Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

\(n_{H_2}=\dfrac{2,24}{22,4}=0,1\left(mol\right)\)
\(CH_3COOH+Na\rightarrow CH_3COONa+\dfrac{1}{2}H_2\)
0,1 0,1
\(C_2H_5OH+Na\rightarrow C_2H_5ONa+\dfrac{1}{2}H_2\)
0,1 0,1
Theo pthh có: \(n_A=2nH_2=2.0,1=0,2\left(mol\right)\)
Gọi x, y là số mol của rượu và axit có trong hh A.
có hệ: \(\left\{{}\begin{matrix}x+y=0,2\\60x+46y=10,6\end{matrix}\right.\)
=> x = y = 0,1
=> \(\left\{{}\begin{matrix}\%_{m_{CH_3COOH}}=\dfrac{60.0,1.100}{10,6}=56,6\%\\\%_{m_{C_2H_5OH}}=100-56,6=43,4\%\end{matrix}\right.\)
\(m_{muối}=m_{CH_3COONa}+m_{C_2H_5ONa}=82.0,1+68.0,1=15\left(g\right)\)
100 - 56,6 sao bằng 43,4%
Xem lại đơn vị

PT; C2H50H + Na \(\rightarrow C_2H_5ONa\) + \(\dfrac{1}{2}\) H2

\(C_2H_5OH+K_2CO_3\rightarrow\left(kopứ\right)\)
\(2CH_3COOH+K_2CO_3\rightarrow2CH_3COOK+CO_2+H_2O\)
2 1 2 1 1 (mol)
0,4 0,2 0,4 0,2 0,2 (mol)
\(nCO_2=\dfrac{4,48}{22,4}=0,2\left(mol\right)\)
\(mCH_3COOH=0,4.60=24\left(g\right)\)
\(mK_2CO_3=0,2.138=27,6\left(g\right)\)
\(mCH_3COOK=0,4.98=39,2\left(g\right)\)
\(mCO_2=0,2.44=8,8\left(g\right)\)
\(mdd=mCH_3COOH+mK_2CO_3+mCH_3COOK-mCO_2\)
\(=24+27,6+39,2-8,8=82\left(g\right)\)
\(C\%m_{CH_3COOH}=\dfrac{24.100}{82}=29,27\%\)
\(C\%m_{K_2CO_3}=\dfrac{27,6.100}{82}=33,66\%\)
câu thứ 2 bn tự lm cho bt:>

\(n_{CO_2}=\dfrac{2,24}{22,4}=0,1\left(mol\right)\\ a,PTHH:MgCO_3+H_2SO_4\rightarrow MgSO_4+H_2O+CO_2\uparrow\\ MgO+H_2SO_4\rightarrow MgSO_4+H_2O\\ \Rightarrow n_{MgCO_3}=n_{CO_2}=0,1\left(mol\right)\\ \Rightarrow m_{MgCO_3}=0,1\cdot84=8,4\left(g\right)\\ \Rightarrow\%_{MgCO_3}=\dfrac{8,4}{10,4}\cdot100\%\approx80,77\%\\ \Rightarrow\%_{MgO}=100\%-80,77\%=19,23\%\)
\(b,m_{MgO}=10,4-8,4=2\left(g\right)\\ \Rightarrow n_{MgO}=\dfrac{2}{40}=0,05\left(mol\right)\\ \Rightarrow\sum n_{H_2SO_4}=n_{MgCO_3}+n_{MgO}=0,15\left(mol\right)\\ \Rightarrow\sum m_{H_2SO_4}=0,15\cdot98=14,7\left(g\right)\\ \Rightarrow\sum m_{dd_{H_2SO_4}}=\dfrac{14,7}{9,8\%}=150\left(g\right)\\ \sum n_{MgSO_4}=\sum n_{H_2SO_4}=0,15\left(mol\right)\\ \Rightarrow\sum m_{MgSO_4}=0,15\cdot120=18\left(g\right)\\ \Rightarrow C\%_{MgSO_4}=\dfrac{18}{10,4+150-0,1\cdot44}\approx11,54\%\)

2NA+2CH3COOH-->2CH3COONA+H2
2NA+2C2H5OH--->2C2H5ONA+H2
ta có n H2=4,48/22,4=0,2 mol
gọi x là số mol của CH3COOH
y là số mol của C2H5OH
theo pt ta có hệ sau
\(\left\{{}\begin{matrix}\dfrac{1}{2}x+\dfrac{1}{2}y=0,2\\60x+46y=21,2\end{matrix}\right.\)\(\Leftrightarrow\)\(\left\{{}\begin{matrix}x=0,2\\y=0,2\end{matrix}\right.\)
suy ra mc2h5oh=0,2*46=9,2 g suy ra %C2H5OH=43.4%
%CH3COOH=56,6%

a, Gọi \(\left\{{}\begin{matrix}n_{C_2H_5OH}=a\left(mol\right)\\n_{CH_3COOH}=b\left(mol\right)\end{matrix}\right.\)
\(n_{H_2}=\dfrac{11,2}{22,4}=0,5\left(mol\right)\)
PTHH:
2C2H5OH + 2Na ---> 2C2H5ONa + H2
a---------------------------------------->0,5a
2CH3COOH + 2Na ---> 2CH3COONa + H2
b------------------------------------------------>0,5b
=> hệ pt \(\left\{{}\begin{matrix}46a+60b=48,8\\0,5a+0,5b=0,5\end{matrix}\right.\Leftrightarrow\left\{{}\begin{matrix}a=0,8\left(mol\right)\\b=0,2\left(mol\right)\end{matrix}\right.\)
\(\Rightarrow\left\{{}\begin{matrix}m_{C_2H_5OH}=0,8.46=36,8\left(g\right)\\m_{CH_3COOH}=0,2.60=12\left(g\right)\end{matrix}\right.\\ \Rightarrow\left\{{}\begin{matrix}\%m_{C_2H_5OH}=\dfrac{36,8}{48,8}.100\%=75,41\%\\\%m_{CH_3COOH}=100\%-75,41\%=24,59\%\end{matrix}\right.\)
b, PTHH:
\(C_2H_5OH+CH_3COOH\xrightarrow[t^o]{H_2SO_4đặc}CH_3COOC_2H_5+H_2O\)
LTL: 0,8 > 0,2 => Rượu dư
\(n_{CH_3COOC_2H_5\left(tt\right)}=0,2.85\%=0,17\left(mol\right)\\ m_{este}=0,17.88=14,96\left(g\right)\)
a.Gọi \(\left\{{}\begin{matrix}n_{C_2H_5OH}=x\\n_{CH_3COOH}=y\end{matrix}\right.\)
\(n_{H_2}=\dfrac{11,2}{22,4}=0,5mol\)
\(2C_2H_5OH+2Na\rightarrow2C_2H_5ONa+H_2\)
x 1/2 x ( mol )
\(2CH_3COOH+Na\rightarrow2CH_3COONa+H_2\)
y 1/2 y ( mol )
Ta có:
\(\left\{{}\begin{matrix}46x+60y=48,8\\\dfrac{1}{2}x+\dfrac{1}{2}y=0,5\end{matrix}\right.\) \(\Leftrightarrow\left\{{}\begin{matrix}x=0,8\\y=0,2\end{matrix}\right.\)
\(\rightarrow m_{C_2H_5OH}=0,8.46=36,8g\)
\(\rightarrow\left\{{}\begin{matrix}\%m_{C_2H_5OH}=\dfrac{36,8}{48,8}.100=75,4\%\\\%m_{CH_3COOH}=100\%-75,4\%=24,6\%\end{matrix}\right.\)
b.\(C_2H_5OH+CH_3COOH\rightarrow\left(H_2SO_4\left(đ\right),t^o\right)CH_3COOC_2H_5+H_2O\)
0,8 < 0,2 ( mol )
0,2 0,2 ( mol )
\(m_{CH_3COOC_2H_5}=0,2.88.85\%=14,96g\)

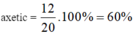

\(2CH_3COOH+CaCO_3\rightarrow\left(CH_3COO\right)_2Ca+CO_2+H_2O\)
\(n_{CO_2}=\dfrac{2,24}{22,4}=0,1\left(mol\right)\)
Theo PT: \(n_{CH_3COOH}=2n_{CO_2}=0,2\left(mol\right)\)
\(\Rightarrow\left\{{}\begin{matrix}\%m_{CH_3COOH}=\dfrac{0,2.60}{21,2}.100\%\approx56,6\%\\\%m_{C_2H_5OH}\approx43,4\%\end{matrix}\right.\)