Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

PTPƯ:
FexOy + 2yHCI -------------> xFeCI2y/x +yH2O
1mol ----------------------------> xmol
16/(56x +16y)mol-------------> 32,5/(56 + 71y/x)
=> 16x/(56x + 16y) = 32,5/(56 + 71y/x)
=> 896x +1136y = 1820x + 520y
=>616y = 924x
=> x/y = 2/3
Vậy CT của oxít sắt là Fe2O3

\(n_{H_2}=\dfrac{8.96}{22.4}=0.4\left(mol\right)\)
\(BTKL:\)
\(m+0.4\cdot2=28.4+7.2\)
\(\Rightarrow m=34.8\left(g\right)\)
\(b.\)
\(m_{Fe}=0.59155\cdot28.4=16.8\left(g\right)\)
\(n_{Fe}=\dfrac{16.8}{56}=0.3\left(mol\right)\)
\(PTHH:\)
\(\dfrac{x}{y}=\dfrac{n_{Fe}}{n_{H_2}}=\dfrac{0.3}{0.4}=\dfrac{3}{4}\)
\(CT:Fe_3O_4\)

\(Fe_xO+yH_2\underrightarrow{^{to}}xFe+yH_2O\)
Ta có:
\(n_{H2}=\frac{11,2}{22,4}=0,5\left(mol\right)\)
\(n_{H2O}=\frac{7,2}{18}=0,4\left(mol\right)\)
\(\Rightarrow H=\frac{0,4}{0,5}.100\%=80\%\)
Ta lại có:
\(m_{Fe}=40.84\%=33,6\left(g\right)\)
\(\Rightarrow n_{Fe}=\frac{33,4}{56}=0,6\left(mol\right)\)
\(n_{H2}=2n_{H2O}=0,4.2=0,8\left(mol\right)\)
\(\rightarrow x:y=0,6:0,8=3:4\)
Vậy CTHH của A là Fe3O4
\(\Rightarrow m_{Fe3O4_{du}}=40-33,6=6,4\left(g\right)\)
\(Fe_3O_4+4H_2\underrightarrow{^{to}}3Fe+4H_2O\)
\(n_{Fe3O4_{tham.gia}}=0,25.n_{H2O}=0,25.0,5=0,1\left(mol\right)\)
\(\Rightarrow m_{Fe3O4_{tham.gia}}=0,1.232=23,2\left(g\right)\)
\(\Rightarrow m=23,2+6,4=29,6\left(g\right)\)

Ta có: \(n_{H_2}=\dfrac{7,392}{22,4}=0,33\left(mol\right)\)
Gọi: nH2 (pư) = a (mol) ⇒ nH2 (dư) = 10%a (mol)
⇒ a + 10%a = 0,33
⇒ a = 0,3 (mol)
Có: \(H_2+O_{\left(trongoxit\right)}\rightarrow H_2O\)
⇒ nO (trong oxit) = 0,3 (mol)
\(\Rightarrow n_{Fe}=\dfrac{16-m_{O\left(trongoxit\right)}}{56}=0,2\left(mol\right)\)
\(\Rightarrow\dfrac{x}{y}=\dfrac{0,2}{0,3}=\dfrac{2}{3}\)
Vậy: CTHH cần tìm là Fe2O3.

\(CT:Fe_xO_y\)
\(Fe_xO_y+yH_2\underrightarrow{^{t^o}}xFe+yH_2O\left(1\right)\)
\(Fe+2HCl\rightarrow FeCl_2+H_2\left(2\right)\)
\(n_{Fe}=n_{H_2\left(2\right)}=\dfrac{4.032}{22.4}=0.18\left(mol\right)\)
\(n_{H_2\left(1\right)}=\dfrac{y}{x}\cdot n_{Fe}=\dfrac{5.376}{22.4}=0.24\left(mol\right)\)
\(\Leftrightarrow\dfrac{y}{x}\cdot0.18=0.24\)
\(\Leftrightarrow\dfrac{x}{y}=\dfrac{3}{4}\)
\(CT:Fe_3O_4\)
\(m_{Fe_3O_4}=\dfrac{0.18}{3}\cdot232=13.92\left(g\right)\)
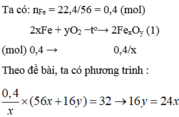
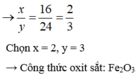
\(n_{Fe_xO_y}=\dfrac{32}{56x+16y}\left(mol\right)\)
PTHH: FexOy + yH2 --to--> xFe + yH2O
\(\dfrac{32}{56x+16y}\)----------->\(\dfrac{32x}{56x+16y}\)
=> \(\dfrac{32x}{56x+16y}=\dfrac{22,4}{56}=0,4\)
=> \(\dfrac{x}{y}=\dfrac{2}{3}\)
=> CTHH: Fe2O3
\(n_{Fe}=\dfrac{22,4}{56}=0,4mol\)
\(Fe_xO_y+yH_2\rightarrow xFe+yH_2O\)
\(\dfrac{0,4}{x}\) 0,4
\(\Rightarrow M=\dfrac{32}{\dfrac{0,4}{x}}=80x\)
Mà \(M_{Fe_xO_y}=56x+16y=80x\Rightarrow16y=24x\Rightarrow\dfrac{x}{y}=\dfrac{16}{24}=\dfrac{2}{3}\)
\(\Rightarrow\left\{{}\begin{matrix}x=2\\y=3\end{matrix}\right.\Rightarrow Fe_2O_3\)