Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

\(a.C_2H_5OH+3O_2-^{t^o}\rightarrow2CO_2+3H_2O\\ n_{C_2H_5OH}=0,3\left(mol\right)\\ n_{CO_2}=2n_{C_2H_5OH}=0,6\left(mol\right)\\ \Rightarrow V_{CO_2}=0,6.22,4=13,44\left(l\right)\\ b.n_{O_2}=3n_{C_2H_5OH}=0,6\left(mol\right)\\ MàV_{O_2}=\dfrac{1}{5}V_{kk}\\ \Rightarrow V_{kk}=V_{O_2}.5=0,6.22,4.5=67,2\left(l\right)\\ c.n_{NaOH}=0,9\left(mol\right)\\ Tacó:\dfrac{n_{NaOH}}{n_{CO_2}}=\dfrac{0,9}{0,6}=1,5\\ \Rightarrow Tạora2muốiNaHCO_3vàNa_2CO_3\\ Đặt:n_{NaHCO_3}=x\left(mol\right);n_{Na_2CO_3}=y\left(mol\right)\\ \Rightarrow\left\{{}\begin{matrix}x+y=0,6\left(BTnguyento\left(C\right)\right)\\x+2y=0,9\left(BTnguyento\left(Na\right)\right)\end{matrix}\right.\\ \Rightarrow\left\{{}\begin{matrix}x=0,3\\y=0,3\end{matrix}\right.\\ \Rightarrow m_{muối}=0,3.84+0,3.106=57\left(g\right)\)

a, \(n_{C_2H_6O}=\dfrac{92}{46}=2\left(mol\right)\)
PT: \(C_2H_6O+3O_2\underrightarrow{t^o}2CO_2+3H_2O\)
Theo PT: \(n_{O_2}=3n_{C_2H_6O}=6\left(mol\right)\Rightarrow V_{O_2}=6.22,4=134,4\left(l\right)\)
b, \(V_{kk}=5V_{O_2}=672\left(l\right)\)

\(n_{C_2H_5OH}=\dfrac{23}{46}=0,5\left(mol\right)\\a, PTHH:C_2H_5OH+3O_2\rightarrow\left(t^o\right)2CO_2+3H_2O\\ b,n_{O_2}=3.0,5=1,5\left(mol\right)\\ V_{O_2\left(đktc\right)}=22,4.1,5=33,6\left(l\right)\\ c,V_{C_2H_5OH}=46\%.100=46\left(ml\right)\\ C_2H_5OH+Na\rightarrow C_2H_5ONa+\dfrac{1}{2}H_2\\ n_{C_2H_5OH}=\dfrac{0,8.46}{46}=0,8\left(mol\right)\\ n_{H_2}=\dfrac{0,8}{2}=0,4\left(mol\right)\Rightarrow V_{H_2\left(đktc\right)}=0,4.22,4=8,96\left(l\right)\)

ta có pthh :C2H5OH +3O2 ---------->2CO2 +3H2O
Tỳ lệ :0,2 mol-----0,6 mol-----0,4 mol (hiểu tại sao hông )
VCO2 :0,4 X 22,4 =(MÁY TÍNH ?)
nCO2=0,6mol -------->n KK =0,6 X 100/20 =3 MOL
Vkk =3 x 22,4 =67,2 l

a) C2H5OH + 3O2 --to--> 2CO2 + 3H2O
b) \(n_{C_2H_5OH}=\dfrac{46}{46}=1\left(mol\right)\)
PTHH: C2H5OH + 3O2 --to--> 2CO2 + 3H2O
1----->3----------->2------->3
=> VO2 = 22,4.3 = 67,2 (l)
c) mH2O = 3.18 = 54 (g)
d) VCO2 = 2.22,4 = 44,8 (l)

PTHH : \(C_2H_4\rightarrow2H_2O+2CO_2\)
\(n_{C_2H_4}=\frac{14}{28}=0,5\left(mol\right)\)
Theo phương trình :
\(n_{O_2}=3n_{C_2H_4}=3.0,5=1,5\left(mol\right)\)
\(\Rightarrow V_{O_2}=1,5.22,4=33,6\left(l\right)\)
\(n_{CO_2}=2n_{C_2H_4}=2.0,5=1\left(mol\right)\)
\(\Rightarrow m_{CO_2}=1.44=44\left(gam\right)\)
PTHH: C2H4 (0,5 mol) + 3O2 (1,5 mol) to→ 2CO2 (1 mol) + 2H2O.
Thể tích khí oxi cần dùng ở đktc: Vkhí oxi=1,5x22,4=33,6 (lít).
Khối lượng khí CO2 sinh ra là: mkhí cacbonic=1x44=44 (gam).
Chọn A.
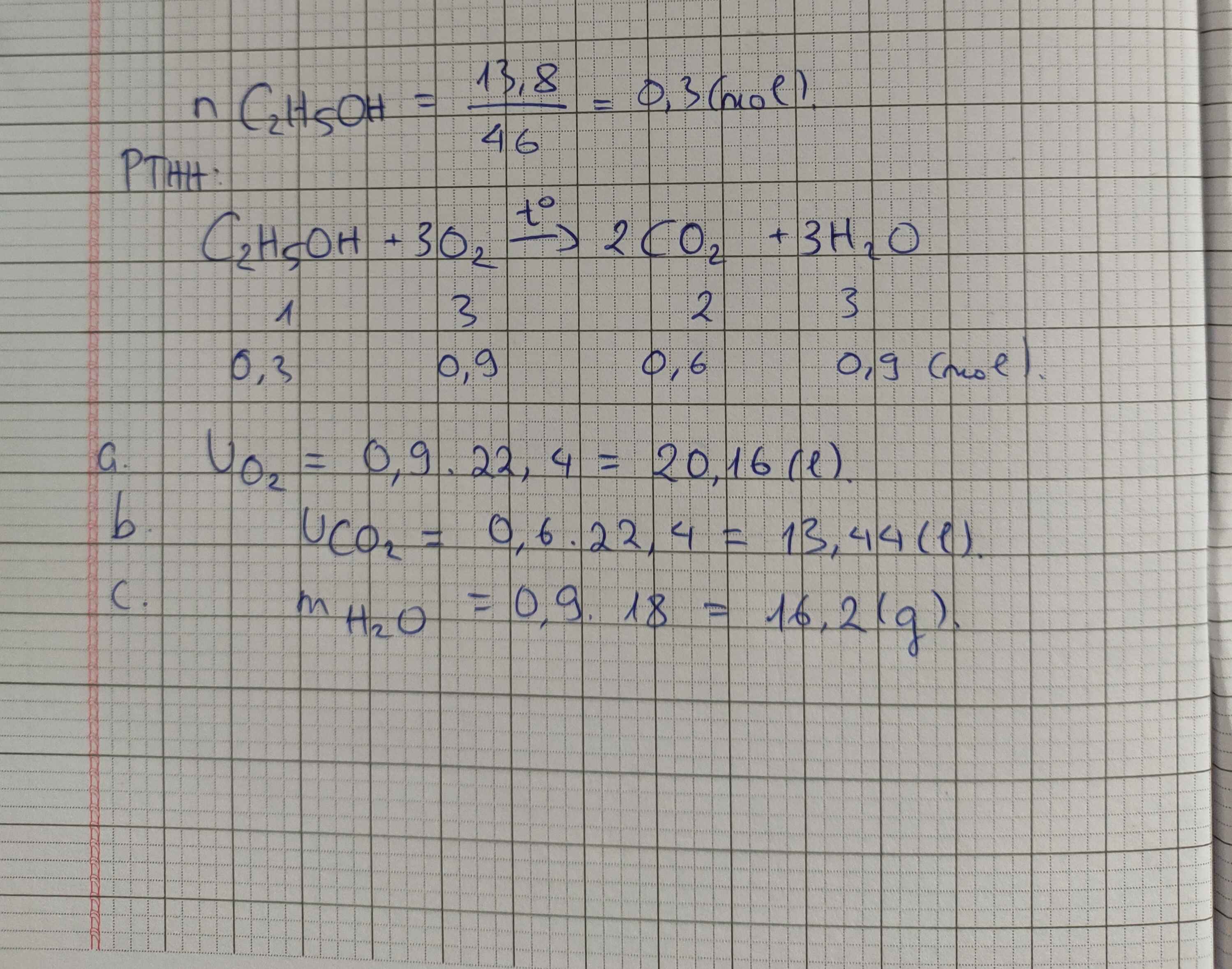
Đáp án: D
n C 2 H 5 O H = 13 , 8 46 = 0 , 3 m o l
C 2 H 5 O H + 3 O 2 → t 0 2 C O 2 + 3 H 2 O
0,3 mol → 0,9 mol
⇒ v o 2 = 0 , 9 . 22 , 4 = 20 , 16