Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

- \(nCH_2=CH_2\underrightarrow{t^o,p,xt}\left(-CH_2-CH_2-\right)_n\)
- \(CH_4+Cl_2\underrightarrow{as}CH_3Cl+HCl\)
- \(CH_2=CH_2+Br_2\rightarrow CH_2Br-CH_2Br\)
- \(CH\equiv CH+2Br_2\rightarrow CHBr_2-CHBr_2\)
- \(C_6H_{12}O_6+Ag_2O\underrightarrow{NH_3}C_6H_{12}O_7+2Ag\)

a) \(CH_4+Cl_2\underrightarrow{as}CH_3Cl+HCl\) (pư thế)
b) \(C_2H_4+Br_2\rightarrow C_2H_4Br_2\) (pư cộng)

Em coi xem là metan và axetilen hay metan và etilen nha?
Hoặc xem tăng 2,8gam hay 2,6 gam?
Check kĩ đề giúp anh nha!

\(n_{hh}=\dfrac{3,36}{22,4}=0,15mol\)
\(n_{Br_2}=\dfrac{2,4}{160}=0,015mol\)
\(C_2H_2+2Br_2\rightarrow C_2H_2Br_4\)
0,0075 0,015 ( mol )
\(V_{C_2H_2}=0,0075.22,4=0,168l\)
\(V_{CH_4}=3,36-0,168=3,192l\)
\(\%V_{C_2H_2}=\dfrac{0,168}{3,36}.100=5\%\)
\(\%V_{CH_4}=100\%-5\%=95\%\)

a)
C2H4 + Br2 --> C2H4Br2
C2H2 + 2Br2 --> C2H2Br4
b) Gọi số mol C2H4, C2H2 là a, b (mol)
=> \(a+b=\dfrac{11,2}{22,4}=0,5\) (1)
PTHH: C2H4 + Br2 --> C2H4Br2
a---->a
C2H2 + 2Br2 --> C2H2Br4
b---->2b
=> a + 2b = \(\dfrac{112}{160}=0,7\) (2)
(1)(2) => a = 0,3 (mol); b = 0,2 (mol)
\(\left\{{}\begin{matrix}\%V_{C_2H_4}=\dfrac{0,3}{0,5}.100\%=60\%\\\%V_{C_2H_2}=\dfrac{0,2}{0,5}.100\%=40\%\end{matrix}\right.\)
\(\left\{{}\begin{matrix}\%m_{C_2H_4}=\dfrac{0,3.28}{0,3.28+0,2.26}.100\%=61,765\%\\\%m_{C_2H_2}=\dfrac{0,2.26}{0,3.28+0,2.26}.100\%=38,235\%\end{matrix}\right.\)

a.b.
\(n_{H_2O}=\dfrac{8,64}{18}=0,48mol\)
\(n_{hh}=\dfrac{7,437}{24,79}=0,3mol\)
Gọi \(\left\{{}\begin{matrix}n_{C_2H_2}=x\\n_{C_2H_4}=y\end{matrix}\right.\)
\(2C_2H_2+5O_2\rightarrow\left(t^o\right)4CO_2+2H_2O\)
x x ( mol )
\(C_2H_4+3O_2\rightarrow\left(t^o\right)2CO_2+2H_2O\)
y 2y ( mol )
Ta có:
\(\left\{{}\begin{matrix}x+y=0,3\\x+2y=0,48\end{matrix}\right.\) \(\Leftrightarrow\left\{{}\begin{matrix}x=0,12\\y=0,18\end{matrix}\right.\)
\(\rightarrow\left\{{}\begin{matrix}\%V_{C_2H_2}=\dfrac{0,12}{0,3}.100=40\%\\\%V_{C_2H_4}=100\%-40\%=60\%\end{matrix}\right.\)
c.
\(C_2H_2+2Br_2\rightarrow C_2H_2Br_4\)
0,12 0,24 ( mol )
\(C_2H_4+Br_2\rightarrow C_2H_4Br_2\)
0,18 0,18 ( mol )
\(C_{M_{Br_2}}=\dfrac{0,24+0,18}{0,2}=2,1M\)
\(n_{hh}=\dfrac{7,437}{24,79}=0,3\left(mol\right)\\ n_{H_2O}=\dfrac{8,64}{18}=0,48\left(mol\right)\)
Gọi \(\left\{{}\begin{matrix}n_{C_2H_2}=a\left(mol\right)\\n_{C_2H_4}=b\left(mol\right)\end{matrix}\right.\)
PTHH:
C2H4 + 3O2 --to--> 2CO2 + 2H2O
b--------------------------------->2b
2C2H2 + 5O2 --to--> 4CO2 + 2H2O
a------>2,5a
=> \(\left\{{}\begin{matrix}a+b=0,3\\2b+2,5a=0,48\end{matrix}\right.\) => nghiệm âm

\(CH_4+Cl_2\rightarrow\left(as\right)CH_3Cl+HCl\\ C_2H_4+Br_2\rightarrow C_2H_4Br_2\\ C_2H_2+2Br_{2\left(dư\right)}\rightarrow C_2H_2Br_4\\ nCH_2=CH_2\underrightarrow{^{to,P,xt}}\left(-CH_2-CH_2-\right)_n\\ CH_4+2O_2\rightarrow\left(t^o\right)CO_2+2H_2O\\ C_2H_4+3O_2\rightarrow\left(t^o\right)2CO_2+2H_2O\\ 2C_2H_2+5O_2\rightarrow\left(t^o\right)4CO_2+2H_2O\\ C_aH_{2a+2}+\dfrac{3a+1}{2}O_2\rightarrow\left(t^o\right)aCO_2+\left(a+1\right)H_2O\)

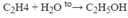
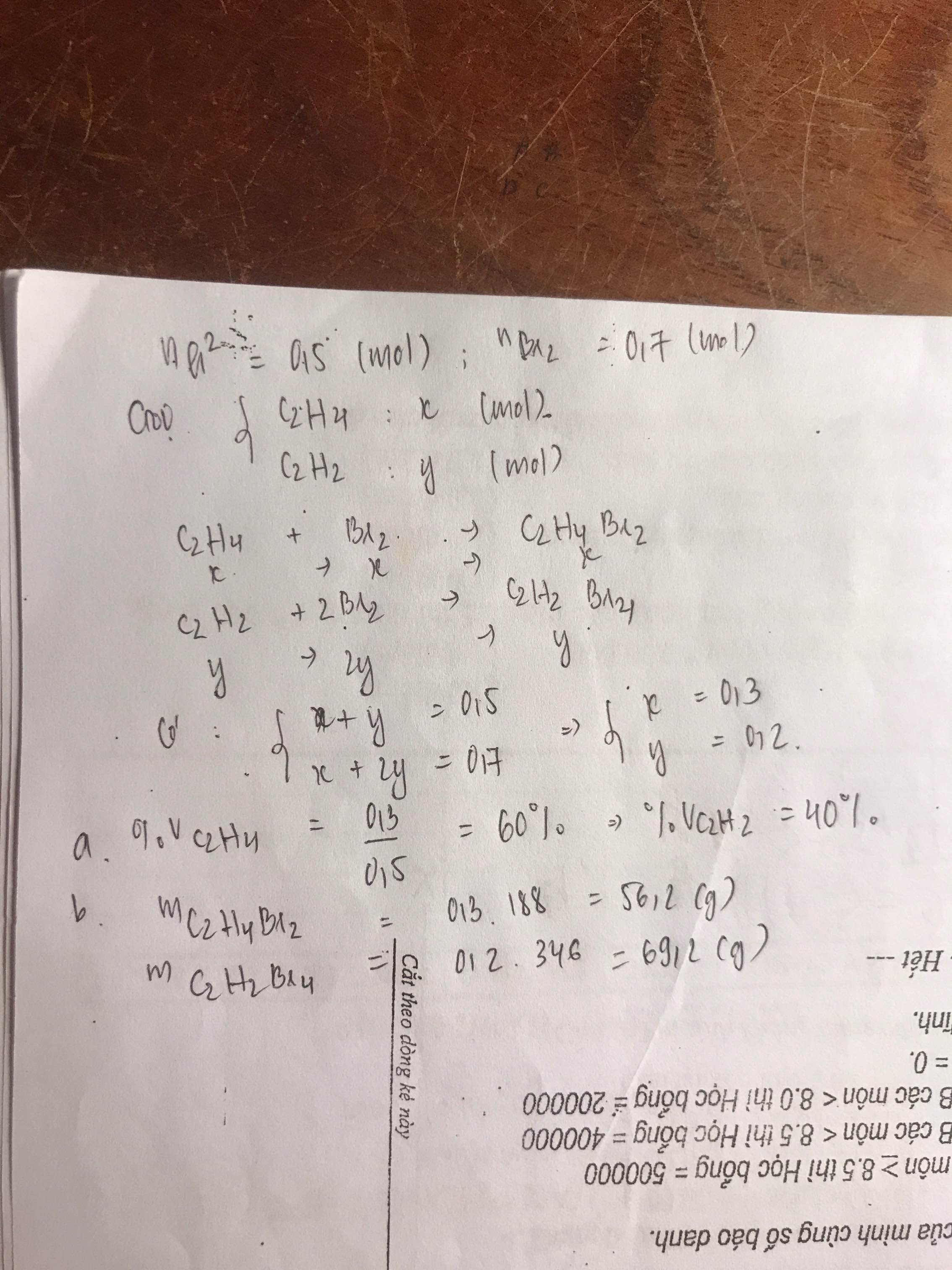
Cái chỗ \(\rightarrow\)chẳng nhìn thấy chữ ở trên , nhỏ quá