Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

\(n_{H_2}=\dfrac{4}{2}=2\left(mol\right)\)
PT: \(Fe_3O_4+4H_2\underrightarrow{^{t^o}}3Fe+4H_2O\)
Theo PT: \(n_{Fe}=\dfrac{3}{4}n_{H_2}=1,5\left(mol\right)\Rightarrow m_{Fe}=1,5.56=84\left(g\right)\)

\(n_{Fe_2O_3}=\dfrac{14.4}{160}=0.09\left(mol\right)\)
\(Fe_2O_3+3H_2\underrightarrow{^{t^0}}2Fe+3H_2O\)
\(0.09.........0.27...0.18\)
\(V_{H_2}=0.27\cdot22.4=6.048\left(l\right)\)
\(m_{Fe}=0.18\cdot56=10.08\left(g\right)\)

a, \(FeO+H_2\underrightarrow{t^o}Fe+H_2O\)
b, \(n_{FeO}=\dfrac{7,2}{72}=0,1\left(mol\right)\)
\(n_{Fe}=n_{FeO}=0,1\left(mol\right)\Rightarrow m_{Fe}=0,1.56=5,6\left(g\right)\)
c, \(n_{H_2}=n_{FeO}=0,1\left(mol\right)\Rightarrow V_{H_2}=0,1.22,4=2,24\left(l\right)\)

\(PTHH:Fe_3O_4+4H_2\rightarrow^{t^o}3Fe+4H_2O\\ n_{Fe}=\dfrac{30,24}{56}=0,54\left(mol\right)\\ \Rightarrow n_{Fe_3O_4}=\dfrac{1}{3}n_{Fe}=0,18\left(mol\right)\\ \Rightarrow m_{Fe_3O_4}=0,18\cdot232=41,76\left(g\right)\)

\(n_{FeO}=\dfrac{3.2}{72}=\dfrac{2}{45}\left(mol\right)\)
\(FeO+H_2\underrightarrow{^{^{t^0}}}Fe+H_2O\)
\(\dfrac{2}{45}....\dfrac{2}{45}....\dfrac{2}{45}\)
\(V_{H_2}=\dfrac{2}{45}\cdot22.4=1\left(l\right)\)
\(Fe+\dfrac{3}{2}Cl_2\underrightarrow{^{t^0}}FeCl_3\)
\(\dfrac{2}{45}.............\dfrac{2}{45}\)
\(m_{FeCl_3}=\dfrac{2}{45}\cdot162.5=7.22\left(g\right)\)
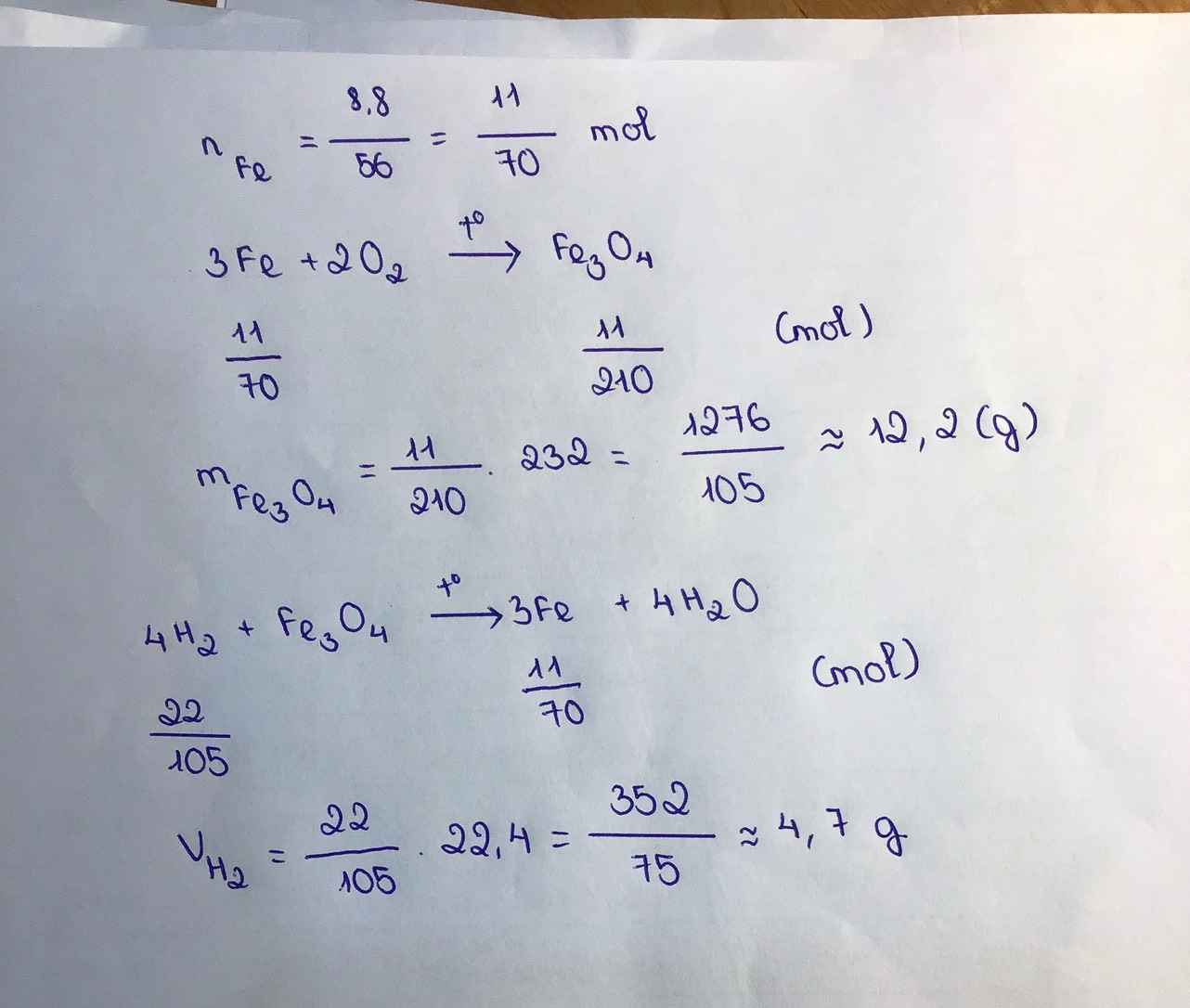

Theo bài ra ta có :
8 gam H\(_2\) tạo ra 168 gam Fe
Vậy:4 gam H\(_2\) tạo ra 84 gam Fe