Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

a, PT: \(CH_3COOH+C_2H_5OH\underrightarrow{_{H_2SO_{4\left(đ\right)}}}CH_3COOC_2H_5+H_2O\)
b, Ta có: \(n_{CH_3COOH}=\dfrac{30}{60}=0,5\left(mol\right)\)
\(n_{C_2H_5OH}=\dfrac{27,6}{46}=0,6\left(mol\right)\)
Xét tỉ lệ: \(\dfrac{0,5}{1}< \dfrac{0,6}{1}\), ta được C2H5OH.
Theo PT: \(n_{CH_3COOC_2H_5\left(LT\right)}=n_{CH_3COOH}=0,5\left(mol\right)\)
\(\Rightarrow m_{CH_3COOC_2H_5\left(LT\right)}=0,5.88=44\left(g\right)\)
Mà: m CH3COOC2H5 (TT) = 35,2 (g)
\(\Rightarrow H\%=\dfrac{35,2}{44}.100\%=80\%\)
Bạn tham khảo nhé!

\(a) C_2H_5OH + CH_3COOH \buildrel{{H_2SO_4}}\over\rightleftharpoons CH_3COOC_2H_5 + H_2O\\ b) n_{CH_3COOH} = n_{C_2H_5OH} = \dfrac{9,2}{46} = 0,2(mol)\\ m_{CH_3COOH} = 0,2.60 = 12(gam)\\ c) n_{CH_3COOC_2H_5} = n_{C_2H_5OH} = 0,2(mol)\\ m_{CH_3COOC_2H_5} = 0,2.88 = 17,6(gam)\)

\(n_{CH_3COOC_2H_5}=\dfrac{4,4}{88}=0,05\left(mol\right)\)
PTHH: CH3COOH + C2H5OH --H2SO4(đ),to--> CH3COOC2H5 + H2O
0,05<--------------------------------------0,05
=> \(m_{CH_3COOH\left(lý.thuyết\right)}=0,05.60=3\left(g\right)\)
=> \(m_{CH_3COOH\left(tt\right)}=\dfrac{3.100}{60}=5\left(g\right)\)

CH3COOH=0,15 mol
C2H5OH=0,1 mol
C2H5OH+CH3COOH->CH3COOC2H5+H2O
0,075-------------------------------0,075
=>CH3COOH dư
n este =0,075 mol
=>H=\(\dfrac{0,075}{0,1}\)100=75%
\(n_{CH_3COOH}=\dfrac{9}{60}=0,15\left(mol\right)\\ n_{C_2H_5OH}=\dfrac{4,6}{46}=0,1\left(mol\right)\\ CH_3COOH+C_2H_5OH⇌CH_3COOC_2H_5+H_2O\\ LTL:\dfrac{0,15}{1}>\dfrac{0,1}{1}\\ \Rightarrow TínhtheosốmolC_2H_5OH\\\Rightarrow n_{CH_3COOC_2H_5\left(lt\right)}=n_{C_2H_5OH}=0,1\left(mol\right)\\ n_{CH_3COOC_2H_5\left(tt\right)}=\dfrac{6,6}{88}=0,075\left(mol\right)\\ \Rightarrow H=\dfrac{0,075}{0,1}.100=75\%\)
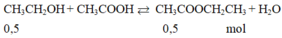
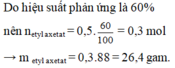

nC2H5OH = 8.05/46 = 0.175 (mol)
nCH3COOH = 36/60 = 0.6 (mol)
nCH3COOC2H5 = 12.32/88 = 0.14 (mol)
C2H5OH + CH3COOH <-H2SO4đ,t0-> CH3COOC2H5 + H2O
1.......................1
0.175................0.6
LTL : 0.175/1 < 0.6/1
=> CH3COOH dư
mCH3COOH (dư) = ( 0.6 - 0.175) * 60 = 25.5 (g)
nCH3COOC2H5 = nC2H5OH = 0.175 (mol)
H% = 0.14/0.175 * 100% = 80%