dùng 6,72 lít khí hidro ở đktc để khử lần lượt là HgO, PbO, FeO, Fe2O3,CuO,Fe3O4. a)Tính khối lượng kim loại được tạo ra. b)Tính khối lượng mỗi oxit cần dùng
Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.


\(n_{CuO}=a\left(mol\right),n_{Fe_2O_3}=b\left(mol\right)\)
\(m=80a+160b=6\left(g\right)\left(1\right)\)
\(n_{H_2}=\dfrac{2.24}{22.4}=0.1\left(mol\right)\)
\(CuO+H_2\underrightarrow{^{^{t^0}}}Cu+H_2O\)
\(Fe_2O_3+3H_2\underrightarrow{^{^{t^0}}}2Fe+3H_2O\)
\(n_{H_2}=a+3b=0.1\left(mol\right)\left(2\right)\)
\(\left(1\right),\left(2\right):a=0.025,b=0.025\)
\(m_{kl}=0.025\cdot64+0.025\cdot2\cdot56=4.4\left(g\right)\)
\(b.\)
\(m_{hh}=3m_{Fe_2O_3}=6\left(g\right)\)
\(\Rightarrow n_{Fe_2O_3}=\dfrac{2}{160}=0.0125\left(mol\right)\)
\(\Rightarrow n_{CuO}=0.0125\left(mol\right)\)
\(m_{kl}=0.0125\cdot2\cdot56+0.0125\cdot64=2.2\left(g\right)\)

Đáp án B
Giải chi tiết:
Quy phản ứng về dạng: [O]Oxit + CO → CO2
=> nCO = nO = 5,6: 22,4 = 0,25 mol
=> mKL = mOxit – mO = 30 – 0,25.16 = 26g

nCu = 8: 80=0,1(mol)
a) PTHH : CuO + H2 -t--> Cu +H2O
0,1-> 0,1------>0,1(mol)
mCu = 0,1.64=6,4(g)
VH2 = 0,1.22,4=2,24(l)

\(n_{Fe_2O_3}=\dfrac{14.4}{160}=0.09\left(mol\right)\)
\(Fe_2O_3+3H_2\underrightarrow{^{t^0}}2Fe+3H_2O\)
\(0.09.........0.27...0.18\)
\(V_{H_2}=0.27\cdot22.4=6.048\left(l\right)\)
\(m_{Fe}=0.18\cdot56=10.08\left(g\right)\)
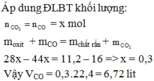

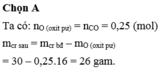
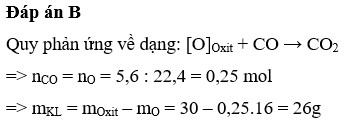
\(n_{H_2}=\dfrac{6,72}{22,4}=0,3\left(mol\right)\)
PTHH: \(HgO+H_2\underrightarrow{t^o}Hg+H_2O\) (1)
\(PbO+H_2\underrightarrow{t^o}PbO+H_2O\) (2)
\(FeO+H_2\underrightarrow{t^o}Fe+H_2O\) (3)
\(Fe_2O_3+3H_2\underrightarrow{t^o}2Fe+3H_2O\) (4)
\(CuO+H_2\underrightarrow{t^o}Cu+H_2O\) (5)
\(Fe_3O_4+4H_2\underrightarrow{t^o}3Fe+4H_2O\) (6)
a) \(n_{Hg\left(1\right)}=0,3\left(mol\right)\Rightarrow m_{Hg\left(1\right)}=0,3.201=60,3\left(g\right)\)
\(n_{Pb\left(2\right)}=0,3\left(mol\right)\Rightarrow m_{Pb\left(2\right)}=0,3.207=62,1\left(g\right)\)
\(n_{Fe\left(3\right)}=0,3\left(mol\right)\Rightarrow m_{Fe\left(3\right)}=0,3.56=16,8\left(g\right)\)
\(n_{Fe\left(4\right)}=0,2\left(mol\right)\Rightarrow m_{Fe\left(4\right)}=0,2.56=11,2\left(g\right)\)
\(n_{Cu\left(5\right)}=0,3\left(mol\right)\Rightarrow m_{Cu\left(5\right)}=0,3.64=19,2\left(g\right)\)
\(n_{Fe\left(6\right)}=0,225\left(mol\right)\Rightarrow m_{Fe\left(6\right)}=0,225.56=12,6\left(g\right)\)
b)
\(n_{HgO\left(1\right)}=0,3\left(mol\right)\Rightarrow m_{HgO\left(1\right)}=0,3.217=65,1\left(g\right)\)
\(n_{PbO\left(2\right)}=0,3\left(mol\right)\Rightarrow m_{PbO\left(2\right)}=0,3.223=66,9\left(g\right)\)
\(n_{FeO\left(3\right)}=0,3\left(mol\right)\Rightarrow m_{FeO\left(3\right)}=0,3.72=21,6\left(g\right)\)
\(n_{Fe_2O_3\left(4\right)}=0,1\left(mol\right)\Rightarrow m_{Fe_2O_3\left(4\right)}=0,1.160=16\left(g\right)\)
\(n_{CuO\left(5\right)}=0,3\left(mol\right)\Rightarrow m_{CuO\left(5\right)}=0,3.80=24\left(g\right)\)
\(n_{Fe_3O_4\left(6\right)}=0,075\left(mol\right)\Rightarrow m_{Fe_3O_4}=0,075.232=17,4\left(g\right)\)