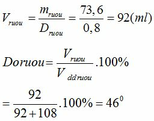Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

a, \(C_2H_6O+3O_2\underrightarrow{t^o}2CO_2+3H_2O\)
b, \(n_{CO_2}=\dfrac{4,48}{22,4}=0,2\left(mol\right)\)
Theo PT: \(n_{O_2}=\dfrac{3}{2}n_{CO_2}=0,3\left(mol\right)\)
\(\Rightarrow V_{O_2}=0,3.22,4=6,72\left(l\right)\)
c, \(n_{C_2H_6O}=\dfrac{1}{2}n_{CO_2}=0,1\left(mol\right)\)
\(\Rightarrow m_{C_2H_6O}=0,1.46=4,6\left(g\right)\)
\(\Rightarrow V_{C_2H_6O}=\dfrac{4,6}{0,8}=5,75\left(ml\right)\)
Độ rượu = \(\dfrac{5,75}{50}.100=11,5^o\)

Gọi \(\left\{{}\begin{matrix}n_{C_2H_5OH}=a\left(mol\right)\\n_{H_2O}=b\left(mol\right)\end{matrix}\right.\)
\(n_{H_2}=\dfrac{5,6}{22,4}=0,25\left(mol\right)\)
PTHH:
2C2H5OH + 2Na ---> 2C2H5ONa + H2
a --------------------------------------------> 0,5a
2H2O + 2Na ---> 2NaOH + H2
b --------------------------------> 0,5b
Hệ pt \(\left\{{}\begin{matrix}46a+18b=20,2\\0,5a+0,5b=0,25\end{matrix}\right.\Leftrightarrow\left\{{}\begin{matrix}a=0,4\left(mol\right)\\b=0,1\left(mol\right)\end{matrix}\right.\)
\(\rightarrow\left\{{}\begin{matrix}m_{C_2H_5OH}=0,4.46=18,4\left(g\right)\\m_{H_2O}=0,1.18=1,8\left(g\right)\end{matrix}\right.\rightarrow\left\{{}\begin{matrix}V_{C_2H_5OH}=\dfrac{18,4}{0,8}=23\left(ml\right)\\V_{H_2O}=\dfrac{1,8}{1}=1,8\left(ml\right)\end{matrix}\right.\)
=> Độ rượu là: \(\dfrac{23}{23+1,8}=92,74^o\)

a, \(C_2H_6O+3O_2\underrightarrow{t^o}2CO_2+3H_2O\)
b, \(n_{C_2H_6O}=\dfrac{23}{46}=0,5\left(mol\right)\)
Theo PT: \(n_{O_2}=3n_{C_2H_6O}=1,5\left(mol\right)\)
\(\Rightarrow V_{O_2}=1,5.22,4=33,6\left(l\right)\)
c, \(V_{C_2H_6O}=\dfrac{100.46}{100}=46\left(ml\right)\)
\(\Rightarrow m_{C_2H_6O}=46.0,8=36,8\left(g\right)\)
\(\Rightarrow n_{C_2H_6O}=\dfrac{36,8}{46}=0,8\left(mol\right)\)
PT: \(C_2H_5OH+Na\rightarrow C_2H_5ONa+\dfrac{1}{2}H_2\)
Theo PT: \(n_{H_2}=\dfrac{1}{2}n_{C_2H_5ONa}=0,4\left(mol\right)\)
\(\Rightarrow V_{H_2}=0,4.22,4=8,96\left(l\right)\)

Đáp án: A
Vì dung dịch rượu gồm rượu etylic và nước nên ta gọi:
n H 2 O = x m o l và n C 2 H 5 O H = y m o l
PTHH:
2 N a + 2 H 2 O → 2 N a O H + H 2 ↑ ( 1 )
x mol → 0,5.x mol
2 N a + 2 C 2 H 5 O H → 2 C 2 H 5 O N a + H 2 ↑
y mol → 0,5.y mol
Ta có hệ phương trình:
18 x + 46 y = 10 , 1 0 , 5 x + 0 , 5 y = 0 , 125 ⇒ x = 0 , 05 y = 0 , 2
V C 2 H 5 O H nguyên chất = m D = 0 , 2 . 46 0 , 8 = 11 , 5 m l
V H 2 O = m D = 10 , 1 - 9 , 2 1 = 0 , 9 m l
=> V d d r ư ợ u = V H 2 O + V C 2 H 5 O H = 0,9 + 11,5 = 12,4 ml
=> Độ rượu D 0 = V C 2 H 5 O H V d d r u o u . 100 = 11 , 5 12 , 4 . 100 = 92 , 74 0

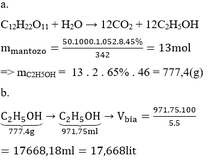
\(a,V_{C_2H_5OH}=\dfrac{10.96}{100}=9,6\left(ml\right)\\ m_{C_2H_5OH}=9,6.0,8=7,68\left(g\right)\\ n_{C_2H_5OH}=\dfrac{7,68}{46}=\dfrac{96}{575}\left(mol\right)\)
PTHH: 2C2H5OH + 2Na ---> 2C2H5ONa + H2
\(\dfrac{96}{575}\)------------------------------------->\(\dfrac{48}{575}\)
\(V_{H_2}=\dfrac{48}{575}.22,4=1,87\left(l\right)\)
\(b,V_{dd}=12+10,6=20,6\left(ml\right)\\ Đ_r=\dfrac{9,6}{20,6}.100=46,6^o\)

a, \(n_{CH_3COOH}=0,2.1=0,2\left(mol\right)\)
PT: \(Mg+2CH_3COOH\rightarrow\left(CH_3COO\right)_2Mg+H_2\)
Theo PT: \(n_{Mg}=n_{H_2}=\dfrac{1}{2}n_{CH_3COOH}=0,1\left(mol\right)\)
\(\Rightarrow m=m_{Mg}=0,1.24=2,4\left(g\right)\)
\(V=V_{H_2}=0,1.22,4=2,24\left(l\right)\)
b, \(C_2H_5OH+O_2\underrightarrow{^{mengiam}}CH_3COOH+H_2O\)
Theo PT: \(n_{C_2H_5OH}=n_{CH_3COOH}=0,2\left(mol\right)\)
\(\Rightarrow m_{C_2H_5OH}=0,2.46=9,2\left(g\right)\)
\(\Rightarrow V_{ddC_2H_5OH}=\dfrac{9,2}{0,8}=11,5\left(ml\right)\)

nH2 = 85,12 : 22,4 = 3,8 (mol) ; nH2O = VH2O.D = 108 (g) => nH2O = 108/18 = 6 (mol)
PTHH:
2Na + 2C2H5OH → 2C2H5ONa + H2↑
x → 0,5x (mol)
2Na + 2H2O → 2NaOH + H2↑
6 → 3 (mol)
Ta có: nH2 = 0,5x + 3 = 3,8
=> x = 1,6 (mol) = nC2H5OH
mC2H5OH = 1,6.46 = 73,6 (g)