Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

a) PTHH: \(Mg+CuSO_4\rightarrow MgSO_4+Cu\)
a_______a________a_____a (mol)
\(Fe+CuSO_4\rightarrow FeSO_4+Cu\)
b_______b_______b_____b (mol)
\(MgSO_4+2NaOH\rightarrow Mg\left(OH\right)_2\downarrow+Na_2SO_4\)
\(FeSO_4+2NaOH\rightarrow Fe\left(OH\right)_2\downarrow+Na_2SO_4\)
\(Mg\left(OH\right)_2\xrightarrow[]{t^o}MgO+H_2O\)
\(4Fe\left(OH\right)_2+O_2\xrightarrow[]{t^o}2Fe_2O_3+4H_2O\)
b) Ta có: \(n_{CuSO_4}=0,3\cdot1=0,3\left(mol\right)=n_{Cu}\)
\(\Rightarrow m_{Fe\left(dư\right)}=24,8-0,3\cdot64=5,6\left(g\right)\) \(\Rightarrow m_{Fe\left(p/ứ\right)}+m_{Mg}=16-5,6=10,4\left(g\right)\)
Ta lập hệ phương trình: \(\left\{{}\begin{matrix}24a+56b=10,4\\a+b=0,3\end{matrix}\right.\) \(\Leftrightarrow\left\{{}\begin{matrix}a=0,2\\b=0,1\end{matrix}\right.\)
\(\Rightarrow\left\{{}\begin{matrix}\%m_{Mg}=\dfrac{0,2\cdot24}{16}\cdot100\%=30\%\\\%m_{Fe}=70\%\end{matrix}\right.\)
c) Theo các PTHH: \(\left\{{}\begin{matrix}n_{MgO}=n_{Mg}=0,2\left(mol\right)\\n_{Fe_2O_3}=\dfrac{1}{2}n_{Fe\left(p/ứ\right)}=0,05\left(mol\right)\end{matrix}\right.\)
\(\Rightarrow m_{rắn}=m_{MgO}+m_{Fe_2O_3}=0,2\cdot40+0,05\cdot160=11\left(g\right)\)

PTHH: \(FeCl_2+2KOH\rightarrow2KCl+Fe\left(OH\right)_2\downarrow\)
a) Ta có: \(\left\{{}\begin{matrix}n_{FeCl_2}=\dfrac{25,4}{127}=0,2\left(mol\right)\\n_{KOH}=\dfrac{28}{56}=0,5\left(mol\right)\end{matrix}\right.\)
Xét tỉ lệ: \(\dfrac{0,2}{1}< \dfrac{0,5}{2}\) \(\Rightarrow\) FeCl2 p/ứ hết, KOH dư
\(\Rightarrow n_{Fe\left(OH\right)_2}=0,2\left(mol\right)\) \(\Rightarrow m_{Fe\left(OH\right)_2}=0,2\cdot90=18\left(g\right)\)
b)
+) Nung trong không khí
PTHH: \(4Fe\left(OH\right)_2+O_2\xrightarrow[]{t^o}2Fe_2O_3+4H_2O\)
Theo PTHH: \(n_{Fe_2O_3}=\dfrac{1}{2}n_{Fe\left(OH\right)_2}=0,1\left(mol\right)\) \(\Rightarrow m_{Fe_2O_3}=0,1\cdot160=16\left(g\right)\)
+) Nung trong chân không
PTHH: \(Fe\left(OH\right)_2\xrightarrow[]{t^o}FeO+H_2O\)
Theo PTHH: \(n_{FeO}=n_{Fe\left(OH\right)_2}=0,2\left(mol\right)\) \(\Rightarrow m_{FeO}=0,2\cdot72=14,4\left(g\right)\)

a)PTHH: \(Ba\left(OH\right)+Na_2CO_3\rightarrow2NaOH+BaCO_3\downarrow\)
\(BaCO_3\underrightarrow{t^o}BaO+CO_2\uparrow\)
Ta có: \(n_{Ba\left(OH\right)_2}=0,4\cdot0,2=0,08\left(mol\right)\)
\(\Rightarrow n_{NaOH}=0,16mol\) \(\Rightarrow C_{M_{NaOH}}=\dfrac{0,16}{0,4}=0,4\left(M\right)\) (Coi Vdd thay đổi không đáng kể)
b) Theo PTHH: \(n_{BaCO_3}=n_{Ba\left(OH\right)_2}=n_{BaO}=0,08mol\) \(\Rightarrow m_{BaO}=0,08\cdot153=12,24\left(g\right)\)

a. Ba(OH)2 +Na2CO3 ➝ BaCO3 + 2NaOH
BaCO3 ➝ BaO + CO2
nBa(OH)2 = 0,08 mol
=> nNaOH = 2nBa(OH)2 = 0,16 mol
=> CM = 0,4 M
b) Bảo toàn Ba: nBaO = nBa(OH)2 = 0,08 mol
=> m = 12,24 g

Đáp án:
m =32,4g
mddH2SO4 = 49g
Giải thích các bước giải:
a) MgCO3 + H2SO4 → MgSO4 + H2O +CO2 ↑
MgSO4 + 2NaOH → Mg(OH)2 + Na2SO4
$Mg{(OH)_2}\buildrel {to} \over
\longrightarrow MgO + {H_2}O$
b) nCO2 = 2,24 : 22,4 = 0,1mol
nMgCO3 = nCO2 = 0,1 mol
nMgO = 12:40=0,3mol
nMgSO4 = nMgO - nMgCO3 = 0,3 - 0,1 = 0,2mol
m = mMgCO3 + mMgSO4
= 0,1 .84+0,2.120=32,4g
nH2SO4 = nCO2 = 0,1 mol
mH2SO4 = 0,1.98=9,8g
mddH2SO4 = 9,8:20.100=49g
chúc bạn học tốt

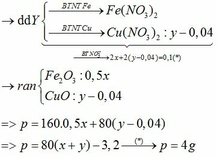
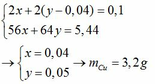
\(n_{FeCl_2}=\dfrac{150\cdot12.7\%}{127}=0.15\left(mol\right)\)
\(n_{NaOH}=\dfrac{350\cdot4\%}{40}=0.35\left(mol\right)\)
\(FeCl_2+2NaOH\rightarrow Fe\left(OH\right)_2+2NaCl\)
\(0.15...........0.3................0.15............0.3\)
\(m_{Fe\left(OH\right)_3}=0.15\cdot90=13.5\left(g\right)\)
\(m_{dd}=150+350-13.5=486.5\left(g\right)\)
\(C\%_{NaCl}=\dfrac{0.3\cdot58.5}{486.5}\cdot100\%=3.61\%\)
\(C\%_{NaOH\left(dư\right)}=\dfrac{\left(0.35-0.3\right)\cdot40}{486.5}\cdot100\%=0.4\%\)
\(Fe\left(OH\right)_2\underrightarrow{^{^{t^0}}}FeO+H_2O\)
\(0.15..........0.15\)
\(m_{FeO}=0.15\cdot72=10.8\left(g\right)\)
\(4Fe\left(OH\right)_2+O_2\underrightarrow{^{^{t^0}}}2Fe_2O_3+4H_2O\)
\(0.15.........................0.075\)
\(m_{Fe_2O_3}=0.075\cdot160=12\left(g\right)\)