Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

\(a) n_{Fe_3O_4} = a(mol) ; n_{CuO} = b(mol)\\ \Rightarrow 232a + 80b = 117,6(1)\\ Fe_3O_4 + 4H_2 \xrightarrow{t^o} 3Fe + 4H_2O\\ CuO + H_2 \xrightarrow{t^o} Cu + H_2O\\ n_{H_2} = 4a + b = \dfrac{40,32}{22,4}=1,8(2)\\ (1)(2)\Rightarrow a = 0,3 ;b = 0,6\\ \%m_{Fe_3O_4} = \dfrac{0,3.232}{117,6}.100\% =59,18\%\\ \%m_{CuO} = 100\%-59,18\% = 40,82\%\)
\(b)\\ n_{Fe} = 3a = 0,9(mol)\\ n_{Cu} = b = 0,6(mol)\\ \%m_{Fe} = \dfrac{0,9.56}{0,9.56+0,6.64}.100\% = 56,76\%\\ \%m_{Cu} = 100\% - 56,76\% = 43,24\%\)

\(n_{H_2}=\dfrac{17.92}{22.4}=0.8\left(mol\right)\)
\(n_{Fe_3O_4}=\dfrac{69.6}{232}=0.3\left(mol\right)\)
\(Fe_3O_4+4H_2\underrightarrow{t^0}3Fe+4H_2O\)
\(0.2..............0.8\)
\(m_{Fe_3O_4\left(dư\right)}=\left(0.3-0.2\right)\cdot232=23.2\left(g\right)\)
\(Zn+2HCl\rightarrow ZnCl_2+H_2\)
\(0.8......................................0.8\)
\(m_{Zn}=0.8\cdot65=52\left(g\right)\)

\(n_{Fe}=\dfrac{12.6}{56}=0.225\left(mol\right)\)
\(n_{O_2}=\dfrac{4.2}{22.4}=0.1875\left(mol\right)\)
\(3Fe+2O_2\underrightarrow{^{^{t^0}}}Fe_3O_4\)
\(3.........2\)
\(0.225......0.1875\)
Lập tỉ lệ : \(\dfrac{0.225}{3}< \dfrac{0.1875}{2}\Rightarrow O_2dư\)
\(m_{O_2\left(dư\right)}=\left(0.1875-0.225\cdot\dfrac{2}{3}\right)\cdot32=1.2\left(g\right)\)
\(m_{Fe_3O_4}=\dfrac{0.225}{3}\cdot232=17.4\left(g\right)\)

\(a,PTHH:Fe_3O_4+4H_2\xrightarrow{t^o}3Fe+4H_2O\\ n_{H_2}=\dfrac{6,72}{22,4}=0,3(mol);n_{Fe_3O_4}=\dfrac{46,4}{232}=0,2(mol)\)
Vì \(\dfrac{n_{H_2}}{4}<\dfrac{n_{Fe_3O_4}}{1}\) nên \(Fe_3O_4\) dư
\(n_{Fe_3O_4(dư)}=0,2-\dfrac{0,3}{4}=0,125(mol)\\ \Rightarrow m_{Fe_3O_4(dư)}=0,125.232=29(g)\\ b,n_{Fe}=\dfrac{3}{4}n_{H_2}=0,225(mol)\\ \Rightarrow m_{Fe}=0,225.56=12,6(g)\)

a) PTHH: \(CuO+H_2\underrightarrow{t^o}Cu+H_2O\)
b+c) Ta có: \(n_{H_2}=\dfrac{2,24}{22,4}=0,1\left(mol\right)=n_{CuO}=n_{Cu}\)
\(\Rightarrow\left\{{}\begin{matrix}m_{Cu}=0,1\cdot64=6,4\left(g\right)\\m_{CuO}=80\cdot0,1=8\left(g\right)\end{matrix}\right.\)
d) Ta có: \(n_{CuO}=\dfrac{12}{80}=0,15\left(mol\right)\)
\(\Rightarrow\) CuO còn dư, Hidro p/ứ hết
\(\Rightarrow n_{CuO\left(dư\right)}=0,05\left(mol\right)\) \(\Rightarrow m_{CuO\left(dư\right)}=80\cdot0,05=4\left(g\right)\)

\(n_{H_2}=\dfrac{11,2}{22,4}=0,5\left(m\right)\);\(n_{Fe_2O_3}=\dfrac{80}{160}=0,5\left(m\right)\)
\(PTHH:Fe_2O_3+3H_2\xrightarrow[]{t^o}2Fe+3H_2O\)
ta có tỉ lệ:\(\dfrac{0,5}{1}>\dfrac{0,5}{3}\)=>\(Fe_2O_3\) dư
H2 phản ứng hết
\(PTHH:Fe_2O_3+3H_2\xrightarrow[]{t^o}2Fe+3H_2O\)
tỉ lệ :1 3 2 3
số mol :0,17 0,5 0,3 0,5
\(m_{Fe_2O_3}=0,3.160=48\left(g\right)\)

mFe3O4 = 34.8 / 232 = 0.15 (mol)
nH2 = 8.96 / 22.4 = 0.4 (mol)
Fe3O4 + 4H2 -t0-> 3Fe + 4H2O
Bđ: 0.15.......0.4
Pư: 0.1..........0.4........0.3........0.4
Kt: 0.05.........0............0.3.......0.4
mFe3O4(dư) = 0.05 * 232 = 11.6 (g)
mFe = 0.3 * 56 = 16.8 (g)
VH2O = 0.4 * 22.4 = 8.96 (l)
2Fe + 6H2SO4(đ) => Fe2(SO4)3 + 3SO2 + 6H2O
0.3...........0.9.................0.15...........0.45
mH2SO4 = 0.9 * 98 = 88.2 (g)
C% H2SO4 = 88.2 * 100 / 98 = 90 %
VSO2 = 0.45 * 22.4 = 10.08 (l)
mX = 16.8 + 98 - 0.45 * 64 = 86 (g)
C% Fe2(SO4)3 = 0.15 * 400 / 86 * 100% = 69.76%
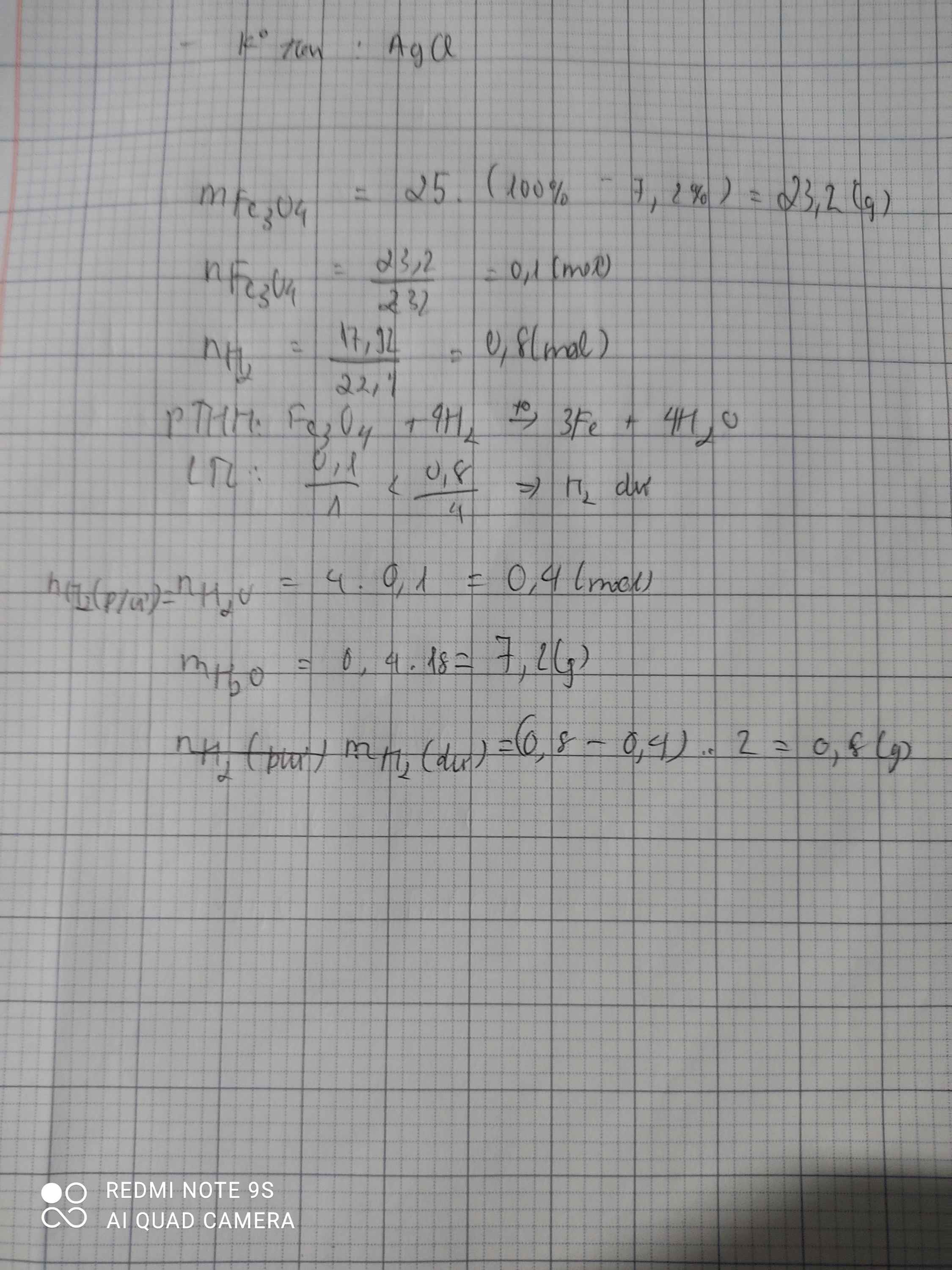
PTHH: 4H2 + Fe3O4 ➞ 3Fe + 4H2O
a) \(n_{H_2}=\dfrac{17,92}{22,4}=0,8\) (mol)
\(n_{Fe_3O_4}=\dfrac{69,6}{232}=0,3\) (mol)
Lập tỉ số: \(\dfrac{0,8}{4}=0,2\) < \(\dfrac{0,3}{1}=0,3\)
⇒ H2 hết, Fe3O4 dư
Theo PT: \(n_{Fe_3O_4}=\dfrac{1}{4}n_{H_2}=\dfrac{1}{4}.0,8=0,2\) (mol)
⇒ \(n_{Fe_3O_4}\)dư = 0,3 - 0,2 = 0,1 (mol)
⇒ \(m_{Fe_3O_4}\)dư = 0,1 . 232 = 23,2 (g)
b) Theo PT: \(n_{Fe}=\dfrac{3}{4}n_{H_2}=\dfrac{3}{4}.0,8=0,6\) (mol)
⇒ mFe= 0,6 . 56 = 33,6 (g)
Fe3O4 + 4H2 -> 3Fe + 4H2O
nH2=0,8(mol)
nFe3O4=0,3(mol)
Vì \(\dfrac{0,8}{4}< 0,3\) nên sau PƯ thì Fe3O4 dư 0,1(mol)
mFe3O4 dư=0,1.232=23,2(g)
Theo PTHH ta có:
nFe=\(\dfrac{3}{4}\)nH2=0,6(mol)
mFe=56.0,6=33,6(g)