Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

\(n_{NaOH}=0,1.0,02=0,002\left(mol\right)\\ n_{SO_3}=n_{H_2SO_4}=\dfrac{0,002}{2}=0,001\left(mol\right)\\ SO_3+H_2O\rightarrow H_2SO_4\\ m_{SO_3}=0,001.80=0,08\left(g\right)\\ m_{oleum\left(lấy\right)}=\dfrac{8,36}{100}=0,0836\left(g\right)\\ Đặt:oleum:ySO_3.H_2O\\ m_{H_2O}=0,0836-0,08=0,0036\left(g\right)\Rightarrow n_{H_2O}=\dfrac{0,0036}{18}=0,0002\left(mol\right)\\ n_{SO_3}=\dfrac{0,001}{0,0002}=5\\ \Rightarrow CTPToleum:5SO_3.H_2O\)

\(Đặt:n_{HCl}=3a\left(mol\right);n_{H_2SO_4}=a\left(mol\right)\\n_{NaOH}=0,05.0,5=0,025\left(mol\right)\\ NaOH+HCl\rightarrow NaCl+H_2O\\ 2NaOH+H_2SO_4 \rightarrow Na_2SO_4+2H_2O\\ \rightarrow n_{NaOH\left(tổng\right)}=3a+2a=5a\left(mol\right)\\ \rightarrow5a=0,025\\ \Leftrightarrow a=0,005\left(mol\right)\\ C_{MddHCl}=\dfrac{0,005.3}{0,1}=0,15\left(M\right)\\ C_{MddH_2SO_4}=\dfrac{0,005}{0,1}=0,05\left(M\right)\)

Đáp án A
Gọi số mol HCl và H2SO4 lần lượt là 2x và 3x mol
HCl + NaOH → NaCl + H2O
2x 2x mol
H2SO4+ 2NaOH→ Na2SO4+ 2H2O
3x 6x mol
nNaOH= 2x+6x=0,5.0,8 suy ra x= 0,05 mol
Do đó CM HCl= 2.0,05/0,1=1M; CMH2SO4= 3.0,05/0,1=1,5M

a, \(n_{H^+}=n_{OH^-}=9.10^{-3}\left(mol\right)\Rightarrow C_{M\left(H_2SO_4\right)}=\dfrac{\dfrac{9.10^{-3}}{2}}{0,05}=0,09M\)
b, \(\left[SO_4^{2-}\right]=\dfrac{4,5.10^{-3}}{0,05+0,15}=0,6M\)
\(\left[Na^+\right]=\dfrac{0,15.0,06}{0,05+0,15}=0,045M\)
\(\left[H^+\right]=\left[OH^-\right]=\dfrac{9.10^{-3}}{0,05+0,15}=0,045M\)

a) \(n_{NaOH}=0,2.1=0,2\left(mol\right)\); \(n_{HNO_3}=0,2.0,5=0,1\left(mol\right)\)
\(NaOH+HNO_3\rightarrow NaNO_3+H_2O\)
0,2.............0,1
Lập tỉ lệ : \(\dfrac{0,2}{1}>\dfrac{0,1}{1}\) => Sau phản ứng NaOH dư
Dung dịch D gồm NaNO3 và NaOH dư
\(n_{NaNO_3}=n_{HNO_3}=0,1\left(mol\right)\)
\(n_{NaOH\left(pứ\right)}=n_{HNO_3}=0,1\left(mol\right)\)
\(n_{NaOH\left(dư\right)}=0,2-0,1=0,1\left(mol\right)\)
Ion trong dung dịch D : Na+ , NO3-, OH-
\(\left[Na^+\right]=\dfrac{0,1+0,1}{0,2}=1M\)
\(\left[NO_3^-\right]=\dfrac{0,1}{0,2}=0,5M\)
\(\left[OH^-\right]=\dfrac{0,1}{0,2}=0,5M\)
b)Trong dung dịch D chỉ có NaOH dư phản ứng
\(2NaOH+H_2SO_4\rightarrow Na_2SO_4+H_2O\)
0,1................0,05
=> \(V_{H_2SO_4}=\dfrac{0,05}{1}=0,05\left(l\right)\)

\(n_{KOH}=0.1\cdot1=0.1\left(mol\right)\)
\(n_{H_2SO_4}=0.3\cdot0.5=0.15\left(mol\right)\)
\(2KOH+H_2SO_4\rightarrow K_2SO_4+H_2O\)
\(0.1..........0.05...............0.05\)
Dung dịch D : 0.05 (mol) K2SO4 , 0.1 (mol) H2SO4
\(\left[K^+\right]=\dfrac{0.05\cdot2}{0.1+0.3}=0.25\left(M\right)\)
\(\left[H^+\right]=\dfrac{0.1\cdot2}{0.1+0.3}=0.5\left(M\right)\)
\(\left[SO_4^{2-}\right]=\dfrac{0.05+0.1}{0.1+0.3}=0.375\left(M\right)\)
\(2NaOH+H_2SO_4\rightarrow K_2SO_4+H_2O\)
\(0.2..................0.1\)
\(V_{dd_{NaOH}}=\dfrac{0.2}{1}=0.2\left(l\right)\)

Đáp án C
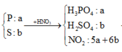
P + 5HNO3 → H3PO4 + 5NO2↑ + H2O
a →5a → a (mol)
S + 6HNO3 → H2SO4 + 6NO2↑ + 2H2O
b→ 6b → b (mol)
∑ nHNO3 pư = (5a + 6b) mol
=> nHNO3 dư = (5a + 6b).20% = a + 1,2b (mol)
∑ nH+ = nHNO3 + 3nH3PO4 + 2nH2SO4 = ( a + 1,2b) + 3a + 2b
=> ∑ nH+ = 4a + 3,2b (mol)
Phản ứng trung hòa: H+ + OH-→ H2O
=> nNaOH = nOH- = nH+ = 4a + 3,2b (mol)

Đáp án D
→ n N a O H = 3 a + 2 b + 0 , 2 ( 5 a + 6 b ) = 4 a + 3 , 2 b
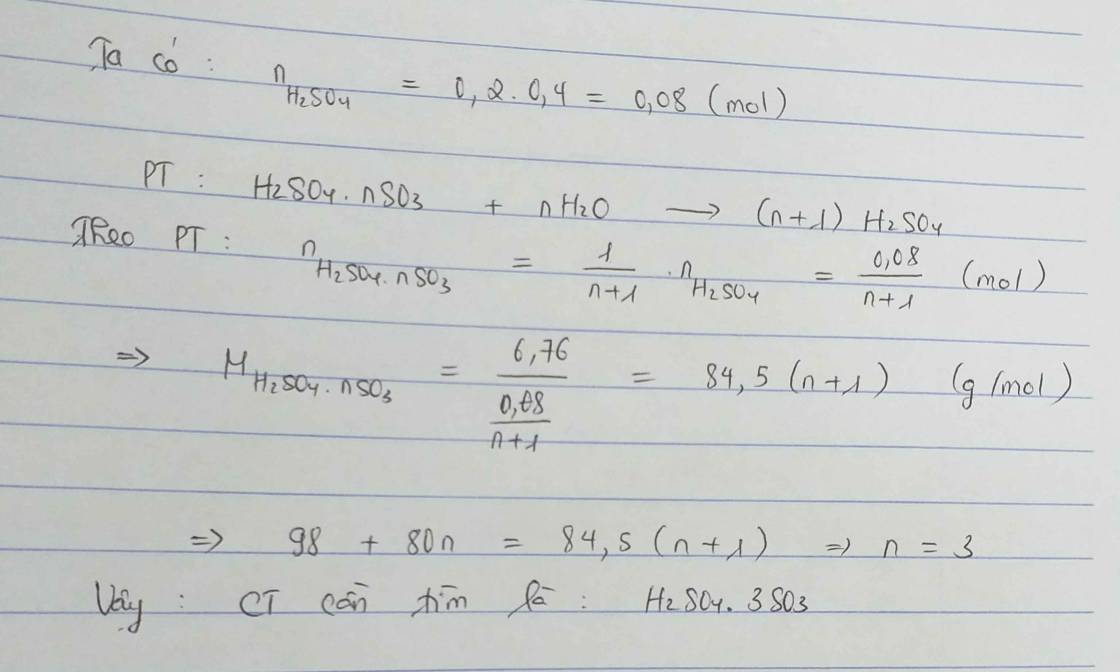
\(CT:H_2SO_4\cdot nSO_3\left(x\right)\)
\(n_{NaOH}=0.1\cdot0.175=0.0175\left(mol\right)\)
\(H^++OH^-\rightarrow H_2O\)
\(0.0175...0.0175\)
\(n_{H_2SO_4}=8.75\cdot10^{-3}\left(mol\right)\)
\(BTS:\)
\(n_{H_2SO_4}=x+xn=8.75\cdot10^{-3}\left(1\right)\)
\(m=98x+80xn=0.826\left(2\right)\)
\(\left(1\right),\left(2\right):\) \(x=7\cdot10^{-3},xn=1.75\cdot10^{-3}\)
\(\Rightarrow n=4\)
\(\dfrac{n_{SO_3}}{n_{H_2SO_4}}=4\)
\(CT:H_2SO_{_{ }4}\cdot4SO_3\)