Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

a.b.
\(n_{Fe}=\dfrac{2,8}{56}=0,05mol\)
\(Fe+2HCl\rightarrow FeCl_2+H_2\)
0,05 0,1 0,05 ( mol )
\(V_{H_2}=0,05.22,4=1,12l\)
\(m_{HCl}=0,1.36,5=3,65g\)
c.
\(CuO+H_2\rightarrow\left(t^o\right)Cu+H_2O\)
0,05 0,05 ( mol )
\(m_{Cu}=0,05.64=3,2g\)

a, \(n_{Zn}=\dfrac{13}{65}=0,2\left(mol\right)\)
PT: \(Zn+2HCl\rightarrow ZnCl_2+H_2\)
Theo PT: \(n_{H_2}=n_{Zn}=0,2\left(mol\right)\Rightarrow V_{H_2}=0,2.22,4=4,48\left(l\right)\)
b, \(n_{HCl}=2n_{Zn}=0,4\left(mol\right)\Rightarrow m_{HCl}=0,4.36,5=14,6\left(g\right)\)
c, \(n_{Fe_2O_3}=\dfrac{16}{160}=0,1\left(mol\right)\)
PT: \(Fe_2O_3+3H_2\underrightarrow{t^o}2Fe+3H_2O\)
Xét tỉ lệ: \(\dfrac{0,1}{1}>\dfrac{0,2}{3}\), ta được Fe2O3 dư.
Theo PT: \(n_{Fe}=\dfrac{2}{3}n_{H_2}=\dfrac{2}{15}\left(mol\right)\Rightarrow m_{Fe}=\dfrac{2}{15}.56=\dfrac{112}{15}\left(g\right)\)

nHCl=18,25/36,5=0,5mol
pt : Mg + 2HCl ------> MgCl2 +H2
npứ: 0,25<-0,5---------->0,25----->0,25
VH2=0,25.22,4=5,6l
mMgCl2 = 0,25.95=23,25g
pt: H2 + CuO --to--> Cu + H2O
npứ;0,25-------------->0,25
mCu=0,25.64=16g
1) \(n_{HCl}=\dfrac{m}{M}=\dfrac{18,25}{36,5}=0,5\left(mol\right)\)
Mg + 2HCl → MgCl2+ H2
1mol 2mol 1mol 1mol
0,25mol 0,5mol 0,25mol 0,25mol
2) \(v_{H_2}=n.22,4=0,25.22,4=5,6\left(l\right)\)
3) \(m_{MgCl_2}=n.M=0,25.95=23,75\left(g\right)\)
4) PT2:
CuO + H2 \(\underrightarrow{t^o}\) Cu + H2O
1mol 1mol 1mol 1mol
0,25mol 0,25mol 0,25mol 0,25mol
mCu= n.M= 0,25.64=16(g)

a, \(Zn+H_2SO_4\rightarrow ZnSO_4+H_2\)
b, \(n_{Zn}=\dfrac{13}{65}=0,2\left(mol\right)\)
Theo PT: \(n_{H_2}=n_{Zn}=0,2\left(mol\right)\Rightarrow V_{H_2}=0,2.22,4=4,48\left(l\right)\)
c, \(n_{Fe_3O_4}=\dfrac{18,56}{232}=0,08\left(mol\right)\)
PT: \(Fe_3O_4+4H_2\underrightarrow{t^o}3Fe+4H_2O\)
Xét tỉ lệ: \(\dfrac{0,08}{1}>\dfrac{0,2}{4}\), ta được Fe3O4 dư.
Theo PT: \(n_{Fe}=\dfrac{3}{4}n_{H_2}=0,15\left(mol\right)\Rightarrow m_{Fe}=0,15.56=8,4\left(g\right)\)

\(n_{Zn}=\dfrac{6,5}{65}=0,1\left(mol\right)\\
pthh:Zn+2HCl\rightarrow ZnCl_2+H_2\)
0,1 0,2 0,1
\(V_{H_2}=0,1.22,4=2,24l\\
m_{HCl}=\left(0,2.36,5\right).10\%=0,73g\)
\(n_{Fe_2O_3}=\dfrac{16}{160}=0,1\left(mol\right)\\
pthh:Fe_2O_3+3H_2\underrightarrow{t^o}2Fe+3H_2O\\
LTL:\dfrac{0,1}{1}>\dfrac{0,1}{3}\)
=> Fe2O3 dư
\(n_{Fe}=\dfrac{2}{3}n_{H_2}=0,067\left(mol\right)\\
m_{Fe}=0,067.56=3,73g\)
a.b.\(n_{Zn}=\dfrac{6,5}{65}=0,1mol\)
\(Zn+2HCl\rightarrow ZnCl_2+H_2\)
0,1 0,2 0,1 ( mol )
\(V_{H_2}=0,1.22,4=2,24l\)
\(m_{ddHCl}=\dfrac{0,2.36,5}{10\%}=73g\)
c.\(n_{Fe_2O_3}=\dfrac{16}{160}=0,1mol\)
\(Fe_2O_3+3H_2\rightarrow\left(t^o\right)2Fe+3H_2O\)
0,1 > 0,1 ( mol )
0,1 1/15 ( mol )
\(m_{Fe}=\dfrac{1}{15}.56=3,73g\)

a, \(Zn+2HCl\rightarrow ZnCl_2+H_2\)
b, \(n_{Zn}=\dfrac{6,5}{65}=0,1\left(mol\right)\)
Theo PT: \(n_{H_2}=n_{Zn}=0,1\left(mol\right)\Rightarrow V_{H_2}=0,1.22,4=2,24\left(l\right)\)
c, \(n_{CuO}=\dfrac{12}{80}=0,15\left(mol\right)\)
PT: \(CuO+H_2\underrightarrow{t^o}Cu+H_2O\)
Xét tỉ lệ: \(\dfrac{0,15}{1}>\dfrac{0,1}{1}\), ta được CuO dư.
Theo PT: \(n_{Cu}=n_{H_2}=0,1\left(mol\right)\Rightarrow m_{Cu}=0,1.64=6,4\left(g\right)\)

a) \(n_{Zn}=\dfrac{16,25}{65}=0,25\left(mol\right)\)
PTHH: Zn + 2HCl --> ZnCl2 + H2
0,25-------------------->0,25
=> VH2 = 0,25.22,4 = 5,6 (l)
b)
PTHH: CuO + H2 --to--> Cu + H2O
0,25--->0,25
=> mCu = 0,25.64 = 16 (g)

\(1,\\ Mg+2HCl\rightarrow MgCl_2+H_2\\ 2,\\ n_{HCl}=\dfrac{18,25}{36,5}=0,5\left(mol\right)\Rightarrow n_{Mg}=n_{MgCl_2}=n_{H_2}=\dfrac{0,5}{2}=0,25\left(mol\right)\\ V_{H_2\left(đktc\right)}=0,25.22,4=5,6\left(l\right)\\ 3,\\ m_{MgCl_2}=95.0,25=23,75\left(g\right)\\ 4,\\ H_2+CuO\rightarrow\left(t^o\right)Cu+H_2O\\ n_{Cu}=n_{H_2}=0,25\left(g\right)\\ m_{Cu}=0,25.64=16\left(g\right)\)
1. \(Mg+2HCl\rightarrow MgCl_2+H_2\)
2. \(n_{HCl}=\dfrac{m_{HCl}}{M_{HCl}}=\dfrac{18,25}{36,5}\approx0,5\left(mol\right)\)
Theo PTHH: \(n_{H_2}=\dfrac{1}{2}n_{HCl}\)
\(\Rightarrow n_{H_2}=\dfrac{1}{2}.0,5=0,25\left(mol\right)\)
\(\Rightarrow V_{H_2}=n_{H_2}.22,4=0,25.22,4=5,6\left(l\right)\)
3. Theo PTHH: \(n_{MgCl_2}=\dfrac{1}{2}n_{HCl}\)
\(\Rightarrow n_{MgCl_2}=0,25\left(mol\right)\)
\(m_{MgCl_2}=n_{MgCl_2}.M_{MgCl_2}=0,25.95=23,75\left(g\right)\)
4. \(H_2+CuO\rightarrow Cu+H_2O\)
Theo PTHH: \(n_{Cu}=n_{H_2}=0,25\left(mol\right)\)
\(\Rightarrow m_{Cu}=n_{Cu}.M_{Cu}=0,25.64=16\left(g\right)\)
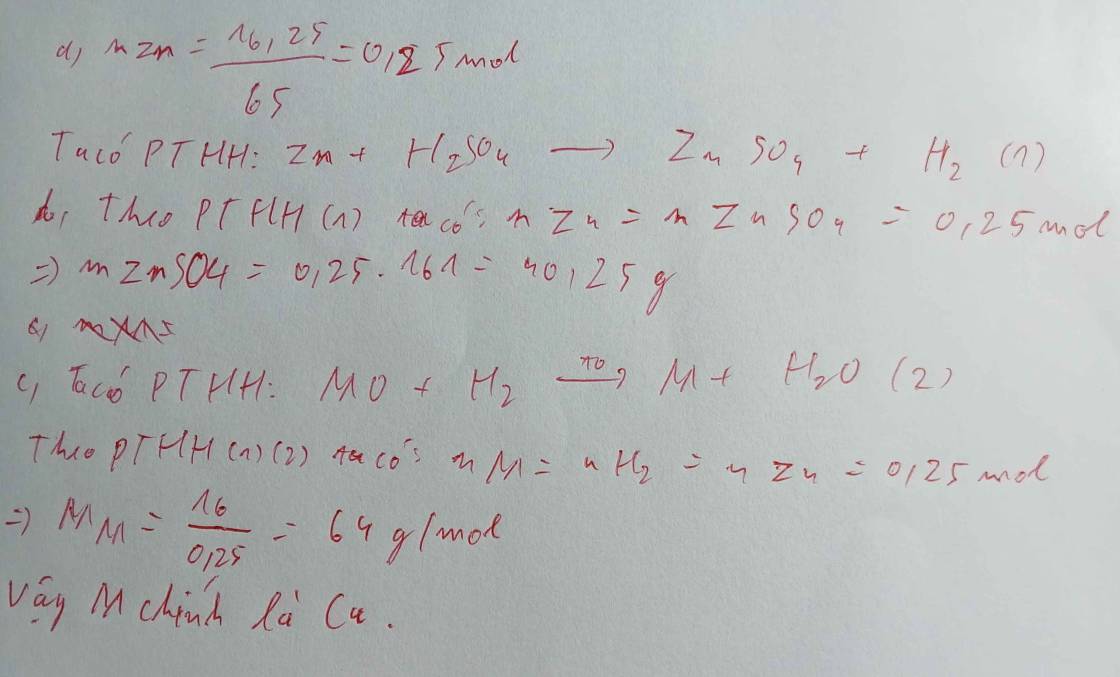
1) Zn + HCl -----> ZnCl2 + H2↑
Theo ĐLBTKL , ta có :
mZnCl2 + mH2 = 16,25 + 7,3 = 23,55 (g)
Câu 2 chả hiểu j cả...
Câu 2 chắc bị nhầm. :))