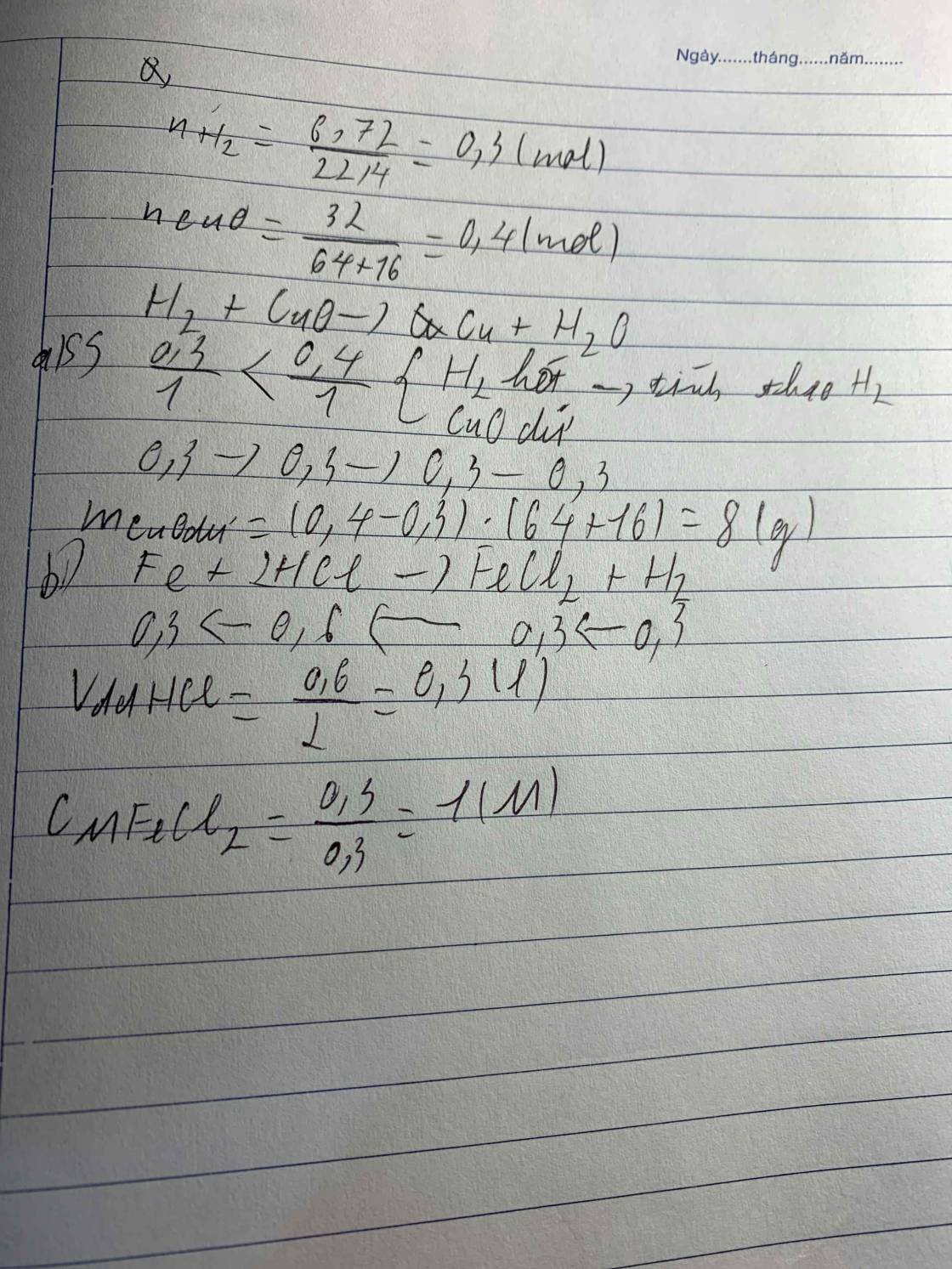Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

a) A gồm Cu, Fe
\(n_O=\dfrac{39,2-29,6}{16}=0,6\left(mol\right)\)
=> \(n_{H_2O}=0,6\left(mol\right)\)
=> \(n_{H_2}=0,6\left(mol\right)\)
=> \(V_{H_2}=0,6.22,4=13,44\left(l\right)\)
b)
Gọi \(\left\{{}\begin{matrix}n_{CuO}=a\left(mol\right)\\n_{Fe_xO_y}=b\left(mol\right)\end{matrix}\right.\)
=> 80a + b(56x + 16y) = 39,2
=> 80a + 56bx + 16by = 39,2 (1)
nO = 0,6 (mol)
=> a + by = 0,6
=> 80a + 80by = 48 (2)
\(n_{H_2}=\dfrac{6,72}{22,4}=0,3\left(mol\right)\)
PTHH: Fe + 2HCl --> FeCl2 + H2
0,3<-------------------0,3
=> nFe = bx = 0,3 (mol)
(2) - (1) => 64by - 56bx = 8,8
=> by = 0,4
Xét \(\dfrac{bx}{by}=\dfrac{x}{y}=\dfrac{0,3}{0,4}=\dfrac{3}{4}\)
=> CTHH: Fe3O4
Có: \(\left\{{}\begin{matrix}80a+232b=39,2\\a+4b=0,6\end{matrix}\right.\)
=> a = 0,2; b = 0,1
=> \(\left\{{}\begin{matrix}m_{CuO}=0,2.80=16\left(g\right)\\m_{Fe_3O_4}=0,1.232=23,2\left(g\right)\end{matrix}\right.\)

a, \(n_{H_2}=\dfrac{0,896}{22,4}=0,04\left(mol\right)\)
PTHH: Fe + 2HCl ---> FeCl2 + H2
0,04<-----------------------0,04
\(m_{Cu}=3,52-0,04.56=1,28\left(g\right)\)
Bảo toàn O: \(\left\{{}\begin{matrix}n_{O\left(oxit\right)}=\dfrac{4,8-3,52}{16}=0,08\left(mol\right)\\n_{O\left(CuO\right)}=n_{Cu}=\dfrac{1,28}{64}=0,02\left(mol\right)\end{matrix}\right.\)
=> \(n_{O\left(Fe_xO_y\right)}=0,08-0,02=0,06\left(mol\right)\)
PTHH:CuO + H2 --to--> Cu + H2O
0,02<--------------0,02
=> \(\left\{{}\begin{matrix}\%m_{CuO}=\dfrac{0,02.80}{4,8}.100\%=33,33\%\\\%m_{Fe_xO_y}=100\%-33,33\%=66,67\%\end{matrix}\right.\)
b, CTHH là FexOy
=> x : y = 0,04 : 0,06 = 2 : 3
=> CTHH là Fe2O3

a) PTHH : \(2Al+6HCl-->2AlCl_3+3H_2\) (1)
\(Fe+2HCl-->FeCl_2+H_2\) (2)
\(H_2+CuO-t^o->Cu+H_2O\) (3)
b) Ta có : \(m_{CR\left(giảm\right)}=m_{O\left(lay.di\right)}\)
=> \(m_{O\left(lay.di\right)}=32-26,88=5,12\left(g\right)\)
=> \(n_{O\left(lay.di\right)}=\frac{5,12}{16}=0,32\left(mol\right)\)
Theo pthh (3) : \(n_{H_2\left(pứ\right)}=n_{O\left(lay.di\right)}=0,32\left(mol\right)\)
=> \(tổng.n_{H_2}=\frac{0,32}{80}\cdot100=0,4\left(mol\right)\)
Đặt \(\hept{\begin{cases}n_{Al}=a\left(mol\right)\\n_{Fe}=b\left(mol\right)\end{cases}}\) => \(27a+56b=11\left(I\right)\)
Theo pthh (1) và (2) : \(n_{H_2\left(1\right)}=\frac{3}{2}n_{Al}=\frac{3}{2}a\left(mol\right)\)
\(n_{H_2\left(2\right)}=n_{Fe}=b\left(mol\right)\)
=> \(\frac{3}{2}a+b=0,4\left(II\right)\)
Từ (I) và (II) => \(\hept{\begin{cases}a=0,2\\b=0,1\end{cases}}\)
=> \(\hept{\begin{cases}m_{Al}=27\cdot0,2=5,4\left(g\right)\\m_{Fe}=56\cdot0,1=5,6\left(g\right)\end{cases}}\)

\(n_{Zn}=\dfrac{6.5}{65}=0.1\left(mol\right)\)
\(Zn+2HCl\rightarrow ZnCl_2+H_2\)
\(0.1.................................0.1\)
\(Đặt:n_{CuO\left(pư\right)}=x\left(mol\right)\)
\(CuO+H_2\underrightarrow{t^0}Cu+H_2O\)
\(x............x\)
\(m_{cr}=6-80x+64x=5.2\left(g\right)\)
\(\Rightarrow x=0.05\)
\(H\%=\dfrac{0.05}{0.075}\cdot100\%=66.67\%\)

PTHH: \(Fe_xO_y+yH_2\underrightarrow{t^o}xFe+yH_2O\)
\(Fe+2HCl\rightarrow FeCl_2+H_2\uparrow\)
Đặt \(\left\{{}\begin{matrix}n_{Fe\left(oxit\right)}=a\left(mol\right)=n_{H_2}\\n_{O\left(oxit\right)}=b\left(mol\right)\end{matrix}\right.\)
Ta có: \(m_{tăng}=m_{Fe}-m_{H_2}\) \(\Rightarrow56a-2a=3,24\) \(\Rightarrow a=n_{Fe}=0,06\left(mol\right)\)
Hỗn hợp D gồm \(\left\{{}\begin{matrix}n_{CO_2\left(dư\right)}=c\left(mol\right)\\n_{H_2O}=n_{O\left(oxit\right)}=b\left(mol\right)\end{matrix}\right.\)
Ta có hệ phương trình: \(\left\{{}\begin{matrix}c+b=0,1\\18b+2c=7,4\cdot2\cdot\left(b+c\right)\end{matrix}\right.\) \(\Leftrightarrow\left\{{}\begin{matrix}b=0,08\\c=0,02\end{matrix}\right.\)
\(\Rightarrow x:y=a:b=0,06:0,08=3:4\)
\(\Rightarrow\) Công thức cần tìm là Fe3O4

Gọi $n_{Al}= a(mol) ; n_{Fe} = b(mol) \Rightarrow 27a + 56b = 4,44(1)$
$4Al + 3O_2 \xrightarrow{t^o} 2Al_2O_3$
$3Fe + 2O_2 \xrightarrow{t^o} Fe_3O_4$
$Fe_3O_4 + 4H_2 \xrightarrow{t^o} 3Fe + 4H_2O$
B gồm : $Al_2O_3, Fe$
$n_{Al_2O_3} = \dfrac{1}{2}n_{Al} = 0,5a(mol)$
Suy ra: $0,5a.102 + 56b = 5,4(2)$
Từ (1)(2) suy ra a = 0,04 ; b = 0,06
$m_{Al} = 0,04.27 =1,08\ gam$
$m_{Fe} = 0,06.56 = 3,36\ gam$