Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

đặt nHCl=a(mol)
=>mHCl=36,5a(g)=>mddHCl=1000a(g)
mNaOH=200.10:100=20g=>nNaOH=0,5(mol)
PTHH:
HCl+NaOH-->NaCl+H2O
0,5__0,5____0,5
theo pt=>mddHCl=500g
=>mddsau pu=500+200=700g
tho pt,ta có:nNaCl=0,5 mol=>mNaCl=0,5.58,5=29,25g
=>C%(NaCl)=(29,25.100%):700=4,2%

NaOH+HCl--->NaCl+H2O
nNaOH=(200.10%)/40=0,5
=>nHCl=nNaOH=0,5
=>mddHCl=(0,5.36,5)/3,65%=500 g
ko biết có đúng ko nữa![]()

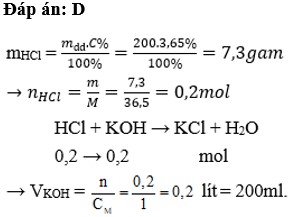
\(n_{HCl}=\dfrac{200.3,65}{100.36,5}=0,2mol\\ KOH+HCl\rightarrow KCl+H_2O\\ n_{HCl}=n_{KOH}=0,2mol\\ V_{KOH}=\dfrac{0,2}{1}=0,2l\)
Ta có: \(m_{HCl}=200.3,65\%=7,3\left(g\right)\Rightarrow n_{HCl}=\dfrac{7,3}{36,5}=0,2\left(mol\right)\)
PT: \(HCl+KOH\rightarrow KCl+H_2O\)
Theo PT: \(n_{KOH}=n_{HCl}=0,2\left(mol\right)\)
\(\Rightarrow V_{KOH}=\dfrac{0,2}{1}=0,2\left(l\right)\)

Trả lời:
mk chx hok wa lớp 9 nên ko giúp đc, thông cảm
HT^^
\(NaOH+HCl->NaCl+H_2O\)
a, \(m_{HCl}=\frac{C\%.m_{\text{dd}HCl}}{100\%}=\frac{7,3\%.200}{100\%}=14.6g\)
\(n_{HCl}=\frac{m_{HCl}}{M_{HCl}}=\frac{14.6}{36.5}=0.4\left(mol\right)\)
Theo PTHH ta có:\(n_{HCl}=n_{NaOH}=0.4\left(mol\right)\)
\(\Rightarrow m_{NaOH}=0,4.40=16g\)
\(\Rightarrow m_{\text{dd}NaOH}=\frac{m_{NaOH}.100\%}{C\%}=\frac{16.100\%}{10\%}=160g\)
b, Ta có \(\frac{C\%_{\text{dd}NaOH}-C\%_{\text{dd}mu\text{ối}}}{C\%_{\text{dd}mu\text{ối}}-C\%_{\text{dd}HCl}}=\frac{m_{\text{dd}HCl}}{m_{\text{dd}NaOH}}\)
\(\Leftrightarrow\frac{10\%-C\%}{C\%-7,3\%}=\frac{200}{160}=\frac{5}{4}\)\(\Rightarrow4\left(10\%-C\%\right)=5\left(C\%-7.3\%\right)\Leftrightarrow40\%-4C\%=5C\%-36.5\%\)
\(\Leftrightarrow9C\%=76.5\%\Leftrightarrow C\%=8,5\%\)

\(n_{HCl}=\dfrac{146.10\%}{100\%.36,5}=0,4(mol)\\ PTHH:NaOH+HCl\to NaCl+H_2O\\ \Rightarrow n_{NaOH}=0,4(mol)\\ \Rightarrow m_{dd_{NaOH}}=\dfrac{0,4.40}{10\%}=160(g)\)

a) \(m_{HCl}=200.10,95\%=21,9\left(g\right)\)
b) \(CaCO_3+2HCl\rightarrow CaCl_2+CO_2+H_2O\)
x_______2x________x____x(mol)
\(n_{HCl}=\dfrac{200.10,95\%}{36,5}=0,6\left(mol\right)\)
Dung dịch A phải có HCl dư mới có thể trung hòa được NaOH.
\(n_{NaOH}=0,05.2=0,1\left(mol\right)\)
\(NaOH+HCl\rightarrow NaCl+H_2O\)
y________y______y(mol)
Ta có hpt:
\(\left\{{}\begin{matrix}2x+y=0,6\\y=0,1\end{matrix}\right.\Leftrightarrow\left\{{}\begin{matrix}x=0,25\\y=0,1\end{matrix}\right.\)
\(\Rightarrow a=m_{CaCO_3}=100x=100.0,25=25\left(g\right)\\ V=V_{CO_2\left(đktc\right)}=22,4x=22,4.0,25=5,6\left(l\right)\)
c)
\(m_{ddA}=25+200-0,25.44=214\left(g\right)\\ C\%_{ddCaCl_2}=\dfrac{0,25.111}{214}.100\approx12,967\%\\ C\%_{ddHCl\left(dư\right)}=\dfrac{0,1.36,5}{214}.100\approx1,706\%\)

\(NaOH+HCl->NaCl+H_2O\\ 2NaOH+H_2SO_4->Na_2SO_4+2H_2O\\ a.V=\dfrac{0,1.1}{2}=0,05\left(L\right)\\ b.m_{ddH_2SO_4}=\dfrac{0,1.1.98}{2.0,1}=49\left(g\right)\)

nNaOH=200x10/100x40=0.5(mol)
NaOH+HCl-->NaCl+H2O
0.5------0.5 (mol)
=>mHCl=0.5x36.5=18.25(g)
=>mddHCl=18.25x100/3.65=500(g)