Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.


\(n_{H_2}=\dfrac{3,36}{22,4}=0,15\left(mol\right)\)
PT: \(Fe+2HCl\rightarrow FeCl_2+H_2\)
\(n_{Fe}=n_{H_2}=0,15\left(mol\right)\Rightarrow m_{Fe}=0,15.56=8,4\left(g\right)\)
Đáp án: B

a, \(Fe+H_2SO_4\rightarrow FeSO_4+H_2\)
b, \(n_{H_2}=\dfrac{4,48}{22,4}=0,2\left(mol\right)\)
Theo PT: \(n_{FeSO_4}=n_{H_2SO_4}=n_{H_2}=0,2\left(mol\right)\)
\(\Rightarrow m_{FeSO_4}=0,2.152=30,4\left(g\right)\)
c, \(C_{M_{H_2SO_4}}=\dfrac{0,2}{0,5}=0,4\left(M\right)\)

\(a/3Fe+2O_2\xrightarrow[]{t^0}Fe_3O_4\\ b/n_{Fe}=\dfrac{1,4}{56}=0,025mol\\ n_{O_2}=\dfrac{0,025.2}{3}=\dfrac{0,05}{3}mol\\ V_{O_2}=\dfrac{0,05}{3}\cdot22,4\approx0,37l\\ c/C_1\\ n_{Fe_3O_4}=\dfrac{0,025}{3}mol\\ m_{Fe_3O_4}=\dfrac{0,025}{3}\cdot232\approx1,93g\\ C_2\\ m_{O_2}=\dfrac{0,05}{3}\cdot32\approx0,53g\\ BTKL:m_{Fe}+m_{O_2}=m_{Fe_3O_4}\\ \Rightarrow m_{Fe_3O_4}=1,4+0,53=1,93g\)


\(\left(1\right)CuCl_2+2NaOH\rightarrow Cu\left(OH\right)_2+NaCl\)
\(\left(2\right)Cu\left(OH\right)_2+H_2SO_4\rightarrow CuSO_4+2H_2O\)
\(\left(3\right)CuSO_4+2NaCl\rightarrow Na_2SO_4+CuCl_2\)

\(Mg+H_2SO_4\rightarrow MgSO_4+H_2\)
a) \(m_{Mg}=\dfrac{3,6}{24}=0,15\left(mol\right)\)
Theo PTHH: \(n_{H_2}=n_{Mg}=0,15\left(mol\right)\)
\(\Rightarrow V_{H_2}=0,15\cdot22,4=3,36\left(l\right)\)
b) Theo PTHH: \(n_{MgSO_4}=n_{Mg}=0,15\left(mol\right)\)
\(\Rightarrow m_{MgSO_4}=0,15\cdot120=18\left(g\right)\)
c) Theo PTHH: \(n_{HCl}=n_{Mg}=0,15\left(mol\right)\)
\(V_{dd,H_2SO_4}=\dfrac{0,15}{2,4}=0,0625\left(l\right)\)
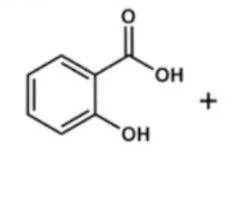
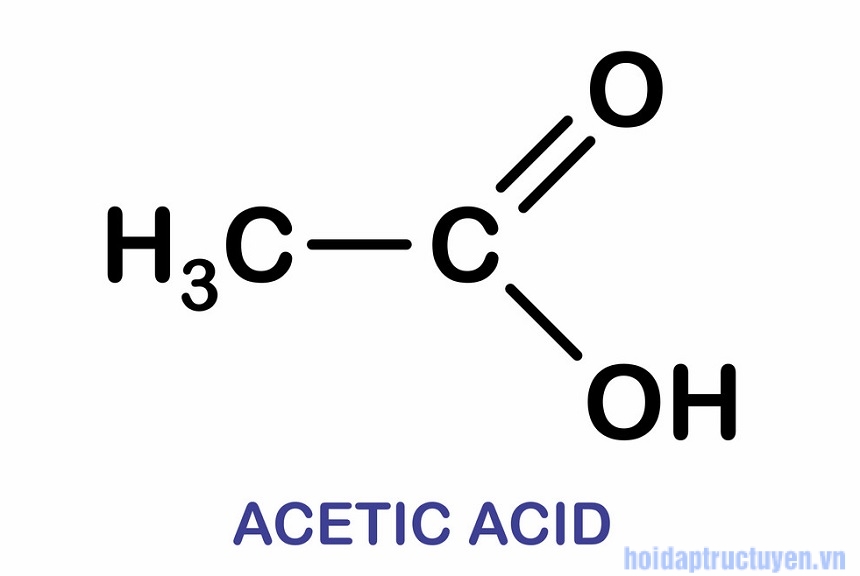
Đáp án: A
A