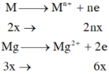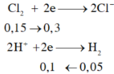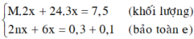Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

\(n_{KMnO_4}=\frac{15,8}{158}=0,1\left(mol\right)\)
PTHH : \(2KMnO_4+16HCl-->2KCl+2MnCl_2+5Cl_2+8H_2O\) (1)
\(Cl_2+H_2-as->2HCl\) (2)
Có : \(m_{ddHCl}=100\cdot1,05=105\left(g\right)\)
=> \(m_{HCl}=105-97,7=7,3\left(g\right)\)
=> \(n_{HCl}=\frac{7,3}{36,5}=0,2\left(mol\right)\)
BT Clo : \(n_{Cl_2}=\frac{1}{2}n_{HCl}=0,1\left(mol\right)\)
Mà theo lí thuyết : \(n_{Cl_2}=\frac{5}{2}n_{KMnO_4}=0,25\left(mol\right)\)
=> \(H\%=\frac{0,1}{0,25}\cdot100\%=40\%\)
Vì spu nổ thu được hh hai chất khí => \(\hept{\begin{cases}H_2\\HCl\end{cases}}\) (Vì H2 dư)
=> \(n_{hh}=\frac{13,44}{22,4}=0,6\left(mol\right)\)
=> \(n_{H_2\left(spu\right)}=n_{hh}-n_{HCl\left(spu\right)}=0,6-0,2=0,4\left(mol\right)\)
BT Hidro : \(\Sigma_{n_{H2\left(trong.binh\right)}}=n_{H_2\left(spu\right)}+\frac{1}{2}n_{HCl}=0,4+0,1=0,5\left(mol\right)\)
đọc thiếu đề câu a wtf
\(C_{M\left(HCl\right)}=\frac{0,2}{0,1}=2\left(M\right)\)

bài 5
Fe+6 HNO3 đặc → Fe(NO3)3+ 3NO2+3 H2O
Cu+ 4HNO3→ Cu(NO3)2+ 2NO2+2 H2O
Đặt nFe= xmol; nCu= y mol
Ta có mhhX= 56x+ 64y= 12,0
Số mol khí NO2 là nNO2= 3x+ 2y= 0,5 mol
Giải hệ có x= 0,1; y=0,1 → %mFe=46,67%
=>%mCu=53,33%
Bài 1 :
\(n_{HCl}=0,7.1=0,7\left(mol\right)\)
\(n_{H2}=\frac{5,6}{22,4}=0,25\left(mol\right)\)
\(2Al+6HCl\rightarrow2AlCl_3+3H_2\)
a______6a_____________3a
\(Fe+2HCl\rightarrow FeCl_2+H_2\)
b_______2b____________b
Giải hệ PT:
\(\left\{{}\begin{matrix}27a+56b=8,3\\3a+b=0,25\end{matrix}\right.\Rightarrow\left\{{}\begin{matrix}a=0,04\\b=0,129\end{matrix}\right.\)
\(\Rightarrow\%_{Fe}=\frac{0,04.56.100}{8,3}=26,98\%\)
\(\Rightarrow\%_{Al}=100\%-26,98\%=73\%\)

\(Đặt:\)
\(\left\{{}\begin{matrix}n_{Mg}=x\left(mol\right)\\\\n_{Fe}=y\left(mol\right)\end{matrix}\right.\)
\(n_{H_2}=\dfrac{6.72}{22.4}=0.3\left(mol\right)\)
\(Mg+2HCl\rightarrow MgCl_2+H_2\\ Fe+2HCl\rightarrow FeCl_2+H_2\)
\(m_{hh}=24x+56y=13.6\left(g\right)\\ n_{H_2}=x+y=0.3\left(mol\right)\)
\(\Rightarrow\left\{{}\begin{matrix}x=0.1\\y=0.2\end{matrix}\right.\)
\(\%Mg=\dfrac{0.1\cdot24}{13.6}\cdot100\%=17.64\%\\ \%Fe=100-17.64=82.36\%\)
\(n_{HCl}=2n_{H_2}=2\cdot0.3=0.6\left(mol\right)\)
\(V_{HCl}=\dfrac{0.6}{2}=0.3\left(l\right)\)
\(m_Y=m_{MgCl_2}+m_{FeCl_2}=0.1\cdot95+0.2\cdot127=34.9\left(g\right)\)

Câu 1
\(n_{H2S}=\frac{8,96}{22,4}=0,4\left(mol\right)\)
\(m_{dd_{NaOH}}=1,28.500=640\left(g\right)\)
\(\Rightarrow n_{NaOH}=\frac{640.25\%}{40}=4\left(mol\right)\)
\(2NaOH+H_2S\rightarrow Na_2S+2H_2O\)
0,8_______0,4______0,4____________
Dư NaOH nên chỉ tạo Na2S
\(\Rightarrow C\%_{Na2S}=\frac{0,4.78.100}{0,4.34+640}=4,77\%\)
Câu 2 :
\(n_{H2S}=0,3\left(mol\right)\)
\(RS+H_2SO_4\rightarrow RSO_4+H_2S\)
0,3____0,3______________0,3
\(\Rightarrow m_{H2SO4}=29,4\left(g\right)\)
\(R+S\underrightarrow{^{to}}RS\)
0,3_____0,3
\(\Rightarrow M_R=\frac{16,8}{0,3}=56\left(Fe\right)\)
Câu 3 :
\(n_{Fe}=0,1\left(mol\right);n_S=0,05\left(mol\right)\)
\(PTHH:Fe+S\rightarrow FeS\)
Ban đầu :__0,1_0,05
Phứng_0,05__0,05
Sau___ 0,05 ___0 ____ 0,05
Nên Fe dư
\(n_{H2S}=n_{FeS}=0,05\left(mol\right)\)
\(2n_{Fe}=2n_{H2}\Rightarrow n_{H2}=0,05\left(mol\right)\)
\(V\%_{H2S}=n\%_{H2S}=\frac{0,05}{0,1}.100\%=50\%\)
\(V\%_{H2}=100\%-50\%=50\%\)
\(n_{HCl\left(pư\right)}=2n_{H2S}+2n_{H2}=0,05.2+0,05.2=2\)
Đổi :
500ml=0,5l
125ml=0,125l
\(n_{NaOH}=0,125.0,1=0,0125\left(mol\right)\)
\(n_{NaOH}=n_{NaCl}=n_{HCl\left(dư\right)}=0,0125\left(mol\right)\)
\(\Sigma n_{HCl\left(bđ\right)}=0,0125+2=2,0125\left(mol\right)\)
\(\Rightarrow CM_{HCl}=\frac{2,0125}{0,5}=4,025M\)
Câu 4 dài ( Mình chưa ngắn ra cho dễ nhìn nha )
a, \(n_{SO2}=\frac{2,24}{22,4}=0,1\left(mol\right)\)
Đổi 800ml = 8l
\(n_{NaOH}=0,8.1=0,8\left(mol\right)\)
\(\frac{n_{NaOH}}{n_{SO2}}=\frac{0,8}{0,1}=8\)
Sinh ra muối Na2SO3, tính theo mol SO2
\(2NaOH+SO_2\rightarrow Na_2SO_3+H_2O\)
0,2________0,1_______0,1_____________
\(CM_{Na2SO3}=\frac{0,1}{0,8}=0,125M\)
\(CM_{NaOH\left(dư\right)}=\frac{0,8-0,2}{0,8}=0,75M\)
b,\(n_{SO2}=0,2\left(mol\right)\)
Đổi 250ml=0,25l
\(n_{NaOH}=0,25\left(mol\right)\)
\(\frac{n_{NaOH}}{n_{SO2}}=\frac{0,25}{0,2}=1,25\)
Sinh ra 2 muối NaHSO3 và Na2SO3
\(n_{NaHSO3}+n_{Na2SO3}=0,2\)
\(n_{NaHSO3}+2n_{Na2SO3}=0,25\)
\(\Rightarrow\left\{{}\begin{matrix}n_{NaHSO3}=0,15\\n_{Na2SO3}=0,05\end{matrix}\right.\)
\(m_{muoi}=m_{NaHSO3}+m_{Na2SO3}\Leftrightarrow m_{muoi}=0,15.104+0,05.126\)
\(\Rightarrow m_{muoi}=21,9\left(g\right)\)
c,\(n_{SO3}=0,03\left(mol\right)\)
\(n_{KOH}=\frac{13,95.1,147.28\%}{56}=0,08\)
\(\frac{m_{KOH}}{n_{SO2}}=\frac{0,08}{0,03}=2,67\)
Sinh ra muối K2SO3 và KOH dư
\(2KOH+SO_2\rightarrow K_2SO_3+H_2O\)
0,06_____0,03______0,03________
\(C\%_{KOH\left(Dư\right)}=\frac{\left(0,08-0,06\right).56}{13,95.1,147+0,03.64}.100\%=6,25\%\)
\(C\%_{K2SO3}=\frac{0,03.158}{13,95.1,147+0,03.64}.100\%=26,45\%\)