Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

Zn+2HCl->Zncl2+H2
0,4----0,8----0,4----0,4
n Zn=0,4 mol
VH2=0,4.22,4=8,96l
m ZnCl2=0,4.136=54,4g
2H2+O2-to>2H2O
0,4------0,2----0,4
n O2=0,2 mol
=>pứ hết
=>m H2O=0,4.18=7,2g
a.b.\(n_{Zn}=\dfrac{26}{65}=0,4mol\)
\(Zn+2HCl\rightarrow ZnCl_2+H_2\)
0,4 0,4 0,4 ( mol )
\(m_{ZnCl_2}=0,4.136=54,4g\)
\(V_{H_2}=0,4.22,4=8,96l\)
c.\(n_{O_2}=\dfrac{4,48}{22,4}=0,2mol\)
\(2H_2+O_2\rightarrow\left(t^o\right)2H_2O\)
0,4 = 0,2 ( mol )
0,4 0,2 0,4 ( mol )
\(m_{H_2O}=0,4.18=7,2g\)

Quy đổi hh Na và K và \(Na_2O\) và \(K_2O\) thành hh Na và K và O
\(n_{NaCl}=n_{Na}=\frac{22.23}{58.5}=0,38mol\)
\(n_{H_2}=\frac{464.2}{22.4}=0,11mol\)
Bảo toàn e: \(n_{Na}+n_K=2n_O+2n_{H_2}\)
\(\rightarrow0,38+n_K=2n_O+0,22\)
\(\rightarrow n_K-2n_O=-0,16\left(1\right)\)
BTKL: \(23n_{Na}+39n_K+16n_O=30,7\)
\(\rightarrow8,74+39n_K+16n_O=30,7\)
\(\rightarrow39n_K+16n_O=21,96\left(2\right)\)
Từ 1 và (2) \(\rightarrow\hept{\begin{cases}n_K=0,44\\n_O=0,3\end{cases}}\)
\(\rightarrow m_{KCl}=0,44.74,5=32,78g\)

PTHH: \(Zn+2HCl\rightarrow ZnCl_2+H_2\uparrow\)
Ta có: \(n_{Zn}=\dfrac{32,5}{65}=0,5\left(mol\right)\)
\(\Rightarrow\left\{{}\begin{matrix}n_{HCl}=1\left(mol\right)\\n_{H_2}=0,5\left(mol\right)\end{matrix}\right.\) \(\Rightarrow\left\{{}\begin{matrix}m_{ddHCl}=\dfrac{1\cdot36,5}{20\%}=182,5\left(g\right)\\V_{H_2}=0,5\cdot22,4=11,2\left(l\right)\end{matrix}\right.\)

* Tóm tắt:
Biết: mZn = 6,5 (g)
CM(dd.HCl) = 0,5M
Hỏi: a) VH2 = ?
b) Vdd HCl = ?
a) \(n_{Zn}=\dfrac{6,5}{65}=0,1\left(mol\right)\)
PTHH: Zn + 2HCl --> ZnCl2 + H2
0,1-->0,2------------->0,1
VH2 = 0,1.22,4 = 2,24 (l)
b) \(V_{\left(dd.HCl\right)}=\dfrac{0,2}{0,5}=0,4\left(l\right)\)

\(n_{Zn}=\dfrac{6,5}{65}=0,1\left(mol\right)\\
pthh:Zn+2HCl\rightarrow ZnCl_2+H_2\uparrow\)
0,1 0,1 0,1
\(V_{H_2}=0,1.22,4=2,24\left(l\right)\\
m_{ZnCl_2}=136.0,1=13,6\left(g\right)\)
\(m_{\text{dd}}=6,5+150-\left(0,1.2\right)=156,3\left(g\right)\\
C\%=\dfrac{13,6}{156,3}.100\%=8,7\%\)

\(n_{Zn}=\dfrac{3,25}{65}=0,05\left(mol\right)\)
a) Pt : \(Zn+2HCl\rightarrow ZnCl_2+H_2|\)
1 2 1 1
0,05 0,1 0,05
b) \(n_{H2}=\dfrac{0,05.1}{1}=0,05\left(mol\right)\)
\(V_{H2\left(dktc\right)}=0,05.22,4=1,12\left(l\right)\)
c) \(n_{HCl}=\dfrac{0,05.2}{1}=0,1\left(mol\right)\)
200ml = 0,2l
\(C_{M_{ddHCl}}=\dfrac{0,1}{0,2}=0,5\left(M\right)\)
Chúc bạn học tốt

Câu 1 :
\(n_{Zn}=\dfrac{16.575}{65}=0.255\left(mol\right)\)
\(Zn+2HCl\rightarrow ZnCl_2+H_2\)
\(0.255................0.255....0.255\)
\(m_{ZnCl_2}=0.255\cdot95=24.225\left(g\right)\)
\(V_{H_2}=0.255\cdot22.4=5.712\left(l\right)\)
Câu 2 :
Điều chế oxi :
- Nguyên liệu : KClO3, KMnO4,...
- Phương pháp : Nhiệt phân các hợp chất giàu oxi
Ví dụ : \(2KClO_3 \xrightarrow{t^o} 2KCl + 3O_2\)
Điều chế hidro :
- Nguyên liệu : Zn,Mg,... và dung dịch HCl,H2SO4
- Phương pháp : Cho kim loại tác dụng dd axit loãng
Ví dụ : \(Zn + 2HCl \to ZnCl_2 + H_2\)
Các thu hai khí này là phương pháp dời nước và đẩy không khí

PTPỨ: Zn + 2HCl ---> ZnCl2 + H2
nZn = 6,5/65 = 0,1 (mol)
Theo ptpứ: nHCl = 2nZn = 0,2 (mol)
=> mHCl = 0,2 x 36,5 = 7,3 (g)
nH2 = nZn = 0,1 (mol)
=> VH2(đktc) = 0,1 x 22,4 = 2,24 (l)
mdd HCl đã dùng = 7,3 x 7,3% = 0,5329(g)
Câu c bạn viết rõ ra đi ak. CM hay C% và của chất j
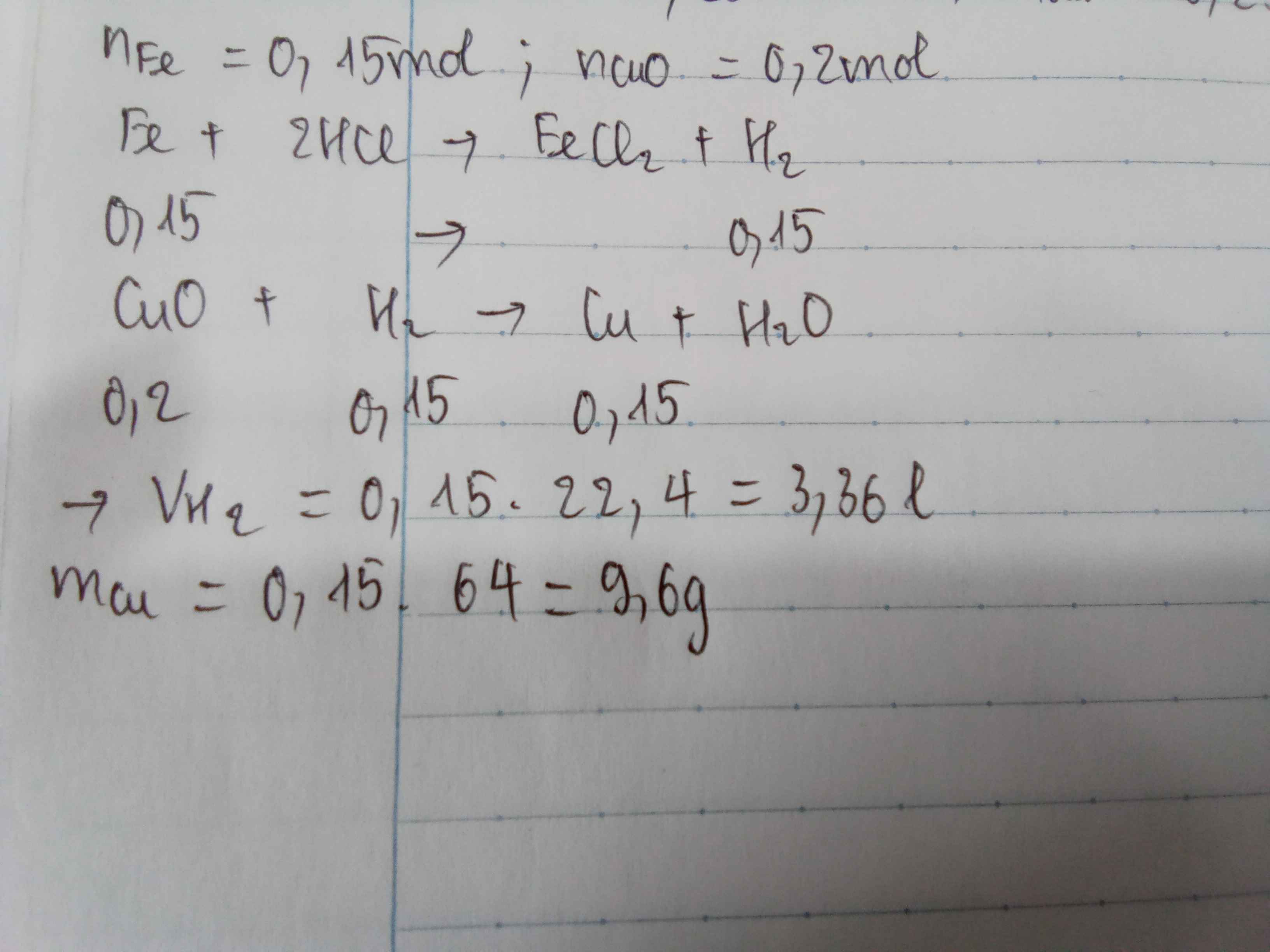

Zn+2Hcl->ZnCl2+H2
0,1------------------0,1
n Zn=0,1 mol
=>VH2=0,1.22,4=2,24l