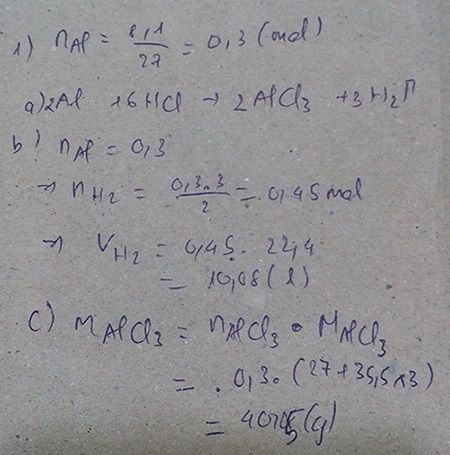Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

\(a,PTHH:Fe+2HCl\rightarrow FeCl_2+H_2\\ n_{Fe}=\dfrac{22,4}{56}=0,4\left(mol\right)\\ b,n_{FeCl_2}=n_{H_2}=n_{Fe}=0,4\left(mol\right)\\ V_{H_2\left(đktc\right)}=0,4.22,4=8,96\left(l\right)\\ c,m_{FeCl_2}=127.0,4=50,8\left(g\right)\)

Phản ứng thuộc phản ứng oxh- khử
VHCl= 400ml => VHCl= 0,4 lít
mZn= 1.3g => nZn= mZn/MZn = 1.3/65 = 0.02(mol)
Zn + 2HCl ---> ZnCl2 + H2
1 2 1 1
mol: 0.02 0.04 0.02 0.02
a) mZnCl2 = nZnCl2 . MZnCl2 = (0.02)( 65+ 35,5.2) =2,72g
b) VH2= nH2. 22,4 = 0.02 . 22.4 = 0.448(lít)
c) CM(HCl)= nHCl/VHCl = 0.04/0.4 = 0,1 M

2Al + 6HCl → 2AlCl3 + 3H2
2 6 2 3
0,3 0,9 0,3 0,45
a). nAl= \(\dfrac{8,1}{27}\)=0,3(mol)
⇒ nHCl= \(\dfrac{0,3.3}{6}\)= 0,9(mol).
⇒ mHCl=n.M= 0,9 . 36.5 =32,85(g).
b). nAlCl3= \(\dfrac{0,9.2}{6}\)= 0,3(mol).
⇒mAlCl3= n.M = 0,3 . 133,5 =40,05(g).
c). nH2= \(\dfrac{0,3.3}{2}\)= 0,45(mol).
⇒VH2= n . 22,4 = 0,45 . 22,4= 10,08(g).

\(Pt: Fe + 2HCl \rightarrow FeCl_2 + H_2\)
\(a.n_{Fe}=\dfrac{11,2}{56}=0,2\left(mol\right)\)
Theo pt: \(nH_2 = nFe = 0,2 mol\)
\(\Rightarrow V_{H_2}=0,2.22,4=4,48l\)
\(b.n_{FeCl_2}=n_{Fe}=0,2\left(mol\right)\)
\(\Rightarrow m_{FeCl_2}=0,2.127=25.4g\)
\(c.n_{HCl}=2nFe=0,4mol\)
\(C_MHCl=\dfrac{0,4}{0,1}=4M\)

a,\(n_{Zn}=\dfrac{6,5}{65}=0,1\left(mol\right)\)
PTHH: Zn + 2HCl → ZnCl2 + H2
Mol: 0,1 0,2 0,1 0,1
b,\(V_{H_2}=0,1.24,79=2,479\left(l\right)\)
c,\(m_{ddHCl}=\dfrac{0,2.36,5.100}{3,65}=200\left(g\right)\)
d,\(m_{ZnCl_2}=0,1.136=13,6\left(g\right)\)
e,mdd sau pứ = 6,5+200-0,1.2 = 206,3 (g)
\(C\%_{ddZnCl_2}=\dfrac{13,6.100\%}{206,3}=6,59\%\)

a, \(n_{CO_2}=\dfrac{0,56}{22,4}=0,025\left(mol\right)\)
PT: \(2CH_3COOH+Na_2CO_3\rightarrow2CH_3COONa+CO_2+H_2O\)
Theo PT: \(n_{CH_3COOH}=2n_{CO_2}=0,05\left(mol\right)\)
\(\Rightarrow C\%_{CH_3COOH}=\dfrac{0,05.60}{100}.100\%=3\%\)
b, Theo PT: \(n_{Na_2CO_3}=n_{CO_2}=0,025\left(mol\right)\Rightarrow m_{Na_2CO_3}=0,025.106=2,65\left(g\right)\)
\(n_{CH_3COONa}=2n_{CO_2}=0,05\left(mol\right)\Rightarrow m_{CH_3COONa}=0,05.82=4,1\left(g\right)\)
c, \(C_2H_5OH+O_2\underrightarrow{^{mengiam}}CH_3COOH+H_2O\)
Theo PT: \(n_{C_2H_5OH\left(LT\right)}=n_{CH_3COOH}=0,05\left(mol\right)\)
Mà: H = 80%
\(\Rightarrow n_{C_2H_5OH\left(TT\right)}=\dfrac{0,05}{80\%}=0,0625\left(mol\right)\)
\(\Rightarrow m_{C_2H_5OH\left(TT\right)}=0,0625.46=2,875\left(g\right)\)
\(\Rightarrow V_{C_2H_5OH}=\dfrac{2,875}{0,8}=3,59375\left(ml\right)\)
\(\Rightarrow V_{C_2H_5OH\left(10^o\right)}=\dfrac{3,59375}{10}.100=35,9375\left(ml\right)\)

a, Ta có: \(n_{Na_2SO_3}=\dfrac{6,3}{126}=0,05\left(mol\right)\)
\(n_{Ca\left(OH\right)_2}=0,1.1=0,1\left(mol\right)\)
PT: \(Na_2SO_3+2HCl\rightarrow2NaCl+H_2O+SO_2\)
_____0,05__________________________0,05 (mol)
Xét tỉ lệ: \(\dfrac{n_{SO_2}}{n_{Ca\left(OH\right)_2}}=0,5< 1\)
⇒ Tạo muối CaSO3.
PT: \(SO_2+Ca\left(OH\right)_2\rightarrow CaSO_3+H_2O\)
____0,05_______________0,05 (mol)
b, \(V_{SO_2}=0,05.22,4=1,12\left(l\right)\)
c, \(m_{CaSO_3}=0,05.120=6\left(g\right)\)
Bạn tham khảo nhé!

\(a.Fe+2HCl\rightarrow FeCl_2+H_2\\b.n_{Fe}=\dfrac{5,6}{56}=0,1\left(mol\right)\\ n_{H_2}=n_{Fe}=0,1\left(mol\right)\\ \Rightarrow V_{H_2}=0,1.22,4=2,24\left(l\right)\\ c.n_{FeCl_2}=n_{Fe}=0,1\left(mol\right)\\ m_{FeCl_2}=0,1.127=12,7\left(g\right) \)

a, PT: \(Na_2SO_3+2HCl\rightarrow2NaCl+SO_2+H_2O\)
Ta có: \(n_{Na_2SO_3}=\dfrac{12,6}{126}=0,1\left(mol\right)\)
Theo PT: \(n_{SO_2}=n_{Na_2SO_3}=0,1\left(mol\right)\)
\(\Rightarrow V_{SO_2}=0,1.22,4=2,24\left(l\right)\)
b, Theo PT: \(n_{NaCl}=n_{HCl}=2n_{Na_2SO_3}=0,2\left(mol\right)\)
\(\Rightarrow m_{NaCl}=0,2.58,5=11,7\left(g\right)\)
c, \(m_{HCl}=0,2.36,5=7,3\left(g\right)\)
\(\Rightarrow m_{ddHCl}=\dfrac{7,3}{10\%}=73\left(g\right)\)
d, Ta có: m dd sau pư = 12,6 + 73 - 0,1.64 = 79,2 (g)
\(\Rightarrow C\%_{NaCl}=\dfrac{11,7}{79,2}.100\%\approx14,77\%\)