Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

a, \(CH_4+2O_2\underrightarrow{t^o}CO_2+2H_2O\)
\(2C_2H_2+5O_2\underrightarrow{t^o}4CO_2+2H_2O\)
Ta có: \(n_{CH_4}+n_{C_2H_2}=\dfrac{6,72}{22,4}=0,3\left(mol\right)\left(1\right)\)
\(n_{CO_2}=n_{CH_4}+2n_{C_2H_2}=\dfrac{7,84}{22,4}=0,35\left(mol\right)\left(2\right)\)
Từ (1) và (2) \(\Rightarrow\left\{{}\begin{matrix}n_{CH_4}=0,25\left(mol\right)\\n_{C_2H_2}=0,05\left(mol\right)\end{matrix}\right.\)
\(\Rightarrow\left\{{}\begin{matrix}\%V_{CH_4}=\dfrac{0,25.22,4}{6,72}.100\%\approx83,33\%\\\%V_{C_2H_2}\approx16,67\%\end{matrix}\right.\)
Theo PT: \(n_{O_2}=2n_{CH_4}+\dfrac{5}{2}n_{C_2H_2}=0,625\left(mol\right)\Rightarrow m_{O_2}=0,625.32=20\left(g\right)\)

\(a,Gọi\left\{{}\begin{matrix}n_{CH_4}=a\left(mol\right)\\n_{C_2H_4}=b\left(mol\right)\\n_{C_2H_2}=c\left(mol\right)\end{matrix}\right.\\ n_{hhkhí}=0,4\left(mol\right)\\ n_{CO_2}=\dfrac{15,68}{22,4}=0,7\left(mol\right)\\ n_{Br_2}=\dfrac{64}{160}=0,4\left(mol\right)\\ PTHH:C_2H_4+Br_2\rightarrow C_2H_4Br_2\\ Mol:a\rightarrow a\\ C_2H_2+2Br_2\rightarrow C_2H_2Br_4\\ Mol:b\rightarrow2b\\ CH_4+2O_2\underrightarrow{t^o}CO_2+2H_2O\\ Mol:a\rightarrow2a\rightarrow a\)
\(C_2H_4+3O_2\underrightarrow{t^o}2CO_2+2H_2O\\ Mol:b\rightarrow3b\rightarrow2b\\ 2C_2H_2+5O_2\underrightarrow{t^o}4CO_2+2H_2O\\ Mol:c\rightarrow2,5c\rightarrow2c\\ Hệ.pt\left\{{}\begin{matrix}a+b+c=0,4\\b+2c=0,4\\a+2b+2c=0,7\end{matrix}\right.\Leftrightarrow\left\{{}\begin{matrix}a=0,1\left(mol\right)\\b=0,2\left(mol\right)\\c=0,1\left(mol\right)\end{matrix}\right.\)
\(\%V_{CH_4}=\%V_{C_2H_2}=\dfrac{0,1}{0,4}=25\%\\ \%V_{C_2H_4}=\dfrac{0,2}{0,4}=50\%\)
\(m_{CH_4}=0,1.16=1,6\left(g\right)\\ m_{C_2H_4}=28.0,2=5,6\left(g\right)\\ m_{C_2H_2}=0,1.26=2,6\left(g\right)\\ \%m_{CH_4}=\dfrac{1,6}{1,6+5,6+2,6}=16,32\%\\ \%m_{C_2H_4}=\dfrac{5,6}{1,6+5,6+2,6}=57,14\%\\ \%m_{C_2H_2}=100\%-16,32\%-57,14\%=26,54\%\)
\(b,PTHH:C_2H_5OH\rightarrow C_2H_4+H_2O\\ Mol:0,2\leftarrow0,2\\ m_{C_2H_5OH}=0,2.46=9,2\left(g\right)\)
Dài quá!!!

\(n_{hh}=\dfrac{3,36}{22,4}=0,15mol\)
\(n_{CO_2}=\dfrac{4,48}{22,4}=0,2mol\)
\(\left\{{}\begin{matrix}n_{CH_4}=x\left(mol\right)\\n_{C_2H_2}=y\left(mol\right)\end{matrix}\right.\)
\(CH_4+2O_2\rightarrow CO_2+2H_2O\)
\(C_2H_2+\dfrac{5}{2}O_2\rightarrow2CO_2+H_2O\)
Từ hai pt trên:\(\Rightarrow\left\{{}\begin{matrix}x+y=0,15\\x+2y=0,2\end{matrix}\right.\Rightarrow\left\{{}\begin{matrix}x=0,1\\y=0,05\end{matrix}\right.\)
\(\%V_{CH_4}=\dfrac{0,1}{0,1+0,05}\cdot100\%=66,67\%\)
\(\%V_{C_2H_2}=100\%-66,67\%=33,33\%\)
\(n_{CO_2}=\dfrac{V_{CO_2}}{22,4}=\dfrac{4,48}{22,4}=0,2mol\)
Gọi \(n_{CH_4}\) là x \(\Rightarrow V_{CH_4}=22,4x\)
\(n_{C_2H_2}\) là y \(\Rightarrow V_{C_2H_2}=22,4y\)
\(CH_4+2O_2\rightarrow\left(t^o\right)CO_2+2H_2O\)
x x ( mol )
\(2C_2H_2+5O_2\rightarrow\left(t^o\right)4CO_2+2H_2O\)
y 2y ( mol )
Ta có:
\(\left\{{}\begin{matrix}22,4x+22,4y=3,36\\x+2y=0,2\end{matrix}\right.\) \(\Leftrightarrow\left\{{}\begin{matrix}x=0,1\\y=0,05\end{matrix}\right.\)
\(\Rightarrow V_{CH_4}=22,4.0,1=2,24l\)
\(\Rightarrow V_{C_2H_2}=22,4.0,05=1,12l\)
\(\%V_{CH_4}=\dfrac{2,24}{3,36}.100=66,67\%\)
\(\%V_{C_2H_2}=100\%-66,67\%=33,33\%\)

Gọi x,y lân lượt là số mol của CH4 và C2H2 ( x,y>0 )
CH4 + 2O2 → CO2 + 2H2O (1)
x...........2x........x..........2x
2C2H2 + 5O2 → 4CO2 + 2H2O (2)
y..............2,5y........2y........y
Từ (1) (2) ta có hệ phương trình
\(\left\{{}\begin{matrix}44x+88y=13,2\\36x+18y=5,4\end{matrix}\right.\)
⇒ \(\left\{{}\begin{matrix}x=0,1\\y=0,1\end{matrix}\right.\)
⇒ mhổn hợp = ( 0,1.16) + ( 0,1.26 ) = 4,2 (g)
⇒ %CH4 = \(\dfrac{0,1.16.100\%}{4,2}\)\(\approx\) 38,1%
⇒ %C2H2 = \(\dfrac{0,1.26.100\%}{4,2}\)\(\approx\)61,9 %

PTHH: \(CH_4+2O_2\xrightarrow[]{t^o}CO_2+2H_2O\)
Ta có: \(n_{CO_2}=\dfrac{1,68}{22,4}=0,075\left(mol\right)=n_{CH_4}\)
Đặt \(\left\{{}\begin{matrix}n_{C_2H_4}=a\left(mol\right)\\n_{C_2H_2}=b\left(mol\right)\end{matrix}\right.\) \(\Rightarrow a+b=\dfrac{5,04}{22,4}-0,075=0,15\) (1)
PTHH: \(C_2H_4+Br_2\rightarrow C_2H_4Br_2\)
\(C_2H_2+2Br_2\rightarrow C_2H_2Br_4\)
Theo PTHH: \(28a+26b=4,1\) (2)
Từ (1) và (2) \(\Rightarrow\left\{{}\begin{matrix}a=n_{C_2H_4}=0,1\left(mol\right)\\b=n_{C_2H_2}=0,05\left(mol\right)\end{matrix}\right.\)
Mặt khác: \(n_{hh}=\dfrac{5,04}{22,4}=0,225\left(mol\right)\) \(\Rightarrow\left\{{}\begin{matrix}\%V_{CH_4}=\dfrac{0,075}{0,225}\cdot100\%\approx33,33\%\\\%V_{C_2H_4}=\dfrac{0,1}{0,225}\cdot100\%\approx44,44\%\\\%V_{C_2H_2}=22,23\%\end{matrix}\right.\)

CH4+2O2-to>CO2+2H2O
x-----------------------------2x
C2H2+\(\dfrac{5}{2}\)O2-to>2CO2+H2O
y-----------------------------------y
=>\(\left\{{}\begin{matrix}x+y=\dfrac{3,36}{22,4}\\2x+y=\dfrac{4,5}{18}\end{matrix}\right.\)
=>\(\left\{{}\begin{matrix}x=0,1\\y=0,05\end{matrix}\right.\)
=>%VCH4=\(\dfrac{0,1.22,4}{3,36}\).100=66,67%
=>%VC2H2=100-66,67%=33,33%
b)
C2H2+2Br2->C2H2Br4
0,05-----0,1 mol
=>m Br2=0,1.160=16g

Gọi số mol của CH4 và C2H2 lần lượt là a và b
CH4 + 2O2 ➝ CO2 + 2H2O
a 2a a
C2H2 + 2,5O2 ➝ 2CO2 + H2O
b 2,5b 2b
CO2 + Ca(OH)2 ➝ CaCO3 + H2O
a + 2b a + 2b
Số mol của hỗn hợp khí: a + b = 4,48/22,4 = 0,2 mol
Số mol kết tủa: a + 2b = 30/100 = 0,3 mol
➝ a = 0,1 mol, b = 0,1 mol
a) %VC2H2 = %VCH4 = 50%
b) Tổng số mol khí O2 đã dùng: a + 2,5b = 0,35 mol
➝ VO2 = 0,35.22,4 = 7,84 lít
➝ V kk = 5.VO2 = 39,2 lít

a) Gọi số mol CH4, C2H2 là a, b (mol)
=> \(a+b=\dfrac{4,48}{22,4}=0,2\) (1)
\(n_{CaCO_3}=\dfrac{30}{100}=0,3\left(mol\right)\)
PTHH: CH4 + 2O2 --to--> CO2 + 2H2O
a--->2a----------->a
2C2H2 + 5O2 --to--> 4CO2 + 2H2O
b------>2,5b-------->2b
Ca(OH)2 + CO2 --> CaCO3 + H2O
0,3<-----0,3
=> a + 2b = 0,3 (2)
(1)(2) => a = 0,1 (mol); b = 0,1 (mol)
=> \(\%V_{CH_4}=\%V_{C_2H_2}=\dfrac{0,1}{0,2}.100\%=50\%\)
b) nO2 = 2a + 2,5b = 0,45 (mol)
=> VO2 = 0,45.22,4 = 10,08 (l)
=> Vkk = 10,08.5 = 50,4 (l)

\(n_{CO_2}=\dfrac{8,96}{22,4}=0,4mol\)
\(m_{tăng}=m_{Br_2}=m_{C_2H_2}=2,6g\)
\(\Rightarrow n_{C_2H_2}=\dfrac{2,6}{26}=0,1mol\)
\(C_2H_2+2Br_2\rightarrow C_2H_2Br_4\)
0,1 0,1
\(CH_4+2O_2\underrightarrow{t^o}CO_2+2H_2O\)
\(C_2H_2+\dfrac{5}{2}O_2\underrightarrow{t^o}2CO_2+H_2O\)
0,1 0,25 0,2
\(\Rightarrow n_{CO_2\left(CH_4\right)}=0,4-0,2=0,2mol\)
\(\Rightarrow n_{CH_4}=0,2mol\Rightarrow n_{O_2}=0,4mol\)
a)\(\%V_{CH_4}=\dfrac{0,2}{0,4}\cdot100\%=50\%\)
\(\%V_{C_2H_2}=100\%-50\%=50\%\)
b)\(\Sigma n_{O_2}=0,4+0,25=0,65mol\)
\(\Rightarrow V_{O_2}=0,65\cdot22,4=14,56l\)
\(\Rightarrow V_{kk}=14,56\cdot5=72,8l\)
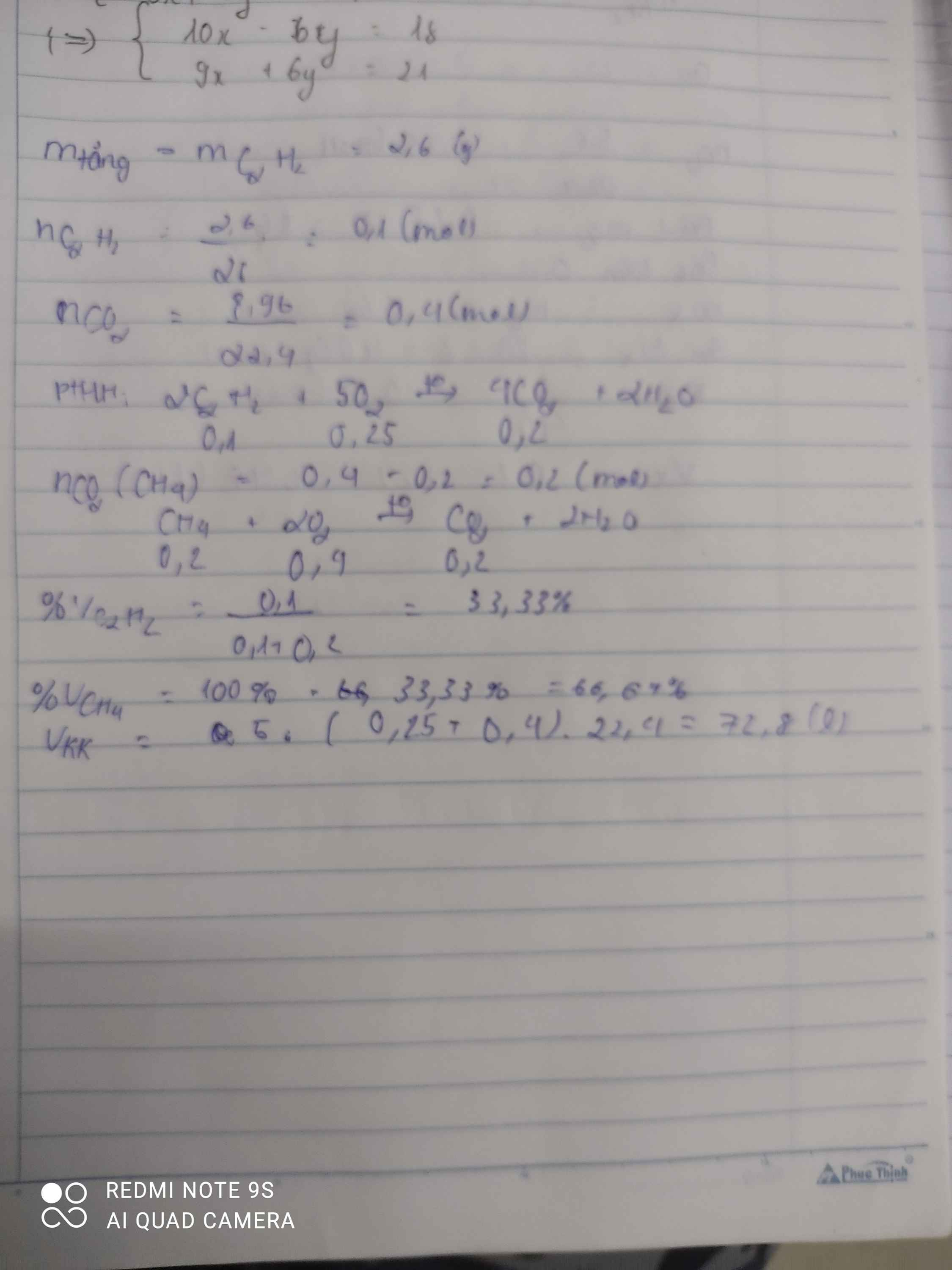
\(n_{C_2H_2}=a\left(mol\right),n_{CH_4}=b\left(mol\right)\)
\(\Rightarrow a+b=0.2\left(1\right)\)
\(n_{CO_2}=\dfrac{13.2}{44}=0.3\left(mol\right)\)
\(\Rightarrow2a+b=0.3\left(2\right)\)
\(\left(1\right),\left(2\right):a=b=0.1\)
\(m_{C_2H_2}=0.1\cdot26=2.6\left(g\right)\)
\(n_{C_2H_2} = a(mol) ; n_{CH_4} = b(mol)\\ \Rightarrow a + b = 0,2(1)\\ C_2H_2 + \dfrac{5}{2}O_2 \xrightarrow{t^o} 2CO_2 + H_2O\\ CH_4 + 2O_2 \xrightarrow{t^o} CO_2 + 2H_2O\\ n_{CO_2} = 2a + b = \dfrac{13,2}{44} = 0,3(2)\\ (1)(2)\Rightarrow a = 0,1 ; b = 0,2\\ m_{C_2H_2} = 0,1.26 = 2,6(gam)\)