Cho 24g hỗn hợp gồm MgO,Fe2O3 vào 500ml dung dịch axit clohidric 2M.Tính khối lượng mỗi axit
Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.


Pt : \(MgO+H_2SO_4\rightarrow MgSO_4+H_2O|\)
1 1 1 1
a 1a 0,05
\(Fe_2O_3+3H_2SO_4\rightarrow Fe_2\left(SO_4\right)_3+3H_2O|\)
1 3 1 3
b 3b 0,05
Gọi a là số mol của MgO
b là số mol của Fe2O3
\(m_{MgO}+m_{Fe2O3}=10\left(g\right)\)
⇒ \(n_{MgO}.M_{MgO}+n_{Fe2O3}.M_{Fe2O3}=10g\)
⇒ 40a + 160b = 10g (1)
\(m_{ct}=\dfrac{5,6.350}{100}=19,6\left(g\right)\)
\(n_{H2SO4}=\dfrac{19,6}{98}=0,2\left(mol\right)\)
⇒ 1a + 6b = 0,2(2)
Từ(1),(2) , ta có hệ phương trình :
40a + 160b = 10
1a + 6b = 0,2
⇒ \(\left\{{}\begin{matrix}a=0,05\\b=0,05\end{matrix}\right.\)
\(m_{MgO}=0,05.40=2\left(g\right)\)
\(m_{Fe2O3}=0,05.160=8\left(g\right)\)
0/0MgO = \(\dfrac{2.100}{10}=20\)0/0
0/0Fe2O3 = \(\dfrac{8.100}{10}=80\)0/0
b) Có : \(n_{MgO}=0,05\left(mol\right)\Rightarrow n_{MgSO4}=0,05\left(mol\right)\)
\(n_{Fe2O3}=0,05\left(mol\right)\Rightarrow n_{Fe2\left(SO4\right)3}=0,05\left(mol\right)\)
\(m_{MgSO4}=0,05.161=8,05\left(g\right)\)
\(m_{Fe2\left(SO4\right)3}=0,05.400=20\left(g\right)\)
Chúc bạn học tốt

Ta có: \(n_{H_2}=\dfrac{11,2}{22,4}=0,5\left(mol\right)\)
a. PTHH:
Zn + H2SO4 ---> ZnSO4 + H2 (1)
MgO + H2SO4 ---> MgSO4 + H2O (2)
b. Theo PT(1): \(n_{Zn}=n_{H_2}=0,5\left(mol\right)\)
=> \(m_{Zn}=0,5.65=32,5\left(g\right)\)
(Sai đề nhé.)
\(n_{H_2}=\dfrac{11,2}{22,4}=0,5mol\)
\(MgO+H_2SO_4\rightarrow MgSO_4+H_2O\)
\(Zn+H_2SO_4\rightarrow ZnSO_4+H_2\uparrow\)
0,5 0,5
b)\(m_{Zn}=0,5\cdot65=32,5\left(g\right)\)
\(m_{ZnO}=\) ko tính đc do lỗi đề

\(n_{MgO}=a\left(mol\right),n_{Fe_2O_3}=b\left(mol\right)\)
\(m=40a+160b=12\left(g\right)\left(1\right)\)
\(MgO+H_2SO_4\rightarrow MgSO_4+H_2O\)
\(Fe_2O_3+3H_2SO_4\rightarrow Fe_2\left(SO_4\right)_3+3H_2O\)
\(m_{Muối}=120a+400y=32\left(g\right)\left(2\right)\)
\(\left(1\right),\left(2\right):a=0.1,b=0.05\)
\(\%MgO=\dfrac{0.1\cdot40}{12}\cdot100\%=33.33\%\)
\(\%Fe_2O_3=66.67\%\)
\(m_{dd}=12+200=212\left(g\right)\)
\(C\%_{MgSO_4}=\dfrac{0.1\cdot120}{212}\cdot100\%=5.66\%\)
\(C\%_{Fe_2\left(SO_4\right)_3}=\dfrac{0.05\cdot400}{212}\cdot100\%=9.42\%\)

a) \(n_{H_2SO_4}=0,2.2=0,4\left(mol\right)\)
\(Fe_2O_3+3H_2SO_4\rightarrow Fe_2\left(SO_4\right)_3+3H_2O\)
\(CuO+H_2SO_4\rightarrow CuSO_4+H_2O\)
Đặt \(n_{Fe_2O_3}=a\left(mol\right);n_{CuO}=b\left(mol\right)\)
Ta có:
\(\left\{{}\begin{matrix}160a+80b=24\\3a+b=0,4\end{matrix}\right.\\ \Leftrightarrow\left\{{}\begin{matrix}a=0,1\\b=0,1\end{matrix}\right.\)
\(\%m_{Fe_2O_3}=\dfrac{160.0,1}{24}.100\%=66,67\%\\ \%m_{CuO}=100\%-66,67\%=33,33\%\)
b) \(Fe_2O_3+6HCl\rightarrow2FeCl_3+3H_2O\\ CuO+2HCl\rightarrow CuCl_2+H_2O\)
\(n_{HCl}=6.0,1+2.0,1=0,9\left(mol\right)\)
\(m_{HCl}=0,9.36,5=32,85\left(g\right)\)
\(m_{ddHCl}=\dfrac{32,85.100}{14,7}=223,47\left(g\right)\)

\(Fe_2O_3 + 6HCl \to 2FeCl_3 + 3H_2O\\ ZnO + 2HCl \to ZnCl_2 + H_2O\\ MgO + 2HCl \to MgCl_2 + H_2O\\ n_{HCl} = 0,5.0,1 = 0,05(mol)\\ n_{H_2O} = \dfrac{1}{2}n_{HCl} = 0,025(mol)\\ \text{Bảo toàn khối lượng : }\\ m_{muối} = m_{hỗn\ hợp} + m_{HCl} - m_{H_2O} = 1,405 + 0,05.36,5 - 0,025.18 = 2,78(gam)\)

\(a)n_{H_2}=\dfrac{5,6}{22,4}=0,25mol\\ 2Al+6HCl\rightarrow2AlCl_3+3H_2\)
\(\dfrac{1}{6}\) \(0,5\) \(\dfrac{1}{6}\) \(0,25\)
\(\%m_{Al}=\dfrac{1:6.27}{25}\cdot100=18\%\\ \%m_{Al_2O_3}=100-18=82\%\\ b)n_{Al_2O_3}=\dfrac{25-1:6.27}{102}=\dfrac{41}{204}mol\\ Al_2O_3+6HCl\rightarrow2AlCl_3+3H_2O\)
\(\dfrac{41}{204}\) \(\dfrac{41}{36}\) \(\dfrac{41}{102}\)
\(m_{ddHCl}=\dfrac{\left(0,5+41:36\right)36,5}{20}\cdot100=299,1g\\ V_{ddHCl}=\dfrac{299,1}{1,1}=271,9ml\)
\(m_{dd}=299,1+25-0,25.2=323,6g\)
\(m_{AlCl_3}=\left(\dfrac{1}{6}+\dfrac{41}{102}\right)\cdot133,5=75,9g\\ C_{\%AlCl_3}=\dfrac{75,9}{323,6}\cdot100=23,45\%\)
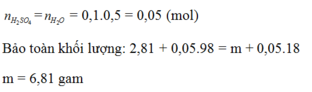
\(n_{MgO}=a\left(mol\right),n_{Fe_2O_3}=b\left(mol\right)\)
\(m_{hh}=40a+160b=24\left(g\right)\left(1\right)\)
\(MgO+2HCl\rightarrow MgCl_2+H_2O\)
\(Fe_2O_3+6HCl\rightarrow2FeCl_3+3H_2O\)
\(n_{HCl}=2a+6b=1\left(mol\right)\left(2\right)\)
\(\left(1\right),\left(2\right):a=0.2,b=0.1\)
\(m_{MgO}=0.2\cdot40=8\left(g\right)\)
\(m_{Fe_2O_3}=16\left(g\right)\)