Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

a)
$C_2H_5OH + O_2 \xrightarrow{men\ giấm} CH_3COOH + H_2O$
V rượu = 57,5.12/100 = 6,9(lít) = 6900(cm3)
=> m rượu = 6900.0,8 = 5520(gam)
Theo PTHH :
n CH3COOH = n C2H5OH = 5520/46 = 120(mol)
m CH3COOH = 120.60 = 7200(gam)
b)
m dd giấm = 7200/4% = 180 000(gam)
\(V_r=57.5\cdot0.12=6.9\left(l\right)\)
\(m_{C_2H_5OH}=6.9\cdot0.8=5.52\left(g\right)\)
\(n_{C_2H_5OH}=\dfrac{5.52}{46}=0.12\left(mol\right)\)
\(n_{C_2H_5OH\left(pư\right)}=0.12\cdot92\%=0.1104\left(mol\right)\)
\(C_2H_5OH+O_2\underrightarrow{mg}CH_3COOH+H_2O\)
\(0.1104........................0.1104\)
\(m_{dd_{CH_3COOH}}=\dfrac{0.1104\cdot60}{4\%}=165.6\left(g\right)\)

a.\(V_{C_2H_5OH}=\dfrac{10.8}{100}=0,8ml\)
\(m_{C_2H_5OH}=0,8.0,8=1,6g\)
\(n_{C_2H_5OH}=\dfrac{1,6}{46}=0,034mol\)
\(C_2H_5OH+O_2\rightarrow\left(men.giấm\right)CH_3COOH+H_2O\)
0,034 0,034 ( mol )
\(m_{CH_3COOH}=0,034.60.80\%=1,632g\)
b.\(m_{dd_{CH_3COOH}}=\dfrac{1,632}{5\%}=32,64g\)

\(V_{C_2H_5OH\left(ng.chất\right)\left(1\right)}=0,4.200=80\left(ml\right)\\ Đặt:a=m_{C_2H_5OH\left(2\right)}\left(a>0\right)\\ \Rightarrow\dfrac{80+\dfrac{a}{0,8}}{200+\dfrac{a}{0,8}}.100\%=60\%\\ \Leftrightarrow a=80\left(gam\right)\)
Vậy phải pha thêm vào 200ml rượu etylic 40 độ với 80 gam rượu etylic để có được rượu etylic 60 độ

2 C 2 H 5 O H → C 4 H 6
n C 4 H 6 = 2/54 = 1/27 kmol
Vì H = 80% nên ta có:
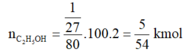
⇒ m C 2 H 5 O H = 5/54.46 = 115/27 kg
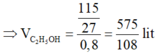
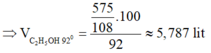
⇒ Chọn A.

\(C_2H_5OH+O_2\rightarrow\left(men.giấm\right)CH_3COOH+H_2O\\ V_{C_2H_5OH}=25.4\%=1\left(l\right)=1000\left(ml\right)\\ m_{C_2H_5OH}=1000.0,8=800\left(g\right)\\ m_{CH_3COOH\left(LT\right)}=\dfrac{800.60}{46}=\dfrac{48000}{46}\left(g\right)\\ m_{CH_3COOH\left(TT\right)}=\dfrac{48000}{46}:92\%=1134,2155\left(gam\right)\\ m_{ddCH_3COOH}=1135,2155:5\%=22684,31\left(g\right)\)

Ta có glucozo → 2C2H5OH + 2CO2
nrượu = 100 . 0,9 . 0,8 : 46 = 1,565 mol
=> mglucozo = 1,565 : 2 : 0,90 . 180 = 156,5 kg
\(V_{C_2H_5OH}=100.1000.90\%=9000\left(ml\right)\\ m_{C_2H_5OH}=9000.0,8=7200\left(g\right)\\ n_{C_2H_5OH}=\dfrac{7200}{46}=\dfrac{3600}{23}\left(mol\right)\)
PTHH: C6H12O6 -men rượu-> CO2 + C2H5OH
\(m_{C_6H_{12}O_6}=\dfrac{180.\dfrac{3600}{23}}{90\%}=\dfrac{720000}{23}\left(g\right)\)

a, \(n_{C_2H_4}=\dfrac{15,68}{22,4}=0,7\left(mol\right)\)
PT: \(C_2H_4+H_2O\underrightarrow{^{t^o,xt}}C_2H_5OH\)
Theo PT: \(n_{C_2H_5OH\left(LT\right)}=n_{C_2H_4}=0,7\left(mol\right)\)
Mà: H = 90%
\(\Rightarrow n_{C_2H_5OH\left(TT\right)}=0,7.90\%=0,63\left(mol\right)\)
\(\Rightarrow m_{C_2H_5OH\left(TT\right)}=0,63.46=28,98\left(g\right)\)
\(\Rightarrow V_{C_2H_5OH}=\dfrac{28,98}{0,8}=36,225\left(ml\right)\)
b, \(C_2H_5OH+O_2\underrightarrow{^{mengiam}}CH_3COOH+H_2O\)
Theo PT: \(n_{CH_3COOH}=n_{C_2H_5OH}=0,63\left(mol\right)\)
\(\Rightarrow m_{CH_3COOH}=0,63.60=37,8\left(g\right)\)
\(\Rightarrow m_{ddCH_3COOH}=\dfrac{37,8}{5\%}=756\left(g\right)\)


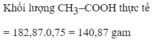
\(V_{C_2H_5OH}=\dfrac{1.1000.40}{100}=400\left(ml\right)\\ \rightarrow m_{C_2H_5OH\left(TT\right)}=400.0,8=320\left(g\right)\\ \rightarrow m_{C_2H_5OH\left(LT\right)}=\dfrac{320.100}{80}=400\left(g\right)\\ \rightarrow n_{C_2H_5OH\left(LT\right)}=\dfrac{400}{23}\left(mol\right)\)
PTHH: \(C_6H_{12}O_6\underrightarrow{\text{men rượu}}2C_2H_5OH+2CO_2\uparrow\)
\(\dfrac{200}{23}\)<-----------------\(\dfrac{400}{23}\)
\(\rightarrow m_{C_6H_{12}O_6}=\dfrac{200}{23}.180=\dfrac{36000}{23}\left(g\right)\)