Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

\(a,PTHH:FeCl_3+3NaOH\rightarrow Fe\left(OH\right)_3\downarrow+3NaCl\\ \Rightarrow n_{Fe\left(OH\right)_2}=n_{FeCl_3}=\dfrac{10,7}{107}=0,1\left(mol\right)\\ \Rightarrow m_{CT_{FeCl_3}}=0,1\cdot162,5=16,25\left(g\right)\\ \Rightarrow m_{dd_{FeCl_3}}=\dfrac{16,25\cdot100\%}{5\%}=325\left(g\right)\\ b,n_{NaOH}=n_{NaCl}=3n_{Fe\left(OH\right)_3}=0,3\left(mol\right)\\ \Rightarrow m_{NaOH}=0,3\cdot40=12\left(g\right)\\ m_{NaCl}=0,3\cdot58,5=17,55\left(g\right)\\ \Rightarrow m_{dd_{NaCl}}=325+150-10,7=464,3\left(g\right)\\ \Rightarrow C\%_{dd_{NaCl}}=\dfrac{17,55}{464,3}\cdot100\%\approx3,78\%\)

PTHH: \(2NaOH+CuSO_4\rightarrow Na_2SO_4+Cu\left(OH\right)_2\downarrow\)
a) Ta có: \(n_{CuSO_4}=\dfrac{320\cdot20\%}{160}=0,4\left(mol\right)=n_{Cu\left(OH\right)_2}\) \(\Rightarrow m_{Cu\left(OH\right)_2}=0,4\cdot98=39,2\left(g\right)\)
b) Theo PTHH: \(n_{NaOH}=2n_{CuSO_4}=0,8mol\) \(\Rightarrow m_{ddNaOH}=\dfrac{0,8\cdot40}{10\%}=320\left(g\right)\)
c) Theo PTHH: \(n_{Na_2SO_4}=n_{CuSO_4}=0,4mol\) \(\Rightarrow m_{Na_2SO_4}=0,4\cdot142=56,8\left(g\right)\)
Mặt khác: \(m_{dd}=m_{ddNaOH}+m_{ddCuSO_4}-m_{Cu\left(OH\right)_2}=600,8\left(g\right)\)
\(\Rightarrow C\%_{Na_2SO_4}=\dfrac{56,8}{600,8}\cdot100\%\approx9,45\%\)

nNa2CO3 = 10,6 / 106 = 0,1 (mol)
Na2CO3 + 2CH3COOH -> 2CH3COONa + H2O + CO2
0,1 0,2 0,2 0,1
mdd CH3COOH = 0,2 * 60 / 5 * 100 = 240 (gam)
CO2 + Ca(OH)2 -> CaCO3 + H2O
0,1 0,1
mCaCO3 = 0,1 * 100 = 10 (gam)
mdd = 240 + 10,6 - 0,1 * 44 = 246,2 (gam)
C% = 82 * 0,2 / 246,2 * 100% = 6,66%
\(n_{Na_2CO_3}=\dfrac{10,6}{106}=0,1\left(mol\right)\)
PTHH:
Na2CO3 + 2CH3COOH ---> 2CH3COONa + CO2 + H2O
0,1---------->0,2----------------->0,2--------------->0,1
CO2 + Ca(OH)2 ---> CaCO3 + H2O
0,1------------------------->0,1
=> \(\left\{{}\begin{matrix}m_{ddCH_3COOH}=\dfrac{0,2.60}{5\%}=240\left(g\right)\\m_{CaCO_3}=0,1.100=10\left(g\right)\end{matrix}\right.\)
\(m_{dd}=10,6+240-0,1.44=246,2\left(g\right)\\ C\%_{CH_3COONa}=\dfrac{82.0,2}{246,2}.100\%=6,66\%\)

Ta có: \(n_{H_2SO_4}=\dfrac{200.19,6\%}{98}=0,4\left(mol\right)\)
\(n_{Cu\left(OH\right)_2}=\dfrac{29,4}{98}=0,3\left(mol\right)\)
PT: \(CuO+H_2SO_4\rightarrow CuSO_4+H_2O\)
Dung dịch A gồm: CuSO4 và H2SO4 dư
\(H_2SO_4+2NaOH\rightarrow Na_2SO_4+2H_2O\)
\(CuSO_4+2NaOH\rightarrow Cu\left(OH\right)_2+Na_2SO_4\)
Đề có cho dữ kiện gì liên quan đến dd NaOH không bạn nhỉ?

\(n_{H_2SO_4}=\dfrac{200.19,6}{100.98}=0,4mol\\ CuO+H_2SO_4\rightarrow CuSO_4+H_2O\\ n_{CuSO_4\left(A\right)}=n_{CuO}=n_{H_2SO_4}=0,4mol\\ n_{Cu\left(OH\right)_2}=\dfrac{29,4}{98}=0,3mol\\ CuSO_4+2NaOH\rightarrow Cu\left(OH\right)_2+Na_2SO_4\\\Rightarrow\dfrac{0,4}{1}>\dfrac{0,3}{1}\Rightarrow CuSO_4.pư.không.hết\)
\(CuSO_4+2NaOH\rightarrow Cu\left(OH\right)_2+Na_2SO_4\)
0,3mol 0,6mol 0,3mol
\(m_{ddB}=0,4.80+200+0,6.40-29,4=226,6g\\ C_{\%Na_2SO_4\left(B\right)}=\dfrac{0,3.142}{226,6}\cdot100=18,8\%\)
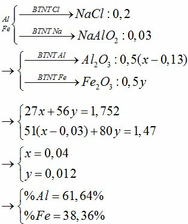
Fe(OH)3 sao khối lượng mol 107 được em?
\(m_{FeCl_3}=\frac{200.16,25\%}{100\%}=32,5\left(g\right)\)
\(n_{FeCl_3}=\frac{32,5}{162,5}=0,2\left(mol\right)\)
\(PTHH:FeCl_3+3NaOH\rightarrow Fe\left(OH\right)_3+3NaCl\)
\(\left(mol\right)\)____\(0,2\)______\(0,6\)________\(0,2\)_______\(0,6\)
a) \(m_{NaOH}=0,6.40=24\left(g\right)\)
\(m_{ddNaOH}=\frac{24.100\%}{20\%}=120\left(g\right)\)
b) \(m_{Fe\left(OH\right)_3}=0,2.107=21,4\left(g\right)\)
c) \(m_{NaCl}=0,6.58,5=35,1\left(g\right)\)
\(m_{ddNaCl}=m_{ddFeCl_3}+m_{ddNaOH}-m_{Fe\left(OH\right)_3}=200+120-21,4=298,6\left(g\right)\)
\(C\%_{NaCl}=\frac{35,1}{298,6}.100\%=11,75\%\)