Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

\(n_{Mg}=\dfrac{2,4}{24}=0,1\left(mol\right)\\ a,2Mg+O_2\rightarrow\left(t^o\right)2MgO\\ n_{O_2}=\dfrac{0,1}{2}=0,05\left(mol\right)\\ V_{O_2\left(đktc\right)}=0,05.22,4=1,12\left(l\right)\\ b,2KClO_3\rightarrow\left(t^o\right)2KCl+3O_2\\ n_{KClO_3}=\dfrac{0,05.2}{3}=\dfrac{1}{30}\left(mol\right)\\ \Rightarrow m_{KClO_3}=\dfrac{122,5}{30}=\dfrac{49}{12}\left(g\right)\)

nMg = 9,6/24 = 0,4 (mol)
2Mg + O2 ---to---> 2MgO
0,4____0,2_________0,4
VO2(đktc) = 0,2.22,4 = 4,48(l)
mMgO = 0,4.40 = 16(g)

mMg = 3.6/24 = 0.15 (mol)
2Mg + O2 -to-> 2MgO
0.15__0.075____0.15
mMgO= 0.15*40 = 6 (g)
VO2 = 0.075*22.4 = 1.68 (l)
2KClO3 -to-> 2KCl + 3O2
0.05_______________0.075
mKClO3 = 0.05*122.5 = 6.125 (g)
PTHH: \(2Mg+O_2\underrightarrow{t^o}2MgO\)
a+b) Ta có: \(n_{Mg}=\dfrac{3,6}{24}=0,15\left(mol\right)\)
\(\Rightarrow\left\{{}\begin{matrix}n_{O_2}=0,075\left(mol\right)\\n_{MgO}=0,15\left(mol\right)\end{matrix}\right.\) \(\Rightarrow\left\{{}\begin{matrix}V_{O_2}=0,075\cdot22,4=1,68\left(l\right)\\m_{MgO}=0,15\cdot40=6\left(g\right)\end{matrix}\right.\)
c) PTHH: \(2KClO_3\xrightarrow[MnO_2]{t^o}2KCl+3O_2\uparrow\)
Theo PTHH: \(n_{KClO_3}=0,05\left(mol\right)\)
\(\Rightarrow m_{KClO_3}=0,05\cdot122,5=6,125\left(g\right)\)

PTHH: \(2Mg+O_2\underrightarrow{t^o}2MgO\)
Gộp cả phần a và b
Ta có: \(n_{Mg}=\dfrac{12}{24}=0,5\left(mol\right)\)
\(\Rightarrow\left\{{}\begin{matrix}n_{O_2}=0,25mol\\n_{MgO}=0,5mol\end{matrix}\right.\) \(\Rightarrow\left\{{}\begin{matrix}m_{O_2}=0,25\cdot22,4=5,6\left(l\right)\\m_{MgO}=0,5\cdot40=20\left(g\right)\end{matrix}\right.\)

a)\(2Mg + O_2 \xrightarrow{t^o} 2MgO\)
b)
\(n_{Mg} = \dfrac{2,4}{24} = 0,1(mol)\)
Theo PTHH :
\(n_{O_2} = \dfrac{1}{2}n_{Mg} = 0,05(mol)\\ \Rightarrow V_{O_2} = 0,05.22,4 = 1,12(lít)\)
c)
\(n_{MgO} = n_{Mg} = 0,1(mol)\\ \Rightarrow m_{MgO} = 0,1.40 = 4(gam)\)
d)
\(V_{không\ khí} = 5V_{O_2} = 1,12.5 = 5,6(lít)\)

a, 2Mg + O2 \(\underrightarrow{t^o}\) 2MgO
b, \(n_{Mg}=\dfrac{4,8}{24}=0,2mol\)
\(n_{O_2}=\dfrac{0,2}{2}=0,1mol\)
\(m_{O_2}=0,1.32=3,2g\)
\(V_{O_2}=0,1.22,4=2,24l\)
c, Cách 1:
\(Theo.ĐLBTKL,ta.có:\\ m_{Mg}+m_{O_2}=m_{MgO}\)
\(\Rightarrow m_{MgO}=4,8+3,2=8g\)
Cách 2:
\(n_{MgO}=\dfrac{0,2.2}{2}=0,2mol\)
\(\Rightarrow m_{MgO}=0,2.40=8g\)

a)
\(n_{Mg}=\dfrac{12}{24}=0,5\left(mol\right)\)
PTHH: 2Mg + O2 --to--> 2MgO
______0,5-->0,25---->0,5
=> VO2 = 0,25.22,4 = 5,6 (l)
=> mMgO = 0,5.40 = 20 (g)
b)
\(n_{O_2}=0,25=>n_{CO_2}=0,25\)
=> mCO2 = 0,25.44 = 11 (g)

$a\big)$
$n_{Fe}=\frac{16,8}{56}=0,3(mol)$
$3Fe+2O_2\xrightarrow{t^o}Fe_3O_4$
Theo PT: $n_{Fe_3O_4}=\frac{1}{3}n_{Fe}=0,1(mol)$
$\to m_{Fe_3O_4}=0,1.232=23,2(g)$
$b\big)$
Theo PT: $n_{O_2}=\frac{2}{3}n_{Fe}=0,2(mol)$
$\to V_{O_2}=0,2.22,4=4,48(l)$
$\to V_{kk}=4,48.5=22,4(l)$
$c\big)$
$2KMnO_4\xrightarrow{t^o}K_2MnO_4+MnO_2+O_2$
Theo PT: $n_{KMnO_4}=2n_{O_2}=0,4(mol)$
$\to m_{KMnO_4(dùng)}=\frac{0,4.158}{80\%}=79(g)$
a, \(n_{Fe}=\dfrac{16,8}{56}=0,3\left(mol\right)\)
PTHH: 3Fe + 2O2 ---to→ Fe3O4
Mol: 0,3 0,2 0,1
\(m_{Fe_3O_4}=0,1.232=23,2\left(g\right)\)
b, \(V_{O_2}=0,2.22,4=4,48\left(l\right)\Rightarrow V_{kk}=4,48.5=22,4\left(l\right)\)
c,
PTHH: 2KMnO4 ---to→ K2MnO4 + MnO2 + O2
Mol: 0,4 0,2
\(m_{KMnO_4\left(lt\right)}=0,4.158=63,2\left(g\right)\)
\(\Rightarrow m_{KMnO_4\left(tt\right)}=\dfrac{63,2}{80\%}=79\left(g\right)\)

a) 2Mg + O2 --to--> 2MgO
b) \(m_{MgO}=2,4.1,667=4\left(g\right)\)
Theo ĐLBTKL: mMg + mO2 = mMgO
=> mO2 = 4-2,4 = 1,6(g)
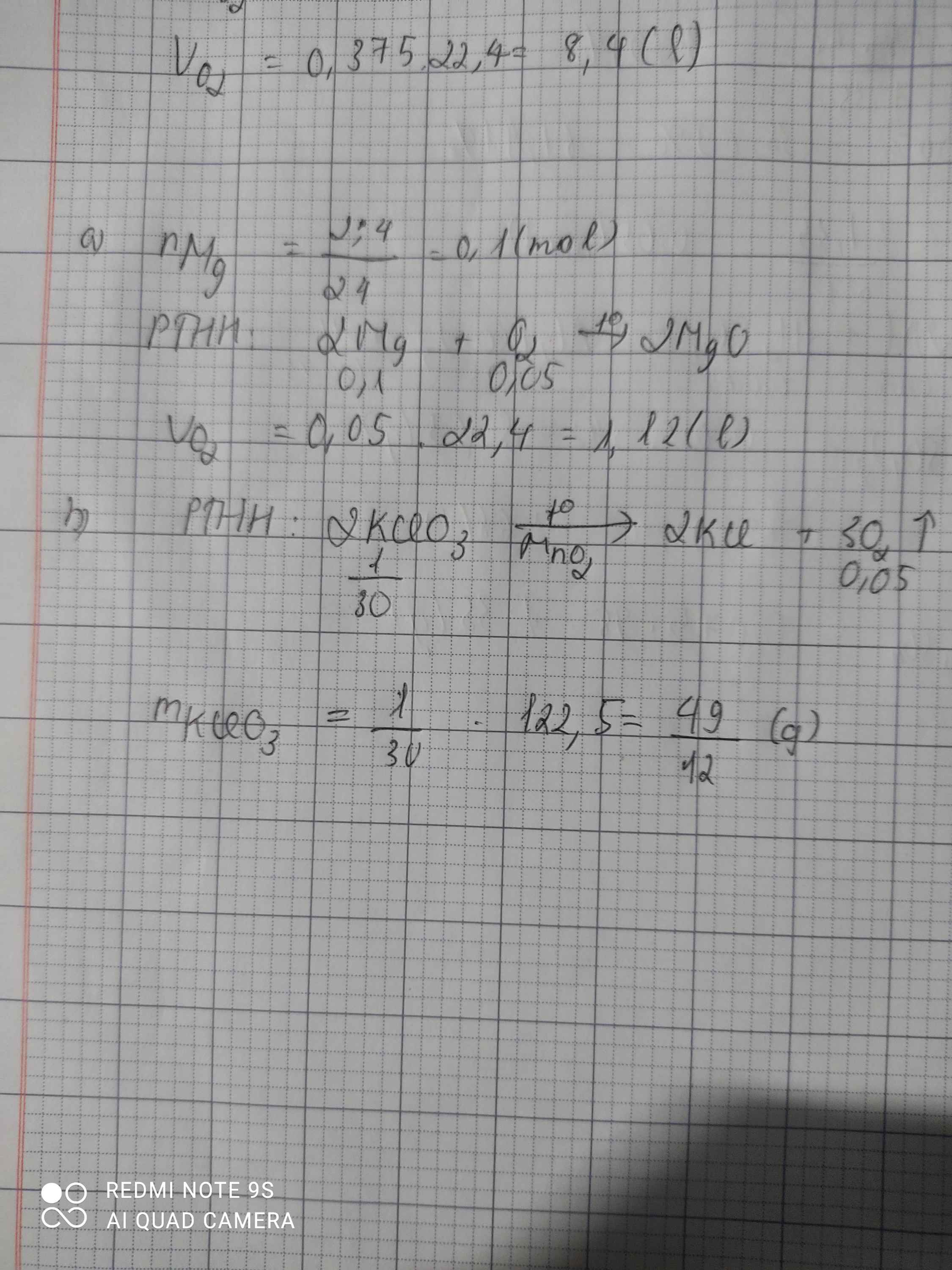
2Mg+O2-to>2MgO
0,1-----0,05-----0,1
n Mg=\(\dfrac{2,4}{24}\)=0,1 mol
=>VO2=0,05.22,4=1,12l
=>m MgO=0,1.40=4g
\(n_{Mg}=\dfrac{2,4}{24}=0,1\left(mol\right)\)
pthh : 2Mg + O2 -t-> 2MgO
0,1 0,05 0,1
=> VO2 = 0,05 . 22,4 = 1,12 (l)
=> mMgO = 0,1 .40 = 4 (g)