Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

Mình thay trên câu a luôn nhé.
5. Số mol của Fe là :
nFe = 5,6/56 = 0,1 (mol)
a) Ta có PTHH :
Fe + 2HCl \(\rightarrow\) FeCl2 + H2\(\uparrow\)
1 mol 2 mol 1 mol 1 mol
0,1 mol 0,2 mol 0,1 mol 0,1 mol
Số mol của Fe là :
nFe = 5,6/56 = 0,1 (mol)
b) Khối lượng của FeCl2 tạo thành sau p.ứng là :
mFeCl2 = 0,1.127 = 12,7 (g)
c) Thể tích khí Hiđro (đktc) tạo thành sau p.ứng là :
VH2 = 0,1.22,4 = 2,24 (l)
4. Công thức của B là : NaxCyOz
+ \(m_{Na}=\frac{106.43,6}{100}\approx46\left(g\right)\)
\(m_C=\frac{106.11,3}{100}\approx12\left(g\right)\)
\(m_O=\frac{106.45,3}{100}\approx48\left(g\right)\)
+ \(n_{Na}=\frac{46}{23}=2\left(mol\right)\)
\(n_C=\frac{12}{12}=1\left(mol\right)\)
\(n_O=\frac{48}{16}=3\left(mol\right)\)
Suy ra trong một p.tử h/c có 2 n.tử Na, 1 n.tử C và 3 n.tử O.
\(\Rightarrow\) CTHH của hợp chất B là Na2CO3.

Đề 15:
1) Theo đề bài , ta có:
NTK(Y)= 2.NTK(Ca)= 2.40=80 (đvC)
=> Nguyên tố Y là brom, KHHH là Br.
2) - Đơn chất là chất do 1nguyên tố tạo thành.
VD: O3; Br2 ; Cl2;......
- Hợp chất là những chất do 2 hay nhiều nguyên tố hóa học trở lên tạo thành.
VD: NaCl, KMnO4, CuSO4, H2O;....
3) Khôi lượng mol? tự trả lời đi !
a) Khối lượng mol của phân tử gồm 2C và 4H
Có nghĩa là khối lượng mol của C2H4
\(M_{C_2H_4}\)= 2.12+4.1=28 (g/mol)
\(M_{H_2S}\)=2.1+32=34(g/mol)

a, PT: \(Fe+2HCl\rightarrow FeCl_2+H_2\)
b, Ta có: \(n_{Fe}=\dfrac{11,2}{56}=0,2\left(mol\right)\)
Theo PT: \(n_{H_2}=n_{Fe}=0,2\left(mol\right)\Rightarrow V_{H_2}=0,2.22,4=4,48\left(l\right)\)
c, Theo PT: \(n_{HCl}=2n_{Fe}=0,4\left(mol\right)\)
\(\Rightarrow m_{HCl}=0,4.36,5=14,6\left(g\right)\)
\(Fe+2HCl\rightarrow FeCl_2+H_2\)
\(1mol\) \(2mol\) \(1mol\)
\(0,2mol\) \(0,4mol\) \(0,2mol\)
\(n_{Fe}=\dfrac{m}{M}=\dfrac{11,2}{56}=0,2\left(mol\right)\)
\(V_{H_2}=n.22,4=0,2.22,4=4,48\left(l\right)\)
\(m_{HCl}=n.M=0,4.=14,6\left(g\right)\)

\(Fe+2HCl\rightarrow FeCl_2+H_2\\ n_{Fe}=\dfrac{14}{56}=0,25\left(mol\right)\\ n_{H_2}=n_{Fe}=0,25\left(mol\right)\\ V_{H_2}=0,25.22,4=5,6\left(l\right)\\ n_{HCl}=2n_{Fe}=0,5\left(mol\right)\\ m_{HCl}=0,5.36,5=18,25\left(g\right)\)

1. \(Zn+2HCl\rightarrow ZnCl_2+H_2\)
2. \(n_{Zn}=\dfrac{13}{65}=0,2\left(mol\right)\)
Theo PT: \(n_{ZnCl_2}=n_{H_2}=n_{Zn}=0,2\left(mol\right)\Rightarrow m_{ZnCl_2}=0,2.136=27,2\left(g\right)\)
3. Ta có: \(n_{Fe_3O_4}=\dfrac{23,2}{232}=0,1\left(mol\right)\)
PT: \(Fe_3O_4+4H_2\underrightarrow{^{t^o}}3Fe+4H_2O\)
Xét tỉ lệ: \(\dfrac{0,1}{1}>\dfrac{0,2}{4}\), ta được Fe3O4 dư.
Theo PT: \(n_{Fe_3O_4\left(pư\right)}=\dfrac{1}{4}n_{H_2}=0,05\left(mol\right)\Rightarrow n_{Fe_3O_4\left(dư\right)}=0,1-0,05=0,05\left(mol\right)\)
\(\Rightarrow m_{Fe_3O_4\left(dư\right)}=0,05.232=11,6\left(g\right)\)
1. \(Zn+2HCl\rightarrow ZnCl_2+H_2\)
2. \(n_{zn}=\dfrac{m_{Zn}}{M_{Zn}}=\dfrac{13}{65}=0,2\left(mol\right)\)
Theo PTHH: \(n_{ZnCl_2}=n_{Zn}=0,2\left(mol\right)\)
\(\Rightarrow m_{ZnCl_2}=n_{ZnCl_2}.M_{ZnCl_2}=0,2.136=27,2\left(g\right)\)
\(\Rightarrow n_{H_2}=n_{Zn}=0,2\left(mol\right)\)
3. \(2H_2+Fe_3O_4\rightarrow3Fe+2H_2O\)
2 mol------1 mol------3 mol--2 mol
\(n_{Fe_3O_4}=\dfrac{m_{Fe_3O_4}}{M_{Fe_3O_4}}=\dfrac{23,2}{232}=0,1\left(mol\right)\)
\(\dfrac{n_{Fe_3O_4}}{1}=\dfrac{0,1}{1}\)
\(\dfrac{n_{H_2}}{2}=\dfrac{0,2}{2}\)
\(\dfrac{n_{Fe_3O_4}}{1}=\dfrac{n_{H_2}}{2}\)
Vậy không có chất nào dư cả

a,\(n_{Fe}=\dfrac{11,2}{56}=0,2\left(mol\right)\)
PTHH: Fe + 2HCl → FeCl2 + H2
Mol: 0,2 0,4 0,2
b, \(V_{H_2}=0,2.22,4=4,48\left(l\right)\)
c, \(m_{HCl}=0,4.36,5=14,6\left(g\right)\)

\(n_{Fe}=\dfrac{11.2}{56}=0.2\left(mol\right)\)
\(Fe+2HCl\rightarrow FeCl_2+H_2\)
\(0.2.......0.4....................0.2\)
\(V_{H_2}=0.2\cdot22.4=4.48\left(l\right)\)
\(m_{HCl}=0.4\cdot36.5=14.6\left(g\right)\)
PTHH : \(Fe+2HCl-->FeCl_2+H_2\uparrow\) (1)
\(n_{Fe}=\dfrac{m}{M}=\dfrac{11.2}{56}=0.2\left(mol\right)\)
Từ (1) => \(n_{Fe}=n_{H_2}=0.2\left(mol\right)\)
=> \(V_{H2\left(đktc\right)}=n.22,4=0,2.22,4=4,48\left(l\right)\)
Từ (1) => \(2n_{Fe}=n_{HCl}=0.4\left(mol\right)\)
=> \(m_{HCl}=n.M=0,4.\left(1+35.5\right)=14.6\left(g\right)\)

nFe = 16,8/56 = 0,3 (mol)
PTHH: Fe + 2HCl -> FeCl2 + H2
Mol: 0,3 ---> 0,6 ---> 0,3 ---> 0,3
VH2 = 0,3 . 24,79 = 7,437 (l)
mHCl = 0,6 . 36,5 = 21,9 (g)
PTHH: CuO + H2 -> (t°) Cu + H2O
Mol: 0,3 <--- 0,3 ---> 0,3
mCu = 0,3 . 64 = 19,2 (g)
mFe = 16,8: 56 =0,3(mol)
pthh : Fe + 2HCl --> FeCl2 + H2 (1)
0,3 ->0,6-----------------> 0,3 (mol)
=> VH2 (đkc) = 0,3 . 24,79 ( l)
=> mHCl = 0,6 . 35,5 = 21,9 (g)
pthh : CuO + H2 -t--> Cu+ H2O
0,3<-----0,3 (mol)
=>mCu = 0,3 . 64 = 19,2 (g)
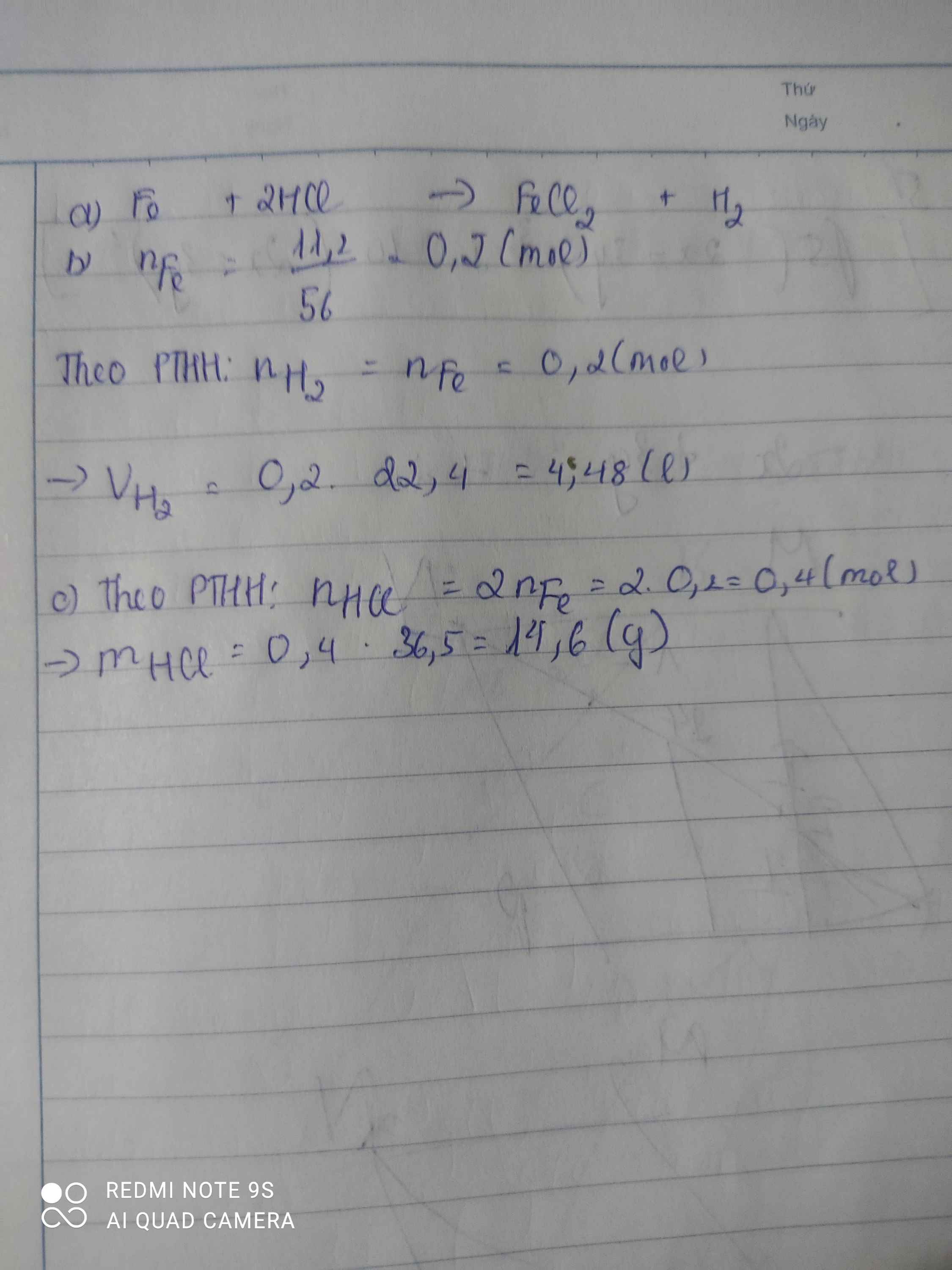
a) \(\left\{{}\begin{matrix}n_{Fe}=\dfrac{11,2}{56}=0,2\left(mol\right)\\n_{HCl}=\dfrac{10,95}{36,5}=0,3\left(mol\right)\end{matrix}\right.\)
PTHH: \(Fe+2HCl\rightarrow FeCl_2+H_2\)
Xét tỉ lệ: \(0,2>\dfrac{0,3}{2}\Rightarrow\) Fe dư
Theo PTHH: \(n_{Fe\left(p\text{ư}\right)}=n_{H_2}=\dfrac{1}{2}n_{HCl}=0,15\left(mol\right)\)
\(\Rightarrow m_{Fe\left(d\text{ư}\right)}=\left(0,2-0,15\right).56=2,8\left(g\right)\)
c) \(V_{H_2}=0,15.22,4=3,36\left(l\right)\)