Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

a. mCuO = % CuO . mhỗn hợp
= 42 % . 70 g = 29.4 g
-> nCuO = mCuO / MCuO
= 29.4 / 80 = ~ 0.37 mol
mFe3O4 = mhỗn hợp - mCuO
= 70 g - 29.4 g = 40.6 g
-> nFe3O4 = mFe3O4 / MFe3O4
= 40.6 / 232 = 0.175 mol
Ta có PƯHH :Fe3O4 +4H2->3Fe+4H2O (1)
CuO + H2 -> Cu + H2O (2)
-> Sau phản ứng thu được các kim loại : PUHH (1) là Fe , PUHH (2) là Cu
Theo PUHH (1) : nFe = 3/1 . nFe3O4
= 3.0,175 = 0.525mol
-> mFe = nFe . MFe = 0,525 . 56 = 29.4 g
Theo PUHH (2) : nCu = nCuO =~ 0.37 mol
-> mCu = nCu . MCu = 0,37 . 64 =~ 23.7 g
Mà mkim loại = mFe + mCu
= 23.7 + 29.4 = 53.1 g
b. Theo PUHH (1) : nH2 = 4/1 . nFe3O4
= 4.0,175 = 0.7mol
-> VH2(1) = nH2 . 22,4 = 0,7 . 22,4 = 15.7(l)
Theo PUHH (2) : nH2 = nCuO = 0.37 mol
-> VH2(2) = nH2 . 22,4 = 0,37.22,4 = 8.3(l)
Mà VH2 cần dùng = VH2(1) + VH2(2)
= 15.7 + 8.3 = 24 (l)
Minh sửa lại nha !
a)
(1) \(CuO+H_2\underrightarrow{t^o}Cu+H_2O\)
(2) \(Fe_3O_4+4H_2\underrightarrow{t^o}3Fe+4H_2O\)
\(m_{CuO}=\frac{42}{100}\cdot70=29,4\left(g\right)\)
\(\rightarrow n_{CuO}=\frac{29,4}{80}=0,3675\left(mol\right)\)
Theo pt (1):\(\Rightarrow n_{Cu}=n_{CuO}=0,3675\left(mol\right)\)
\(\Rightarrow m_{Cu}=0,3675\cdot64=23,52\left(g\right)\)
\(m_{Fe_3O_4}=70-29,4=40,6\left(g\right)\)
\(\rightarrow n_{Fe_3O_4}=\frac{40,6}{232}=0,175\left(mol\right)\)
Theo pt (2): \(\Rightarrow\) nFe = 3. 0,175 = 0,525 (mol)
\(\Rightarrow m_{Fe}=0,525\cdot56=29,4\left(g\right)\)
b)
Theo pt (1): \(n_{H_2}=n_{CuO}=0,3675\left(mol\right)\)
Theo pt (2): n\(H_2\) = 4 . 0,175 = 0,7 (mol)
\(\Rightarrow n_{H_2}\)Cần dùng = n\(H_2\)(1) + n\(H_2\)(2) = 0,3675 + 0,7 = 1,0675 (mol)
\(\Rightarrow V_{H_2}\)Cần dùng = 1,0675 . 22,4 = 23,912 (l)

Bài 2:
Gọi x,y lần lượt là số mol của Fe2O3, CuO
Pt: Fe2O3 + 3CO --to--> 2Fe + 3CO2
.......x................................2x
.....CuO + CO --to--> Cu + CO2
.......y............................y
Ta có hệ pt: \(\left\{{}\begin{matrix}160x+80y=24\\112x+64y=17,6\end{matrix}\right.\Leftrightarrow\left\{{}\begin{matrix}x=0,1\\y=0,1\end{matrix}\right.\)
P/s: tới đây tương tự bài 1, nếu bn ko hỉu thì nt hỏi mình nhé

nH2=0,448/22,4=0,02(mol)
Fe2O3+3H2--->2Fe+3H2O
0,01____<---______0,02
CuO+H2--->Cu+H2O(1)
Fe+2HCl--->FeCl2+H2
0,02_____<---_______0,02
mFe2O3=0,01.160=1,6(g)
=>%mFe2O3=1,6/2,4.100%=66,67%
=>%mCuO=100%-66,67%=33,33%
mFe=0,02.56=1,12(g)
nCuO=(2,4-1,6)/80=0,01(mol)
Theo pt:nCu=nCuO=0,01(mol)
=>mCu=0,01.64=0,64(g)


Gọi a là số mol H2 phản ứng
Bảo toàn H: \(n_{H_2O}=n_{H_2}=a\left(mol\right)\)
Bảo toàn khối lượng: \(24+2a=17,6+18a\)
\(\Rightarrow a=0,4\left(mol\right)\)
\(\Rightarrow m_{H_2O}=7,2\left(gam\right)\)

a) PTHH:
CuO + H2 \(\rightarrow\) Cu + H2O (I)
Fe2O3 + 3H2 \(\rightarrow\) 2Fe + 3H2O (II)
Khối lượng Fe2O3 tham gia phản ứng là:
50 . 80% = 40 (g)
Số mol Fe2O3 tham gia phản ứng là:
40 : (56.2 + 16.3) = 0,25 (mol)
Theo PTHH, số mol Fe thu được là:
0,25 . 2 = 0,5 (mol)
Khối lượng Fe thu được là:
0,5 . 56 = 28 (g)
Khối lượng CuO tham gia phản ứng là:
50 - 40 = 10 (g)
Số mol CuO tham gia phản ứng là:
10 : (64 + 16) = 0,125 (mol)
Theo PTHH, số mol Cu thu được là 0,125 mol.
Khối lượng Cu thu được là:
0,125 . 64 = 8 (g)
b) Theo PTHH, số mol H2 cần dùng ở phản ứng (I) là 0,125 mol.
Số mol H2 cần dùng ở phản ứng (II) là: 0,25 . 3 = 0,75 (mol)
Tổng thể tích H2 cần dùng là:
(0,125 + 0,75) . 22,4 = 19,6 (l)

Ta có:
\(n_{H2}=\frac{0,448}{22,4}=0,02\left(mol\right)\)
\(Fe_2O_3+3H_2\rightarrow2Fe+3H_2O\)
\(CuO+H_2\rightarrow Cu+H_2O\)
\(Fe+2HCl\rightarrow FeCl_2+H_2\)
\(\Rightarrow n_{Fe}=n_{H2}=0,02\left(mol\right)\)
\(\Rightarrow n_{Fe2O3}=\frac{1}{2}n_{Fe}=0,01\left(mol\right)\)
\(\Rightarrow m_{Fe2O3}=0,01.160=1,6\left(g\right)\)
\(\left\{{}\begin{matrix}\%_{Fe2O3}=\frac{1,6}{24}.100\%=6,67\%\\\%m_{CuO}=100\%-6,67\%=93,33\%\end{matrix}\right.\)
\(n_{Cu}=n_{CuO}=0,28\left(mol\right)\)
\(\Rightarrow m=m_{Fe}+m_{Cu}=0,02.56+0,28.64=19,04\left(g\right)\)
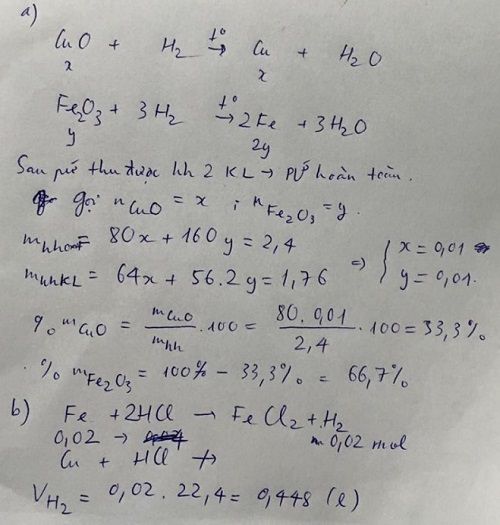
\(a) CuO + H_2 \xrightarrow{t^o} Cu + H_2O\\ Fe_2O_3 + 3H_2 \xrightarrow{t^o} 2Fe + 3H_2O\\ b) n_{CuO} = \dfrac{32.25\%}{80} = 0,1(mol)\\ n_{Fe_2O_3} = \dfrac{32-0,1.80}{160} = 0,15(mol)\\ n_{Cu} = n_{CuO} = 0,1(mol) \Rightarrow m_{Cu} = 0,1.64 = 6,4(gam)\\ n_{Fe} = 2n_{Fe_2O_3} = 0,3(mol) \Rightarrow m_{Fe} = 0,3.56 = 16,8(gam)\)