Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

$n_{AgNO_3} = \dfrac{150.6,8\%}{170} =0,06(mol)$
$Cu+ 2AgNO_3 \to Cu(NO_3)_2 + 2Ag$
Theo PTHH :
$n_{Cu} = \dfrac{1}{2}n_{AgNO_3} = 0,03(mol)$
$m_{Cu} =0,03.64 = 1,92(gam)$
$n_{Ag} = n_{AgNO_3} = 0,06(mol)$
$\Rightarrow m_{dd\ sau\ pư} = 1,92 + 150 - 0,06.108 = 145,44(gam)$
$C\%_{Cu(NO_3)_2} = \dfrac{0,03.188}{145,44}.100\% = 3,88\%$

Ta có: \(m_{CuSO_4}=40.10\%=4\left(g\right)\Rightarrow n_{CuSO_4}=\dfrac{4}{160}=0,025\left(mol\right)\)
PT: \(Zn+CuSO_4\rightarrow ZnSO_4+Cu\)
Theo PT: \(n_{Zn}=n_{ZnSO_4}=n_{Cu}=n_{CuSO_4}=0,025\left(mol\right)\)
\(\Rightarrow m_{Zn}=0,025.65=1,625\left(g\right)\)
Ta có: m dd sau pư = 1,625 + 40 - 0,025.64 = 40,025 (g)
\(\Rightarrow C\%_{ZnSO_4}=\dfrac{0,025.161}{40,025}.100\%\approx10,056\%\)

\(n_{CuSO_4}=\dfrac{200.8}{100.160}=0,1(mol)\\ PTHH:Fe+CuSO_4\to FeSO_4+Cu\\ a,n_{Cu}=n_{Fe}=n_{CuSO_4}=0,1(mol)\\ \Rightarrow m_{Cu}=0,1.64=6,4(g);m_{Fe}=0,1.56=5,6(g)\\ b,n_{FeSO_4}=0,1(mol)\\ \Rightarrow C\%_{FeSO_4}=\dfrac{0,1.152}{5,6+200-6,4}.100\%=7,63\%\)

mAgNO3=5,1g
=> nAgNO3=0,03mol
PTHH: Zn+ 2AgNO3=>Zn(NO3)2+2Ag
0,06 <-0,03 ->0,03 ->0,06
mZn đã dùng:m=0,06.65=3,9g

\(m_{ZnSO_4}=\dfrac{241,5.10}{100}=24,15\left(g\right)=>n_{ZnSO_4}=\dfrac{24,15}{161}=0,15\left(mol\right)\)
PTHH: 2Al + 3ZnSO4 --> Al2(SO4)3 + 3Zn
_____0,1<----0,15-------->0,05----->0,15
=> mAl = 0,1.27 = 2,7(g)
=> mZn = 0,15.65=9,75(g)
b) mdd sau pư = 2,7 + 241,5 - 9,75 = 234,45(g)
=> \(C\%\left(Al_2\left(SO_4\right)_3\right)=\dfrac{0,05.342}{234,45}.100\%=7,294\%\)

nCuSO4 = \(\dfrac{20.10\%}{160}\) = 0,0125 (mol)
Zn + CuSO4 \(\rightarrow\) ZnSO4 + Cu
0,0125 \(\leftarrow\) 0,0125 \(\rightarrow\) 0,0125 \(\rightarrow\) 0,0125 (mol)
mZn pư = 0,0125 . 65= 0,8125 (g)
mdd spư = 20 + 0,8125 - 0,0125.64 = 20,0125 (g)
C%(ZnSO4) = \(\dfrac{0,0125.161}{20,0125}\) . 100%= 10,06%

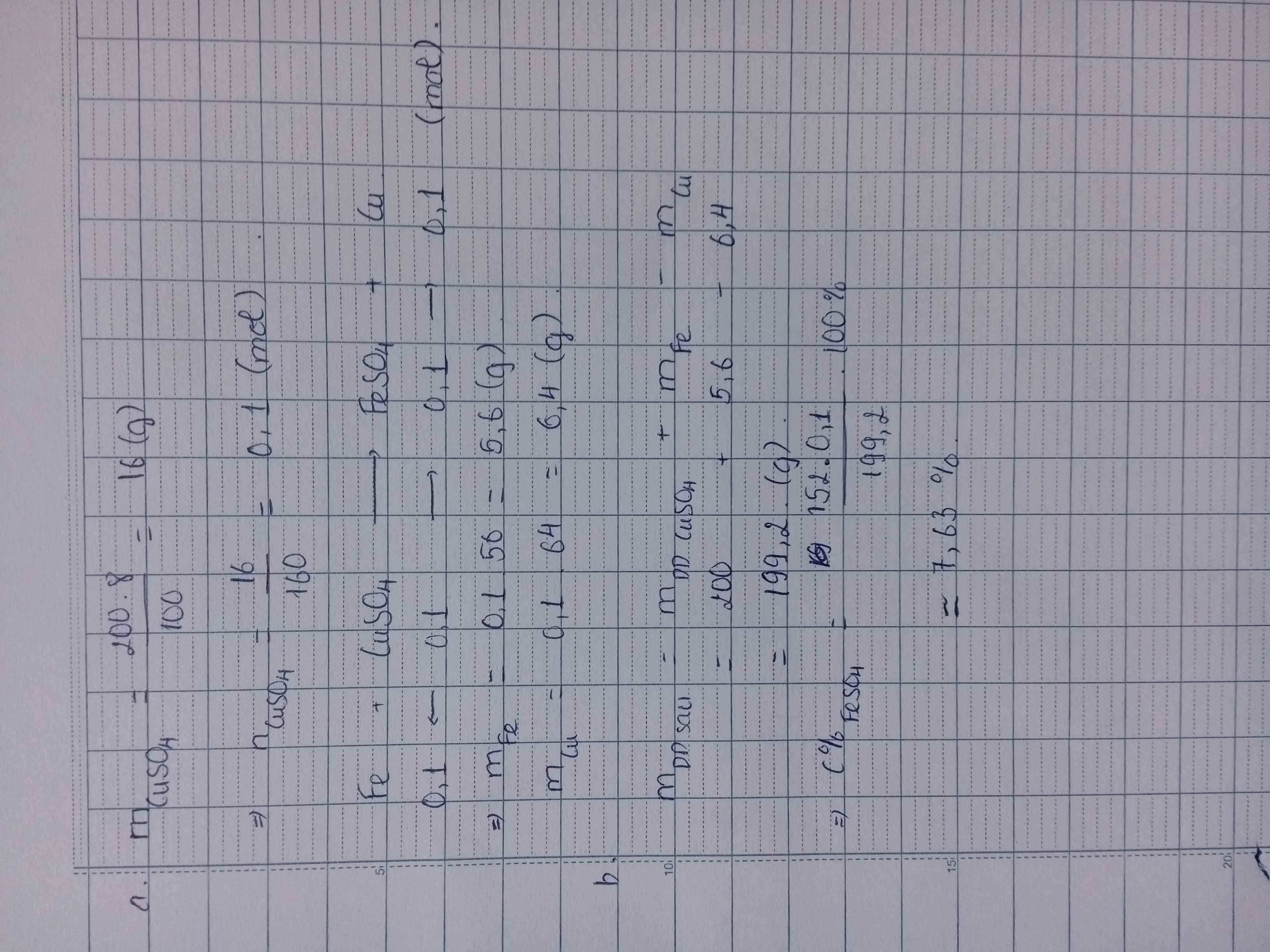
\(n_{AgNO_3}=\dfrac{25.5\cdot10\%}{170}=0.015\left(mol\right)\)
\(Cu+2AgNO_3\rightarrow Cu\left(NO_3\right)_2+2Ag\)
\(0.0075.....0.015\)
\(m_{Cu}=0.0075\cdot64=0.48\left(g\right)\)