\\ 0,25.........0,5.........0,25.......0,25\left(mol\right)\\ a.V_{H_2\left(đktc\right)}=0,25.22,4=5,6\left(l\right)\\ b.m_{HCl}=0,5.36,5=18,25\left(g\right)\\ c.n_{Fe_2O_3}=\dfrac{16}{160}=0,1\left(mol\right)\\ Fe_2O_3+3H_2\underrightarrow{^{to}}2Fe+3H_2O\\ Vì:\dfrac{0,25}{3}< \dfrac{0,1}{1}\\ \Rightarrow Fe_2O_3dư\\ n_{Fe}=\dfrac{2}{3}.0,25=\dfrac{1}{6}\left(mol\right)\\ \Rightarrow m_{Fe}=\dfrac{1}{6}.56\approx9,333\left(g\right)\)
a,\(n_{Mg}=\dfrac{6}{24}=0,25\left(mol\right)\)
PTHH: Mg + 2HCl → MgCl2 + H2
Mol: 0,25 0,5 0,25
\(\Rightarrow V_{H_2}=0,25.22,4=5,6\left(l\right)\)
b,\(m_{HCl}=0,5.36,5=18,25\left(g\right)\)
c,\(n_{Fe_2O_3}=\dfrac{16}{160}=0,1\left(mol\right)\)
PTHH: Fe2O3 + 3H2 → 2Fe + 3H2O
Mol: 0,25 \(\dfrac{1}{6}\)
Ta có: \(\dfrac{0,1}{1}>\dfrac{0,25}{3}\)⇒ Fe2O3 dư, H2 hết
\(m_{Fe}=\dfrac{1}{6}.56=9,33\left(g\right)\)

PTHH: \(Fe_3O_4+8HCl\rightarrow FeCl_2+2FeCl_3+4H_2O\)
Ta có: \(\left\{{}\begin{matrix}n_{Fe_3O_4}=\dfrac{23,2}{232}=0,1\left(mol\right)\\n_{HCl}=\dfrac{300\cdot3,65\%}{36,5}=0,3\left(mol\right)\end{matrix}\right.\)
Xét tỉ lệ: \(\dfrac{0,1}{1}>\dfrac{0,3}{8}\) \(\Rightarrow\) Fe3O4 còn dư, HCl p/ứ hết
\(\Rightarrow\left\{{}\begin{matrix}n_{Fe_3O_4\left(dư\right)}=0,0625\left(mol\right)\\n_{FeCl_2}=0,0375\left(mol\right)\\m_{FeCl_3}=0,075\left(mol\right)\end{matrix}\right.\) \(\Rightarrow\left\{{}\begin{matrix}m_{Fe_3O_4\left(dư\right)}=0,0625\cdot232=14,5\left(g\right)\\m_{muối}=0,0375\cdot127+0,075\cdot162,5=16,95\left(g\right)\end{matrix}\right.\)
nFe3O4= 23,2/232=0,1(mol); nHCl = (300.3,65%)/36,5= 0,3(mol)
a) PTHH: Fe3O4 + 8 HCl -> 2 FeCl3 + FeCl2 + 4 H2O
b) Ta có: 0,3/8 < 0,1/1
=> Fe3O4 dư, HCl hết, tính theo nHCl.
=> nFe3O4(p.ứ)= nFeCl2= nHCl/8=0,3/8= 0,0375(mol)
=> mFe3O4(dư)= (0,1- 0,0375).232=14,5(g)
c) nFeCl3= 2/8. 0,3= 0,075(mol)
=> mFeCl3= 0,075.162,5=12,1875(g)
mFeCl2= 0,0375. 127=4,7625(g)
=>m(muối)= 12,1875+ 4,7625= 16,95(g)

Tìm số mol Fe tham gia phản ứng :
n Fe = n H = 0,15 mol, suy ra m Fe = 8,4 gam.
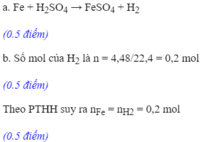

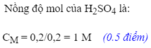
Ta có: \(n_{H_2}=\dfrac{1,22}{22,4}\approx0,05\left(mol\right)\)
a. PTHH: Fe + H2SO4 ---> FeSO4 + H2
b. Theo PT: \(n_{Fe}=n_{H_2}=0,05\left(mol\right)\)
=> \(m_{Fe}=0,05.56=2,8\left(g\right)\)
c. Theo PT: \(n_{H_2SO_4}=n_{Fe}=0,05\left(mol\right)\)
=> \(m_{H_2SO_4}=0,05.98=4,9\left(g\right)\)