Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

\(n_{CH_4}=\dfrac{5,6}{22,4}=0,25\left(mol\right)\)
PTHH: CH4 + 2O2 --to--> CO2 + 2H2O
0,25-->0,5-------->0,25
=> \(V_{O_2}=0,5.22,4=11,2\left(l\right)\)
\(V_{CO_2}=0,25.22,4=5,6\left(l\right)\)

\(V_{O_2}=\dfrac{336}{5}=67,2\left(ml\right)=0,0672\left(l\right)\\ n_{O_2}=\dfrac{0,0672}{22,4}=0,003\left(mol\right)\\ CH_4+2O_2\rightarrow\left(t^o\right)CO_2+2H_2O\\ n_{CO_2}=n_{CH_4}=\dfrac{0,003}{2}=0,0015\left(mol\right)\\ a,V_{CH_4\left(đktc\right)}=0,0015.22,4=0,0336\left(l\right)\\ b,V_{CO_2\left(đktc\right)}=V_{CH_4\left(đktc\right)}=0,0336\left(l\right)\)

\(CH_4+2O_2\underrightarrow{t^o}CO_2+2H_2O\)
0.15 0.3 0.15
\(n_{CH_4}=\dfrac{3.36}{22.4}=0.15mol\)
\(V_{O_2}=0.3\times22.4=6.72l\)
\(V_{CO_2}=0.15\times22.4=3.36l\)

\(n_{CH_4}=\dfrac{2,24}{22,4}=0,1mol\)
\(CH_4+2O_2\underrightarrow{t^o}CO_2+2H_2O\)
0,1 0,2 0,1
\(V_{O_2}=0,2\cdot22,4=4,48l\)
\(V_{CO_2}=0,1\cdot22,4=2,24l\)

nCH4 = 2.24/22.4 = 0.1 (mol)
CH4 + 2O2 -to-> CO2 + 2H2O
0.1____0.2______0.1
VO2 = 0.2*22.4 = 4.48 (l)
VCO2 = 0.1*22.4=2.24 (l)

\(CH_4+2O_2\rightarrow\left(t^o\right)CO_2+2H_2O\\ n_{CH_4}=\dfrac{11,2}{22,4}=0,5\left(mol\right)\Rightarrow n_{CO_2}=n_{CH_4}=0,5\left(mol\right);n_{O_2}=2.n_{CH_4}=2.0,5=1\left(mol\right)\\ V_{O_2\left(đktc\right)}=n_{O_2}.22,4=1.22,4=22,4\left(l\right)\\ V_{CO_2\left(đktc\right)}=n_{CO_2}.22,4=0,5.22,4=11,2\left(l\right)\)

a, PT: \(CH_4+2O_2\underrightarrow{t^o}CO_2+2H_2O\)
\(2H_2+O_2\underrightarrow{t^o}2H_2O\)
Ta có: \(n_{CH_4}+n_{H_2}=\dfrac{11,2}{22,4}=0,5\left(mol\right)\left(1\right)\)
Theo PT: \(n_{H_2O}=2n_{CH_4}+n_{H_2}=\dfrac{16,2}{18}=0,9\left(mol\right)\left(2\right)\)
Từ (1) và (2) \(\Rightarrow\left\{{}\begin{matrix}n_{CH_4}=0,4\left(mol\right)\\n_{H_2}=0,1\left(mol\right)\end{matrix}\right.\)
\(\Rightarrow\left\{{}\begin{matrix}\%V_{CH_4}=\dfrac{0,4.22,4}{11,2}.100\%=80\%\\\%V_{H_2}=20\%\end{matrix}\right.\)
b, Theo PT: \(n_{CO_2}=n_{CH_4}=0,4\left(mol\right)\Rightarrow V_{CO_2}=0,4.22,4=8,96\left(l\right)\)

Bài 2.
\(n_{C_2H_2}=\dfrac{4,48}{22,4}=0,2mol\)
\(n_{O_2}=\dfrac{6,72}{22,4}=0,3mol\)
\(2C_2H_2+5O_2\rightarrow\left(t^o\right)4CO_2+2H_2O\)
0,2 > 0,3 ( mol )
0,3 0,24 0,12 ( mol )
\(m_{CO_2}=0,24.44=10,56g\)
\(m_{H_2O}=0,12.18=2,16g\)
PTHH: 2CO + O2→2CO2
C2H4 + 3O2→ 2CO2 +2 H2O
nH2O= mM=\(\dfrac{1,8}{18}\)=0,1(mol)
nC2H4=\(\dfrac{1}{2}\).nH2O=\(\dfrac{1}{2}\).0,1=0,05(mol)
=> VC2H4=n.22,4=0,05.22,4=1,12(lít)
->VCO=4,48 − 1,12= 3,36(lít)
b) nCO2 (1)=nCO=\(\dfrac{3,36}{22,4}\)=0,15(mol)
mCO2 (1)=n.M=0,15.44=6,6(g)
nCO2 (2)=2.nC2H4=2.0,05=0,1(mol)
mCO2 (2)=n.M=0,1.44=4,4(g)
mCO2 sau pư=6,6 + 4,4= 11(g)
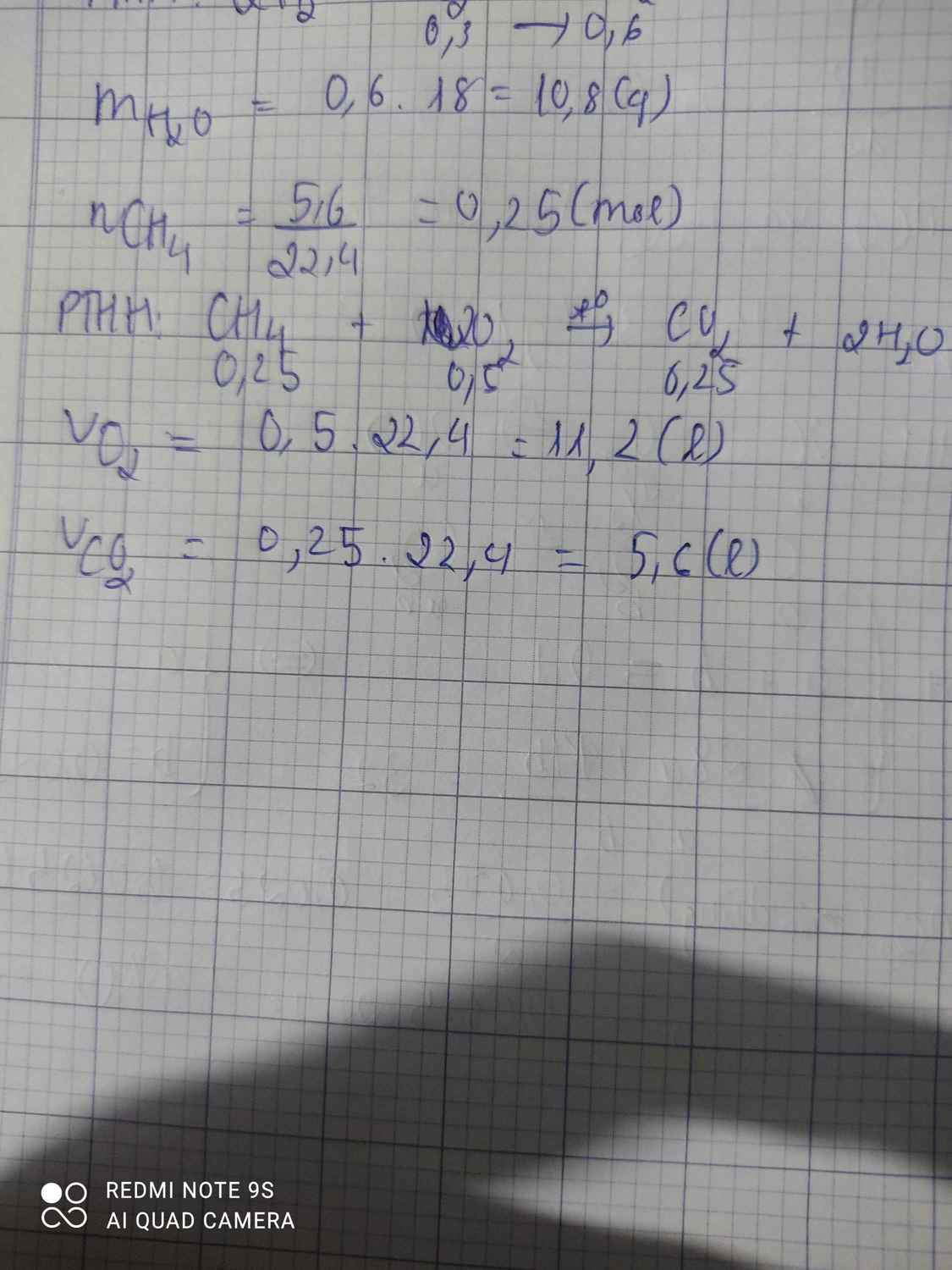
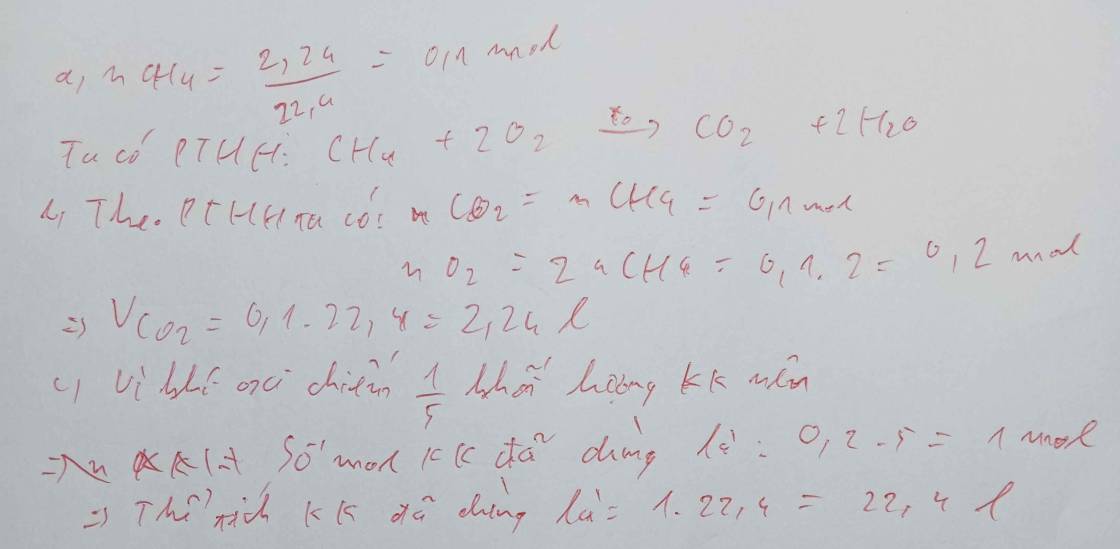
\(n_{CH_4}=\dfrac{3.2}{22.4}=\dfrac{1}{7}\left(mol\right)\)
\(CH_4+2O_2\underrightarrow{t^0}CO_2+2H_2O\)
\(\dfrac{1}{7}.............\dfrac{1}{7}\)
\(V_{CO_2}=\dfrac{1}{7}\cdot22.4=3.2\left(l\right)\)
Cảm ơn câu trả lời của bạn nha ( ˙꒳˙ )