Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

a, \(n_{HCl}=0,2.0,1=0,02\left(mol\right)=n_{H^+}=n_{Cl^-}\)
\(n_{H_2SO_4}=0,2.0,15=0,03\left(mol\right)=n_{SO_4^{2-}}\) \(\Rightarrow n_{H^+}=2n_{H_2SO_4}=0,06\left(mol\right)\)
\(\Rightarrow\Sigma n_{H^+}=0,02+0,06=0,08\left(mol\right)\)
\(n_{Ba\left(OH\right)_2}=0,3.0,05=0,015\left(mol\right)=n_{Ba^{2+}}\)
\(\Rightarrow n_{OH^-}=2n_{Ba\left(OH\right)_2}=0,03\left(mol\right)\)
\(H^++OH^-\rightarrow H_2O\)
0,03___0,03 (mol) ⇒ nH+ dư = 0,05 (mol)
\(Ba^{2+}+SO_4^{2-}\rightarrow BaSO_4\)
0,015___0,015______0,015 (mol) ⇒ nSO42- dư = 0,015 (mol)
⇒ m = mBaSO4 = 0,015.233 = 3,495 (g)
\(\left[Cl^-\right]=\dfrac{0,02}{0,2+0,3}=0,04\left(M\right)\)
\(\left[H^+\right]=\dfrac{0,05}{0,2+0,3}=0,1\left(M\right)\)
\(\left[SO_4^{2-}\right]=\dfrac{0,015}{0,2+0,3}=0,03\left(M\right)\)
b, pH = -log[H+] = 1

nH+=0,04 mol nOH-=0,03 mol
H+ + OH- --------> H20
0,04 0,03
0,03 0,03 0,03
0,01
a/ [H+] du=0,01/0,2=0,05 M
[SO42-]=0,01/0,2=0,05 M
[K+]=0,01/0,2=0,05 M
[Ba2+]=0,01/0,2=0,05M
b/ nH+ du=0,01/0,2=0,05 M
pH=-log(0,05)=1,3
c/ khoi luong chat ran thu duoc sau phan ung la
mcr= mSO42- + mK+ + mBa2+
=0,01.96+0,01.39+0,01.137
=2,72g
ta có : \(\Sigma n_{H^+}=n_{HCl}+2n_{H_2SO_4}=0,04\left(mol\right)\)
\(\Sigma n_{OH^-}=n_{KOH}+2n_{Ba\left(OH\right)_2}=0,03\left(mol\right)\)
\(n_{SO_4^{2-}}=0,01\left(mol\right)\) ; \(n_{Ba^{2+}}=0,01\left(mol\right)\)
a, PT : \(H^++OH^-\rightarrow H_2O\)
0,03 0,03 0,03 (mol)
\(\Rightarrow n_{H^+}dư=0,01\left(mol\right)\)
đến đây tự tính đc nha. dùng ct \(CM=\dfrac{n}{V}\)
b, \(PH=-log[H^+]=-log\left(\dfrac{0,01}{0,2}\right)\simeq1,3\)
c, \(Ba^{2+}+SO_4^{2-}\rightarrow BaSO_4\downarrow\)
0,01 0,01 0,01 (mol)\(mcr=m\downarrow+m_{K^+}=m_{BaSO_4}+m_{K+}=\left(0,01\times233\right)+\left(0,01\times39\right)=2,72\left(g\right)\)

a) trong 100 ml dung dịch HCl và H2SO4
CM[H+]=[Cl-]=0,02 M
[SO4 2-]=0,01M
[H+] =2.0,01=0,02 M
trong 100ml dung dịch KOH và Ba(OH)2
[K+]=[OH-]=0,01M
[Ba2+]=0,01M
[OH-]=0,02M
b)n(H+)=0,02+0,02=0,04mol
n(OH-)=0,01+0,02=0,03mol
khi trộn : H+ + OH- =>H2O
0,03<--0,03
=> nH+ dư=0,01mol
=> [H+]=0,05M
=> pH=-lg(0,05)=1,3

a)
$KOH + HCl \to KCl + H_2O$
$n_{KOH} = 0,3(mol) < n_{HCl} = 1,05(mol)$ nên HCl dư
$n_{HCl\ dư} = 1,05 -0 ,3 = 0,75(mol)$
$n_{KCl} = n_{KOH} = 0,3(mol)$
$V_{dd} = 0,3+ 0,7 = 1(lít)$
Suy ra :
$[K^+] = \dfrac{0,3}{1} = 0,3M$
$[Cl^-] = \dfrac{0,75 + 0,3}{1} = 1,05M$
$[H^+] = \dfrac{0,75}{1} = 0,75M$
b)
$Ba(OH)_2 + 2HCl \to BaCl_2 + 2H_2O$
$n_{Ba(OH)_2} = \dfrac{1}{2}n_{HCl} = 0,375(mol)$
$V_{Ba(OH)_2} = \dfrac{0,375}{1,5} = 0,25(lít)$
\(n_{KOH}=0.3\cdot1=0.3\left(mol\right)\)
\(n_{HCl}=0.7\cdot1.5=1.05\left(mol\right)\)
\(KOH+HCl\rightarrow KCl+H_2O\)
\(0.3...........0.3..........0.3\)
Dung dịch D gồm : 0.3 (mol) KCl , 0.75 (mol) HCl dư
\(\left[K^+\right]=\dfrac{0.3}{0.3+0.7}=0.3\left(M\right)\)
\(\left[Cl^-\right]=\dfrac{0.3+0.75}{0.3+0.7}=1.05\left(M\right)\)
\(\left[H^+\right]=\dfrac{0.75}{0.3+0.7}=0.75\left(M\right)\)
\(Ba\left(OH\right)_2+2HCl\rightarrow BaCl_2+2H_2O\)
\(0.375..................0.75\)
\(V_{dd_{Ba\left(OH\right)_2}}=\dfrac{0.375}{1.5}=0.25\left(l\right)\)

a, \(n_{Ba\left(OH\right)_2}=0,1.0,1=0,01\left(mol\right)=n_{Ba^{2+}}\)
\(\Rightarrow n_{OH^-}=2n_{Ba\left(OH\right)_2}=0,02\left(mol\right)\)
\(n_{NaOH}=0,1.0,1=0,01\left(mol\right)=n_{Na^+}=n_{OH^-}\)
⇒ ΣnOH- = 0,02 + 0,01 = 0,03 (mol)
\(n_{H_2SO_4}=0,4.0,0175=0,007\left(mol\right)=n_{SO_4^{2-}}\)
\(\Rightarrow n_{H^+}=2n_{H_2SO_4}=0,014\left(mol\right)\)
\(H^++OH^-\rightarrow H_2O\)
0,014___0,014 (mol) ⇒ nOH- dư = 0,03 - 0,014 = 0,016 (mol)
\(Ba^{2+}+SO_4^{2-}\rightarrow BaSO_4\)
0,007____0,007_____0,007 (mol) ⇒ nBa2+ dư = 0,01 - 0,007 = 0,003 (mol)
⇒ m = 0,007.233 = 1,631 (g)
\(\left[OH^-\right]=\dfrac{0,016}{0,1+0,4}=0,032\left(M\right)\)
\(\left[Ba^{2+}\right]=\dfrac{0,003}{0,1+0,4}=0,006\left(M\right)\)
\(\left[Na^+\right]=\dfrac{0,01}{0,1+0,4}=0,02\left(M\right)\)
b, pH = 14 - (-log[OH-]) ≃ 12,505
\(n_{Ba^{2+}}=0,1.0,1=0,01\left(mol\right)\)
\(n_{SO_4^{2-}}=0,4.0,0175=7.10 ^{-3}\left(mol\right)\)
\(Ba^{2+}+SO_4^{2-}\rightarrow BaSO_4\downarrow\)
\(\Rightarrow m=m_{BaSO_4}=7.10^{-3}.233=1,631\left(g\right)\)
Ta có:
\(n_{H^+}=0,4.0,0175.2=0,014\left(mol\right)\)
\(n_{OH^-}=0,1.0,1.2+0,1.0,1=0,03\left(mol\right)\)
Trong dung dịch X:
\(n_{OH^-}=0,03-0,014=0,016\left(mol\right)\)\(\Rightarrow\left[OH^-\right]=\dfrac{0,016}{0,1+0,4}=0,032\left(M\right)\)
\(n_{Ba^{2+}}=0,01-7.10^{-3}=3.10^{-3}\left(mol\right)\Rightarrow\left[Ba^{2+}\right]=\dfrac{3.10^{-3}}{0,1+0,4}=6.10^{-3}\left(M\right)\)
\(n_{Na^+}=0,1.0,1=0,01\left(mol\right)\Rightarrow\left[Na^+\right]=0,02\)
\(pOH=-lg\left(0,032\right)\approx1,5\Rightarrow pH=14-1,5=12,5\)

\(n_{HCl}=Cm.V=1.0,1=1mol\)
\(n_{H_2SO_4}=Cm.V=0,5.0,1=0,05mol\)
Thể thích của dd D là 200ml = 0,2l
\([H^+]=\frac{n_{HCl}+2.n_{H_2SO_4}}{V}=\frac{0,1+0,1}{0,2}=1M\)
\([Cl^-]=\frac{n_{HCl}}{V}=\frac{0,1}{0,2}=0,5M\)
\([SO_4^{2-}]=\frac{n_{H_2SO_4}}{V}=\frac{0,05}{0,2}=0,25M\)
Khi cho dd D vào \(Ba\left(OH\right)_2\) chỉ có \(H_2SO_4\) tác dụng, tạo kết tủa
\(H_2SO_4+Ba\left(OH\right)_2\rightarrow BaSO_4+2H_2O\)
\(0,05....\rightarrow0,05mol\)
\(\rightarrow m_{BaSO_4}=n.M=0,05.233=11,65g\)

Đáp án A
Có n H C l = 0 , 15 ; n C O 2 = 0 , 045 ; n B a C O 3 = 0 , 15
Gọi n N a 2 C O 3 = a ; n N a H C O 3 = b
Các phản ứng xảy ra theo thứ tự sau:
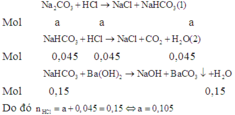
Áp dụng định luật bảo toàn nguyên tố C, ta có:
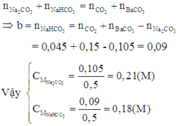
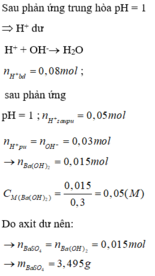
\(n_{Ba\left(OH\right)_2}=0,3.0,1=0,03\left(mol\right)\\ n_{HCl}=0,2.0,15=0,03\left(mol\right)\\ Ba\left(OH\right)_2+2HCl\rightarrow BaCl_2+2H_2O\\ Vì:\dfrac{n_{Ba\left(OH\right)_2\left(đề\right)}}{n_{Ba\left(OH\right)_2\left(PTHH\right)}}=\dfrac{0,03}{1}>\dfrac{n_{HCl\left(đề\right)}}{n_{HCl\left(PTHH\right)}}=\dfrac{0,03}{2}\\ \Rightarrow Ba\left(OH\right)_2dư\\ n_{Ba\left(OH\right)_2\left(p.ứ\right)}=\dfrac{n_{HCl}}{2}=\dfrac{0,03}{2}=0,015\\ n_{Ba\left(OH\right)_2\left(dư\right)}=0,03-0,015=0,015\left(mol\right)\\ \left[OH^-\right]=2.\left[Ba\left(OH\right)_2\left(dư\right)\right]=\dfrac{0,015}{0,3+0,2}=0,03\left(M\right)\\ \Rightarrow pH=14+log\left[OH^-\right]=14+log\left[0,03\right]\approx12,477\)
Nồng độ mol/lít các ion trong dd A:
\(\left[OH^-\left(dư\right)\right]=0,06\left(M\right)\left(nt\right)\\\left[Cl^-\right]=2.\left[BaCl_2\right]=2.\left(\dfrac{0,015}{0,5}\right)=0,06\left(M\right)\\ \left[Ba^{2+}\right]=0,03+ 0,03=0,06\left(M\right)\)